Diagnostic bronchoscopy--current and future perspectives
Introduction
Lung cancer is the leading cause of cancer-related mortality worldwide. Overall 5-year survival is poor at 15% with little improvement over the last twenty years (1). Lung cancer presenting at a late stage is largely responsible for this poor survival; only 20% are potentially curative with surgical resection. Anti-smoking campaigns and government legislation will reduce lung cancer burden in younger generations but a significant proportion of the older generation who have smoked in the past remain at risk.
Innovative bronchoscopic techniques diagnose lung cancer earlier and more accurately to improve patient outcomes. Narrow band imaging (NBI) and autofluorescence bronchoscopy (AFB) detect pre-invasive malignancy. Endobronchial ultrasound (EBUS) and Electromagnetic Navigation Bronchoscopy (ENB) are safer alternatives to mediastinoscopy and computed tomography guided transthoracic needle aspiration (CT-TTNA). Emerging technologies such as optical coherence tomography (OCT) and confocal microscopy (CM) allow lesional assessment on a cellular level.
This article describes these technologies and explains how they enhance lung cancer diagnosis and staging.
Standard bronchoscopy
Gustav Killian invented the rigid bronchoscope in 1898. In 1967 Ikeda pioneered the flexible bronchoscope, and video bronchoscopes became mainstream in the early 1980’s. Despite quickly becoming the cornerstone of lung cancer diagnosis, the shortcomings of white light bronchoscopy (WLB) are immediately obvious. Due to the branching nature of the bronchial tree, the airway diameter rapidly reduces the more distal one advances towards lung periphery. A standard 5.9 mm bronchoscope can only access the 4-5th generation bronchi with visualization of the next 1-2 generations, covering only 1/3 of the approximately 23-generation bronchial tree. The majority of lesions visualized on CT are therefore beyond direct bronchoscopic vision. Aberrations in bronchial mucosa, such as angiogenic squamous dysplasia (ASD) or squamous cell carcinoma (SqCC) in situ (CIS) are indistinct and easily missed when lit by white light (2). Similarly extra-bronchial structures such as mediastinal lymph nodes cannot be visualized, and blind needle aspiration (cTBNA) is possible for a limited number of nodal stations (3).
Meta-analysis by Rivera from studies between 1970-2001 found the sensitivity and specificity for bronchoscopically visible lesions was 88% and 100% respectively (4). Diagnostic accuracy for bronchoscopically invisible lesions under fluroscopic guidance varied from 36% to 88%, dependent on biopsy method (transbronchial biopsy vs. cytology brush vs. bronchoalveolar lavage), the number of samples taken, and lesion size. Yield was most affected by lesion size-sensitivity for peripheral pulmonary lesions (PPLs) >2 cm was 63%, decreasing to 34% for PPLs <2 cm. Whilst CT-TTNA is undoubtably more accurate than WLB (pooled sensitivity 0.9; 95% confidence interval 0.88-0.91) its complication rate is higher (5).
Thus, WLB is effective at diagnosing bronchoscopically visible lesions, but is limited in diagnosing bronchoscopically invisible lesions, in situ tumours, and for mediastinal staging of lung cancer.
Endobronchial/mucosal lesions
AFB
AFB takes advantage of endogenous fluorophores in bronchial tissue to inform about metabolic state and biochemical composition of tissues. Normal bronchial tissue fluoresces strongly in green when illuminated by violet or blue light, however as the epithelium becomes dysplastic, progresses to in-situ carcinoma and finally to invasive cancer, the amount of green autofluorescence decreases and red fluorescence also decreases although to a lesser degree. These abnormal areas contrast sharply with normal mucosa.
Two meta-analyses have studied the value of AFB combined with WLB versus WLB alone for detection of intraepithelial neoplasia and invasive lung cancer. The first (14 studies, n=1,358) demonstrated a pooled sensitivity and specificity of AFB + WLB of 0.9 and 0.56, compared to 0.66 and 0.69 for WLB alone (6). The second (21 studies, n=3,266) showed the relative sensitivity on a per lesion basis of AFB + WLB versus WLB alone to detect CIS and invasive cancer was 2.04 and 1.15 respectively (2). AFB + WLB is less specific than WLB alone because false positives are common with AFB due to inflammation, mucous gland hyperplasia and inter-observer error, however specificity and inter- and intra-observer variation can be improved to 80% by combining the quantitative red/green fluorescence ratio (R/G) with bronchoscopic findings (7).
NBI
NBI visualizes bronchial mucosa with blue light (415 nanometers) and green light (540 nanometers) to accentuate superficial capillaries and deeper submucosal vessels respectively, while at the same time reducing light scatter from other wavelengths seen with white light. NBI detects the characteristic abnormal angiogenesis associated with dysplastic lesions. Most of the data detailing the benefits of NBI are from gastroenterological studies and head and neck cancers, however pulmonologists are increasingly using NBI to detect early stage bronchial mucosal lesions.
In 2003 Shibuya et al. studied 48 patients with sputum cytology suspicious or positive for malignancy (8). These patients underwent WLB and AFB, with suspicious areas subsequently interrogated with NBI of differing wavelengths and biopsied for histology. Dotted vessel diameter seen on NBI-B1 (400-430 nm) most closely agreed with pathological examination of ASD vessel diameter.
Shibuya et al. also studied differing NBI characteristics of ASD, CIS, micro-invasive tumour, and invasive SqCC (9). By identifying tortuous vessel networks, dotted vessels, and spiral and screw type vessels, the authors could confidently differentiate between the different stages of multistep carcinogenesis of SqCC.
NBI has a higher specificity and equivalent sensitivity to AFB. Herth et al. evaluated diagnostic yields of NBI alone, and combined with AFB and WLB, in 62 patients referred for airway cancer screening (10). All abnormal lesions underwent forceps biopsy. NBI was less sensitive but more specific than AFB, when compared to WLB. There was no benefit in combining AFI and NBI, a finding confirmed in a more recent study (11). NBI detects dysplasia or malignancy in 23% of patients with normal WLB (12). NBI after WLB led to a change in therapeutic decisions in approximately 10% of patients (13). It is uncertain how NBI compares to high definition WLB (which provides substantially higher resolution images than conventional WLB) for detection of early bronchial mucosal abnormalities.
The natural history of pre-invasive malignancy remains uncertain and treatment at this early stage has not been shown to improve survival however it is likely that at least some of these lesions, if left untreated, will progress to invasive carcinoma. AFB and NBI have higher sensitivity and specificity for detecting mucosal lesions compared to WLB, and diagnostic bronchoscopists should be familiar with their use.
OCT
OCT provides cellