Efficacy and safety of anlotinib combined with etoposide and platinum-based regimens in the first-line treatment of extensive-stage small-cell lung cancer: a systematic review and meta-analysis
Highlight box
Key findings
• Anlotinib in combination with etoposide and platinum-based regimens was evaluated as first-line therapy for extensive-stage small-cell lung cancer (ES-SCLC). Pooled analysis revealed an objective response rate of 83.0%, with pooled median progression-free survival and overall survival (OS) of 7.29 and 14.99 months, respectively. These results indicate that anlotinib in combination with etoposide and platinum-based regimens has potentially promising efficacy and safety in the first-line treatment of ES-SCLC.
What is known and what is new?
• Anlotinib has obtained approval for the treatment of patients diagnosed with refractory ES-SCLC in China.
• The efficacy and safety of anlotinib in untreated patients with ES-SCLC remain to be determined.
What is the implication, and what should change now?
• Our meta-analysis demonstrated a potentially promising clinical efficacy for OS, suggesting that anlotinib in combination with etoposide and platinum-based therapy could be an effective option for first-line ES-SCLC treatment. However, further large-scale studies enrolling ethnically diverse patient populations and additional randomized controlled trials are warranted to validate these findings.
Introduction
Lung cancer is the leading type of cancer in terms of incidence and mortality rate worldwide (1), and small-cell lung cancer (SCLC) subtypes account for approximately 15% of lung cancer cases (2,3). This type of cancer is characterized by potential for early metastases and strong invasiveness; consequently, approximately 70% of SCLC patients are diagnosed with extensive disease (4,5), which is associated with poor prognosis (5-year survival rate: <7%) (6). It has been reported that the median overall survival (mOS) of patients with extensive-stage SCLC (ES-SCLC) undergoing standard chemotherapy is about 10 months (7,8).
For decades, platinum-etoposide chemotherapy has been the standard first-line treatment for SCLC (9). Despite an initial good response rate to platinum-etoposide chemotherapy, the mOS of SCLC patients remains around 10 months (10). Recently, immune checkpoint inhibitor (ICI) immunotherapy demonstrated clinical activity in the treatment of ES-SCLC (11,12), hence, it has been recommended to use this type of therapy as first-line treatment (13,14). The CASPIAN study results demonstrated that durvalumab in combination with platinum-etoposide significantly improved overall survival (OS) in the first-line treatment of ES-SCLC (7,15,16). However, due to minor improvements in objective response rate (ORR) and progression-free survival (PFS), only a limited survival benefit was obtained, with a mOS of 12.9 months. Hence, there are unmet medical needs in the first-line treatment of ES-SCLC.
Anlotinib is a novel multitarget tyrosine kinase inhibitor, which directly targets endothelial growth factor receptor (EGFR), vascular endothelial growth factor receptor 1–3 (VEGFR1–3), fibroblast growth factor receptor 1–4 (FGFR1–4), and stem cell factor receptor to inhibit tumour angiogenesis (17). The ALTER 1202 study involved 120 Chinese patients with advanced SCLC who received anlotinib as third-line or further-line treatment. The results suggested that, compared with placebo, anlotinib improved PFS (0.7 vs. 4.3 months, respectively; hazard ratio (HR) =0.19, P<0.0001) and OS (4.9 vs. 7.3 months, respectively; HR =0.53, P=0.0029). Moreover, anlotinib treatment was associated with common and acceptable adverse reactions (18). Consequently, anlotinib has obtained approval for the treatment of patients diagnosed with refractory ES-SCLC in China (19).
However, the efficacy and safety of anlotinib in untreated patients with ES-SCLC remain to be determined. Thus, the aim of this systematic review and meta-analysis was to assess the efficacy and safety of anlotinib in combination with platinum-etoposide in the first-line therapy of patients with ES-SCLC. We present this article in accordance with the PRISMA reporting checklist (available at https://jtd.amegroups.com/article/view/10.21037/jtd-2025-1813/rc) (20).
Methods
Inclusion and exclusion criteria
Patients with histologically diagnosed SCLC with Eastern Cooperative Oncology Group (ECOG) performance status score of ≤2, previously untreated for ES-SCLC, were included. No specific restrictions were imposed with regard to factors such as sex, race, or smoking history. Patients were treated with anlotinib combined with platinum-etoposide chemotherapy. Phase II clinical trials or retrospective analyses were included. The outcomes included ORR, PFS, OS, and adverse events (AEs). Reviews, meta-analyses, and studies involving animal experiments were excluded.
Outcome assessment
The main outcomes were the OS, PFS, and ORR of patients with ES-SCLC after treatment. The secondary outcome was the AEs associated with the therapy.
Search strategy and selection
A systematic literature search of Embase, PubMed, Web of Science, and the Cochrane Library was conducted to identify relevant studies published prior to 25 February 2025. Only reports published in English were included in this analysis. The medical subject heading terms and combined text used were: “Carcinoma, Small Cell Lung” and “anlotinib”. The complete search strategy is shown in Appendix 1.
Data extraction
All procedures were conducted independently by two researchers, and any discrepancies were addressed through discussion with a third researcher. The extracted information included the following: first author, year of publication, region, participant characteristics (e.g., age, sex, ethnic origin), number of participants, anlotinib dose, baseline metastatic sites of patients, ECOG performance status scores, the proportion of carboplatin selection, description and doses of therapeutic agents administered, and frequency and types of AEs. The efficacy criteria analyzed were PFS, OS, ORR, and safety.
Assessing the risk of bias and grading quality of evidence
Two researchers conducted independent evaluation and extraction of the required data from all included studies. The revised Methodological Index for Non-Randomized Studies (MINORS) scoring tool was adopted to assess the quality of included studies. The bias risk associated with randomized controlled trials (RCTs) was assessed using the Cochrane risk of bias tool (21), which revealed the presence of proper allocation concealment, blinding procedures, and random sequence generation, along with complete outcome data reporting and no evidence of selective outcome reporting or other biases. GRADE quality evaluation was further conducted by analyzing therapeutic regimens with respect to survival results, drug reaction, and AEs, which confirmed that the outcomes were of high quality. Divergence and discrepancies in the quality assessment were resolved by group discussion.
Statistical analysis
All data included in this meta-analysis were analyzed using RevMan 5.4 provided by the Cochrane Collaboration. The specific statistical methods for the median meta-analysis are provided in Appendix 1. We used the chi-squared test and I2 statistic to assess the level of heterogeneity. The P values <0.1 indicated statistical heterogeneity (22). When P values <0.1 or I2>50%, a random effects model was used in the meta-analysis. Otherwise, a fixed effects model was used to assess the source of the heterogeneity. In addition, sensitivity analysis was conducted to assess the reliability and stability of the combined results.
Results
Results of literature search
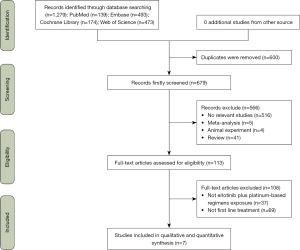
The study flowchart is illustrated in Figure 1. Initially, a total of 1,279 publications were retrieved, of which 600 duplicates were excluded. We read the titles and abstracts of the remaining 679 publications; of those, 516 publications were not relevant, 5 publications were meta-analyses, 4 publications described animal experiments, and 41 publications were reviews. After excluding 566 publications, we carefully screened the remaining 113 articles. Finally, we identified 7 studies that were selected to be included in the current meta-analysis.
Basic characteristics of the included studies
The basic information included the authors, date of publication, participants’ region, age, and treatment (Table 1). In the seven studies included in the meta-analysis (23-29), the anlotinib combined with etoposide and platinum-based regimen groups included a total of 519 cases. Patients were given intravenous chemotherapy; etoposide was administered at a dose of 80–100 mg/m2 every 21 days from days 1–3, in combination with either cisplatin (75–80 mg/m2) or carboplatin (area under the curve: 5–6 mg/mL per min) administered on day 1 of each cycle. Patients received anlotinib orally, administered once each day from day 1 to day 14 of every cycle.
Table 1
| Study | Design | Sample size | Region | Age, years | Male, n [%] | Treatment | Anlotinib dose, mg | Brain metastases, n [%] | Etoposide/carboplatin, n [%] | ECOG PS 0–1, n [%] |
Outcome |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Cheng et al. (23) | RCT | Arm A (n=245) | Asia | – | – | Anlotinib +etoposide/carboplatin | 12 | 22 [9] | 245 [100] | 245 [100] | PFS, OS |
| Arm B (n=247) | Asia | – | – | Etoposide/carboplatin | 26 [10.5] | 247 [100] | 247 [100] | ||||
| Deng et al. (24) | Prospective | 35 | Asia | Mean: 59 | 33 [94.29] | Anlotinib and etoposide with either carboplatin or cisplatin | 12 | 7 [20] | – | 35 [100] | ORR, PFS, OS |
| Gao et al. (25) | Prospective | 30 | Asia | Mean: 65.5 | 28 [93.3] | Anlotinib + etoposide + platinum | 8, 10, or 12 | – | – | – | ORR, PFS |
| Kong et al. (26) | Prospective | 20 | Asia | Mean: 66.2 | 17 [85.0] | Anlotinib and etoposide with either carboplatin or cisplatin | 12 | 3 [15.0] | 6 [30] | 17 [75] | ORR, PFS, OS |
| Liu et al. (27) | Prospective | 30 | Asia | Mean: 62 | 25 [83.8] | Anlotinib and etoposide with either carboplatin or cisplatin | 12 | 10 [27] | 12 [40] | 30 [100] | ORR, PFS, OS |
| Zhang et al. (28) | Prospective | 101 | Asia | Mean ± SD: 61.6±8.0 | 79 [91.9] | Anlotinib and etoposide with either carboplatin or cisplatin | 8, 10, or 12 | 14 [16.3] | 50 [58.1] | 83 [83] | ORR, PFS, OS |
| Zheng et al. (29) | Retrospective | 58 | Asia | Range: 36–81 | 47 [81.0] | Anlotinib plus platinum-etoposide | 12/10 | 5 [8.6] | – | 38 [65.5] | PFS, OS, ORR |
ECOG PS, Eastern Cooperative Oncology Group Performance Status; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; RCT, randomized controlled trial; SD, standard deviation.
Risk of bias for the included studies
The RCT was of high quality (Table 2), and the quality assessment details of noncomparative studies are summarized in Table 3. According to the GRADE method, all of the results were of medium-high quality (Table S1).
Table 2
| Quality assessment item | Evaluation |
|---|---|
| Q1: True randomization | Yes |
| Q2: Allocation concealment | Yes |
| Q3: Similar at baseline | Unclear |
| Q4: Blinding of subjects | Yes |
| Q5: Blinding of the researcher | Yes |
| Q6: Blinding of outcome assessors | Unclear |
| Q7: Identical treatment other than the intervention | Unclear |
| Q8: Follow-up description | Unclear |
| Q9: Intention-to-treat analysis | Unclear |
| Q10: The same outcome measurement | Yes |
| Q11: Reliability of outcome measurements | Yes |
| Q12: Appropriate statistical analysis | Unclear |
| Q13: Appropriate trial design | Unclear |
| Quality grade | High |
Assessment based on Cheng et al. (23).
Table 3
| Study | Q1 | Q2 | Q3 | Q4 | Q5 | Q6 | Q7 | Q8 | Total point |
|---|---|---|---|---|---|---|---|---|---|
| Deng et al. (24) | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 0 | 14 |
| Gao et al. (25) | 2 | 2 | 2 | 2 | 1 | 1 | 0 | 0 | 10 |
| Kong et al. (26) | 2 | 2 | 2 | 2 | 2 | 2 | 1 | 0 | 13 |
| Liu et al. (27) | 2 | 2 | 2 | 2 | 0 | 1 | 0 | 1 | 10 |
| Zhang et al. (28) | 2 | 2 | 2 | 2 | 2 | 2 | 1 | 1 | 14 |
| Zheng et al. (29) | 2 | 2 | 2 | 2 | 2 | 2 | 0 | 0 | 12 |
Q1: clearly stated research objectives. Q2: consecutive enrollment of patients. Q3: prospective data collection. Q4: the endpoints adequately reflect the study objectives. Q5: the objectivity of endpoint assessment. Q6: Is the follow-up duration adequate? Q7: the loss to follow-up was below 5%. Q8: Was the sample size estimation?
PFS
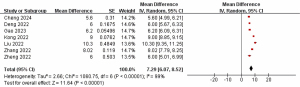
All the studies included in the analysis (23-29) reported PFS for all patients who received anlotinib in combination with platinum-etoposide in the first-line therapy of ES-SCLC. In the random effects model (I2=99.0%, P<0.001), the pooled median PFS (mPFS) was 7.29 months [95% confidence interval (CI): 6.07–8.52 months] (Figure 2).
Overall survival
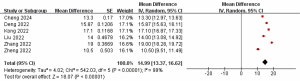
Six studies included in the analysis (23,24,26-29) reported OS for all patients who received anlotinib in combination with platinum-etoposide in the first-line therapy of ES-SCLC. In the random effects model (I2=99.0%, P<0.001), the pooled mOS was 14.99 months (95% CI: 13.37–16.62 months) (Figure 3).
ORR
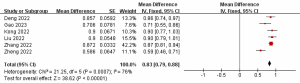
Six studies included in the analysis (24-29) reported ORR for all patients who received anlotinib in combination with platinum-etoposide in the first-line therapy of ES-SCLC. In the random effects model (I2=76.0%, P<0.001), the pooled ORR was 83.0% (95% CI: 79–88%) (Figure 4).
AEs
The most common AEs (all-grade and grade ≥3) associated with anlotinib in combination with platinum-etoposide in the first-line therapy of patients with ES-SCLC are shown in Table 4 and Figures S1-S10. Most patients experienced grades 1–2 AEs, which were well tolerated. The results also identified the commonly reported AEs, including hypertension (28.36%, 95% CI: 22.97–33.75%), fatigue (27.24%, 95% CI: 21.91–32.57%), decreased white blood cell counts (27.20%, 95% CI: 21.87–32.53%), neutropenia (23.88%, 95% CI: 18.78–28.98%), oral mucositis (16.79%, 95% CI: 12.32–21.27%), anemia (16.42%, 95% CI: 11.99–20.86%), hand-foot syndrome (16.04%, 95% CI: 11.65–20.43%), hypertriglyceridemia (8.58%, 95% CI: 5.23–11.93%), hypercholesterolemia (5.59%, 95% CI: 2.84–8.34%), rash (4.10%, 95% CI: 1.73–6.47%), hypophosphatemia (4.10%, 95% CI: 1.73–6.47%), and hypocalcemia (0.75%, 95% CI: −0.28–1.78%). The most common hematologic toxicities included anemia (16.42%, 95% CI: 11.99–20.86%) and neutropenia (23.88%, 95% CI: 18.78–28.98%). The incidence of grade ≥3 AEs was significantly lower, rarely exceeding 16%. Even the incidence rates of the most commonly reported AEs, such as neutropenia, decreased white blood, and hand–foot syndrome, were only 15.67%, 6.34%, and 5.59%, respectively.
Table 4
| Adverse event | All grade | Grade ≥3 | |||
|---|---|---|---|---|---|
| Incidence (95% CI), % | I2, % | Incidence (95% CI), % | I2, % | ||
| Hypertension | 0.2836 (0.2297, 0.3375) | 89 | 0.0373 (0.0146, 0.0599) | 0 | |
| Fatigue | 0.2724 (0.2191, 0.3257) | 82 | 0.0373 (0.0146, 0.0599) | 56 | |
| Decreased white blood | 0.2720 (0.2187, 0.3253) | 93 | 0.0634 (0.0342, 0.0926) | 61 | |
| Neutropenia | 0.2388 (0.1878, 0.2898) | 93 | 0.1567 (0.1132, 0.2002) | 69 | |
| Oral mucositis | 0.1679 (0.1232, 0.2127) | 91 | 0.0448 (0.020, 0.0695) | 85 | |
| Anemia | 0.1642 (0.1199, 0.2086) | 87 | 0.0149 (0.0004, 0.0294) | 27 | |
| Hand-foot syndrome | 0.1604 (0.1165, 0.2043) | 5 | 0.0559 (0.0284, 0.0834) | 75 | |
| Hypertriglyceridemia | 0.0858 (0.0523, 0.1193) | 94 | 0.0149 (0.0004, 0.0294) | 0 | |
| Hypercholesterolemia | 0.0559 (0.0284, 0.0834) | 70 | 0 | 0 | |
| Rash | 0.0410 (0.0173, 0.0647) | 0 | 0.0037 (−0.0036, 0.0109) | 0 | |
| Hypophosphatemia | 0.0410 (0.0173, 0.0647) | 59 | 0.0112 (−0.0036, 0.0238) | 0 | |
| Hypocalcemia | 0.0075 (−0.0028, 0.0178) | 0 | 0.0037 (−0.0014, 0.0109) | 0 | |
CI, confidence interval; ES-SCLC, extensive-stage small-cell lung cancer.
Sensitivity analysis
High heterogeneity was found in mPFS, OS, ORR, and the grade of AEs; thus, we assessed the impact of each study on the pooled results. The results showed that all of the pooled results with 95% CIs were not markedly affected by any individual study (Tables S2-S4). This indicated that the results of the present meta-analysis were both reliable and stable.
Publication bias
Funnel plot tests were utilized for the assessment of publication bias risk. The results are shown in Figures S11,S12. Publication bias could not be reliably assessed.
Discussion
We conducted a meta-analysis by combining six single-arm studies and an RCT study; a total of 519 ES-SCLC cases were included in our analyses. Our findings revealed that in the first-line treatment of ES-SCLC, anlotinib in combination with etoposide and platinum-based regimens showed efficacy and controllable safety, with favorable PFS, ORR, and OS. All-grade AEs, hypertension, fatigue, and decreased white blood cell counts commonly occurred in these patients. Among the 519 patients who were evaluated in the efficacy analysis, the ORR was 83.0%, and the mPFS and mOS were 7.29 and 14.99 months, respectively. No unexpected treatment-related AEs were found.
Use of anlotinib in the first-line treatment of ES-SCLC has potentially promising PFS and OS. The mPFS and mOS linked to platinum-etoposide chemotherapy (i.e., the standard first-line treatment of ES-SCLC for decades) were approximately 5 and 10 months, respectively (30,31). According to the IMpower133 clinical trial (32,33), treatment with programmed death-ligand 1 (PD-L1) antibody atezolizumab plus platinum-etoposide chemotherapy in the first-line therapy for ES-SCLC improved OS (mOS: 12.3 months). Although the combination of atezolizumab regimen resulted in prolonged OS, the survival times were unsatisfactory. Our meta-analysis results showed that the pooled mOS was 14.99 months (95% CI: 13.37–16.62 months). In our study, PFS and OS were very encouraging; nevertheless, comparisons of studies involving treatment with ICIs plus platinum-etoposide chemotherapy should be performed with caution.
Anlotinib is a tyrosine kinase inhibitor that exhibits promising anti-tumour effects by targeting multiple signalling receptors (e.g., VEGFR1–3, FGFR1–4) and promoting vascular matrix reprogramming (34). Recently, the efficacy and safety of anlotinib in patients with SCLC have been reported in clinical studies and meta-analyses. The prospective phase II clinical trial involved 45 patients with relapsed SCLC who received anlotinib treatment. The results demonstrated that the mPFS was 4.1 months and the mOS was 6.1 months. The most common AE was hypertension (13%). Anlotinib is an effective treatment for patients with relapsed SCLC, and the associated side effects are well managed and tolerated (35).
The efficacy and safety of anlotinib in previously treated SCLC were evaluated through a meta-analysis (36). By combining 13 studies including 779 patients with SCLC, the results demonstrated that the PFS and OS of patients in the anlotinib group were 3.46 and 6.86 months, respectively. Compared with the control group, the ORR was significantly higher and the PFS was significantly longer in the anlotinib group; however, there was no significant difference in OS between the anlotinib and control groups (36). These results demonstrate the effectiveness of anlotinib in treating previously treated SCLC. Our meta-analysis combined seven studies including a total of 519 patients with ES-SCLC who were treated with anlotinib combined with platinum-etoposide as first-line therapy. The data revealed a pooled PFS of 7.29 months and a pooled OS of 14.99 months.
Our meta-analysis included an RCT study (23): the randomised phase III trial evaluated anlotinib plus etoposide/carboplatin versus placebo plus etoposide/carboplatin in first-line therapy for ES-SCLC. Compared with the etoposide/carboplatin group, the anlotinib group had a significantly longer mPFS (5.6 vs. 4.2 months, respectively; 95% CI: 0.36–0.55; P<0.001). Regarding mOS, the values of the two groups were 13.3 and 11.9 months, respectively (95% CI: 0.67–11.10; P=0.1723). The results demonstrate that anlotinib plus chemotherapy significantly improves PFS compared to chemotherapy alone in the first-line treatment of ES-SCLC.
Preclinical data showed that VEGF was overexpressed in ES-SCLC (37), thus, angiogenesis inhibitors have been used to treat ES-SCLC. The benefits noted in our meta-analysis were comparable with those obtained after treatment with other angiogenesis inhibitors in the first-line treatment, such as bevacizumab (mPFS: 6.7 months; mOS: 9.8 months) in the GOIRCAIFA trial (38), rh-endostatin (mPFS: 6.4 months; mOS: 12.1 months) (39), sorafenib (mPFS: 5.1 months; mOS: 7.4 months) (40), and apatinib (mPFS: 7.8 months; mOS: 12.1 months) (41). In a previous study, the combination therapy of bevacizumab and platinum-etoposide improved PFS, but did not improve OS (38). Anlotinib exhibits stronger anti-angiogenic activity than other anti-angiogenic inhibitors. In our meta-analysis, a potentially promising clinical efficacy in OS was observed. This finding suggested that anlotinib in combination with etoposide and platinum-based therapy could be an effective option for first-line ES-SCLC treatment.
Our study results revealed that the most frequently reported AEs were hypertension (28.36%), fatigue (27.24%), and decreased white blood (27.20%). Most AEs were of grades 1–2, and the incidence of grade ≥3 AEs was low, rarely exceeding 16%. Importantly, even the incidence rates of the most commonly reported AEs, namely neutropenia, decreased white blood, and hand-foot syndrome were only 15.67%, 6.34%, and 5.59%, respectively. According to these results, the safety of anlotinib in combination with etoposide and platinum-based therapy in the first-line treatment of ES-SCLC is acceptable.
The present meta-analysis had several strengths. We analyzed pooled data from relevant trials comprehensively and provided robust evidence demonstrating the efficacy and safety of anlotinib in combination with etoposide and platinum-based therapy as a first-line treatment for patients with ES-SCLC. Although a meta-analysis on the subsequent-line treatment of SCLC with anlotinib has been published (36), our study is the first to focus on anlotinib in combination with etoposide and platinum-based regimens in the first-line treatment of ES-SCLC. The administration of ICIs plus platinum-etoposide chemotherapy is currently the recommended first-line treatment for advanced ES-SCLC. However, most patients quickly experience tumour progression due to the development of chemotherapy drug resistance easily (42,43). Our meta-analysis provides evidence that anlotinib in combination with etoposide and platinum-based regimens shows potentially promising clinical efficacy in the first-line treatment of patients with ES-SCLC. Therefore, in clinical practice, the addition of anlotinib to etoposide and platinum-based regimens may be considered one of the first-line treatment strategies for ES-SCLC.
Nevertheless, this study had several potential limitations. Firstly, the analysis included only seven studies, and some of which had relatively small sample sizes. The pooled results show extreme heterogeneity. Thus, our conclusions should be interpreted cautiously. Secondly, the trials in this analysis included only Chinese participants; it is unclear whether these results would be consistent for other ethnicities. Thirdly, significant heterogeneity may have resulted from inhomogeneity at both the participant and trial levels, including small samples, exclusively Chinese cohorts, variable anlotinib dosing (8/10/12 mg), and differing proportions of the etoposide with etoposide-carboplatin versus etoposide-cisplatin regimens. Finally, six of the studies were non-randomized single-arm investigations lacking a control group, which may have caused selection bias. Hence, the effectiveness of anlotinib combined with etoposide and platinum-based regimens in the first-line treatment of ES-SCLC warrants further investigation in RCTs.
Conclusions
The results of this meta-analysis implied that anlotinib combined with etoposide and platinum-based regimens has potentially promising clinical efficacy and a favorable safety profile in the first-line treatment of patients with ES-SCLC. To validate these findings, further investigations with large sample sizes, including patients of different ethnicities, are warranted. This would allow researchers to make conclusive comparisons between standard care and the aforementioned combination regimen as first-line treatment.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the PRISMA reporting checklist. Available at https://jtd.amegroups.com/article/view/10.21037/jtd-2025-1813/rc
Peer Review File: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-2025-1813/prf
Funding: This study was supported by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jtd.amegroups.com/article/view/10.21037/jtd-2025-1813/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Kratzer TB, Bandi P, Freedman ND, et al. Lung cancer statistics, 2023. Cancer 2024;130:1330-48. [Crossref] [PubMed]
- Thai AA, Solomon BJ, Sequist LV, et al. Lung cancer. Lancet 2021;398:535-54. [Crossref] [PubMed]
- Kim SY, Park HS, Chiang AC. Small Cell Lung Cancer: A Review. JAMA 2025;333:1906-17. [Crossref] [PubMed]
- Meder L, Orschel CI, Otto CJ, et al. Blocking the angiopoietin-2-dependent integrin β-1 signaling axis abrogates small cell lung cancer invasion and metastasis. JCI Insight 2024;9:e166402. [Crossref] [PubMed]
- Nie Y, Chiang AC. Small-Cell Lung Cancer Updates. Semin Respir Crit Care Med 2025;46:467-78. [Crossref] [PubMed]
- Gutiérrez M, Zamora I, Freeman MR, et al. Actionable Driver Events in Small Cell Lung Cancer. Int J Mol Sci 2023;25:105. [Crossref] [PubMed]
- Paz-Ares L, Dvorkin M, Chen Y, et al. Durvalumab plus platinum-etoposide versus platinum-etoposide in first-line treatment of extensive-stage small-cell lung cancer (CASPIAN): a randomised, controlled, open-label, phase 3 trial. Lancet 2019;394:1929-39. [Crossref] [PubMed]
- Jalal SI, Lavin P, Lo G, et al. Carboplatin and Etoposide With or Without Palifosfamide in Untreated Extensive-Stage Small-Cell Lung Cancer: A Multicenter, Adaptive, Randomized Phase III Study (MATISSE). J Clin Oncol 2017;35:2619-23. [Crossref] [PubMed]
- Lee JH, Saxena A, Giaccone G. Advancements in small cell lung cancer. Semin Cancer Biol 2023;93:123-8. [Crossref] [PubMed]
- Ready NE, Pang HH, Gu L, et al. Chemotherapy With or Without Maintenance Sunitinib for Untreated Extensive-Stage Small-Cell Lung Cancer: A Randomized, Double-Blind, Placebo-Controlled Phase II Study-CALGB 30504 (Alliance). J Clin Oncol 2015;33:1660-5. [Crossref] [PubMed]
- Rudin CM, Awad MM, Navarro A, et al. Pembrolizumab or Placebo Plus Etoposide and Platinum as First-Line Therapy for Extensive-Stage Small-Cell Lung Cancer: Randomized, Double-Blind, Phase III KEYNOTE-604 Study. J Clin Oncol 2020;38:2369-79. [Crossref] [PubMed]
- Yang C, Xuan T, Gong Q, et al. Efficacy and safety of novel immune checkpoint inhibitor-based combinations versus chemotherapy as first-line treatment for patients with extensive-stage small cell lung cancer: A network meta-analysis. Thorac Cancer 2024;15:1246-62. [Crossref] [PubMed]
- Zugazagoitia J, Paz-Ares L. Extensive-Stage Small-Cell Lung Cancer: First-Line and Second-Line Treatment Options. J Clin Oncol 2022;40:671-80. [Crossref] [PubMed]
- Gomez-Randulfe I, Leporati R, Gupta B, et al. Recent advances and future strategies in first-line treatment of ES-SCLC. Eur J Cancer 2024;200:113581. [Crossref] [PubMed]
- Hotta K, Nishio M, Saito H, et al. First-line durvalumab plus platinum-etoposide in extensive-stage small-cell lung cancer: CASPIAN Japan subgroup analysis. Int J Clin Oncol 2021;26:1073-82. [Crossref] [PubMed]
- Goldman JW, Dvorkin M, Chen Y, et al. Durvalumab, with or without tremelimumab, plus platinum-etoposide versus platinum-etoposide alone in first-line treatment of extensive-stage small-cell lung cancer (CASPIAN): updated results from a randomised, controlled, open-label, phase 3 trial. Lancet Oncol 2021;22:51-65. [Crossref] [PubMed]
- Gao Y, Liu P, Shi R. Anlotinib as a molecular targeted therapy for tumors. Oncol Lett 2020;20:1001-14. [Crossref] [PubMed]
- Han B, Li K, Wang Q, et al. Effect of Anlotinib as a Third-Line or Further Treatment on Overall Survival of Patients With Advanced Non-Small Cell Lung Cancer: The ALTER 0303 Phase 3 Randomized Clinical Trial. JAMA Oncol 2018;4:1569-75. [Crossref] [PubMed]
- Gao X, Peng L, Zhang L, et al. Real-world efficacy and safety of anlotinib as third- or further-line treatment in refractory small cell lung cancer. J Cancer Res Clin Oncol 2022;148:2661-71. [Crossref] [PubMed]
- Hutton B, Salanti G, Caldwell DM, et al. The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: checklist and explanations. Ann Intern Med 2015;162:777-84. [Crossref] [PubMed]
- DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials 1986;7:177-88. [Crossref] [PubMed]
- Zhao Y, Liu J, Cai X, et al. Efficacy and safety of first line treatments for patients with advanced epidermal growth factor receptor mutated, non-small cell lung cancer: systematic review and network meta-analysis. BMJ 2019;367:l5460. [Crossref] [PubMed]
- Cheng Y, Yang R, Chen J, et al. Anlotinib plus etoposide/carboplatin (EC) versus placebo plus EC in first-line therapy for extensive-stage small cell lung cancer (ES-SCLC): A randomized, double-blind, parallel controlled, phase III trial (ETER701). ESMO Open 2024;9:102769.
- Deng P, Hu C, Chen C, et al. Anlotinib plus platinum-etoposide as a first-line treatment for extensive-stage small cell lung cancer: A single-arm trial. Cancer Med 2022;11:3563-71. [Crossref] [PubMed]
- Gao F, Zhao Y, Li X, et al. 2005P Anlotinib combined with chemotherapy in the treatment of first-line extensive-stage small cell lung cancer (ES-SCLC): A real-world study. Ann Oncol 2023;34:S1069.
- Kong T, Chen L, Zhao X, et al. Anlotinib plus etoposide and cisplatin/carboplatin as first-line therapy for extensive-stage small cell lung cancer (ES-SCLC): a single-arm, phase II study. Invest New Drugs 2022;40:1095-105. [Crossref] [PubMed]
- Liu C, Liao J, Wu X, et al. A phase II study of anlotinib combined with etoposide and platinum-based regimens in the first-line treatment of extensive-stage small cell lung cancer. Thorac Cancer 2022;13:1463-70. [Crossref] [PubMed]
- Zhang W, Deng P, Kong T, et al. Safety and efficacy of anlotinib in combination with standard chemotherapy as first-line treatment for extensive-stage small cell lung cancer: A multi-center, prospective study (ACTION-2). Lung Cancer 2022;173:43-8. [Crossref] [PubMed]
- Zheng HR, Jiang AM, Gao H, et al. The efficacy and safety of anlotinib combined with platinum-etoposide chemotherapy as first-line treatment for extensive-stage small cell lung cancer: A Chinese multicenter real-world study. Front Oncol 2022;12:894835. [Crossref] [PubMed]
- Shimokawa T, Okamoto H, Machida R, et al. Carboplatin and irinotecan (CI) vs. carboplatin and etoposide (CE) for the treatment of extended-stage small-cell lung cancer in an elderly population: A phase II/III randomized control trial. Lung Cancer 2023;181:107195. [Crossref] [PubMed]
- Kim SH, Jo EJ, Mok J, et al. Real-world evaluation of atezolizumab and etoposide-carboplatin as a first-line treatment for extensive-stage small cell lung cancer. Korean J Intern Med 2023;38:218-25. [Crossref] [PubMed]
- Mansfield AS, Każarnowicz A, Karaseva N, et al. Safety and patient-reported outcomes of atezolizumab, carboplatin, and etoposide in extensive-stage small-cell lung cancer (IMpower133): a randomized phase I/III trial. Ann Oncol 2020;31:310-7. [Crossref] [PubMed]
- Liu SV, Reck M, Mansfield AS, et al. Updated Overall Survival and PD-L1 Subgroup Analysis of Patients With Extensive-Stage Small-Cell Lung Cancer Treated With Atezolizumab, Carboplatin, and Etoposide (IMpower133). J Clin Oncol 2021;39:619-30. [Crossref] [PubMed]
- Shen G, Zheng F, Ren D, et al. Anlotinib: a novel multi-targeting tyrosine kinase inhibitor in clinical development. J Hematol Oncol 2018;11:120. [Crossref] [PubMed]
- Wu D, Nie J, Hu W, et al. A phase II study of anlotinib in 45 patients with relapsed small cell lung cancer. Int J Cancer 2020;147:3453-60. [Crossref] [PubMed]
- Xu H, Cao D, Jie F, et al. The efficacy and safety of anlotinib for subsequent line treatment of small cell lung cancer: a systematic review and meta-analysis. Tumori 2023;109:203-14. [Crossref] [PubMed]
- Montanino A, Manzo A, Carillio G, et al. Angiogenesis Inhibitors in Small Cell Lung Cancer. Front Oncol 2021;11:655316. [Crossref] [PubMed]
- Tiseo M, Boni L, Ambrosio F, et al. Italian multicenter phase III randomized study of cisplatin-etoposide with or without bevacizumab as first-line treatment in extensive stage small cell lung cancer: treatment rationale and protocol design of the GOIRC-AIFA FARM6PMFJM trial. Clin Lung Cancer 2015;16:67-70. [Crossref] [PubMed]
- Lu S, Li L, Luo Y, et al. A multicenter, open-label, randomized phase II controlled study of rh-endostatin (Endostar) in combination with chemotherapy in previously untreated extensive-stage small-cell lung cancer. J Thorac Oncol 2015;10:206-11. [Crossref] [PubMed]
- Sharma N, Pennell N, Nickolich M, et al. Phase II trial of sorafenib in conjunction with chemotherapy and as maintenance therapy in extensive-stage small cell lung cancer. Invest New Drugs 2014;32:362-8. [Crossref] [PubMed]
- Luo H, Zhang L, Yang B, et al. A randomized phase 2 trial of apatinib vs observation as maintenance treatment following first-line induction chemotherapy in extensive- stage small cell lung cancer. Invest New Drugs 2020;38:148-59. [Crossref] [PubMed]
- Guo C, Wan R, He Y, et al. Therapeutic targeting of the mevalonate-geranylgeranyl diphosphate pathway with statins overcomes chemotherapy resistance in small cell lung cancer. Nat Cancer 2022;3:614-28. [Crossref] [PubMed]
- Deng Y, Guo C, Zhang T, et al. Comprehensive analysis of a real-world cohort identifies geranylgeranyl diphosphate synthase 1 as a predictor of chemoresistance in small cell lung cancer. Int J Cancer 2025;157:2173-86. [Crossref] [PubMed]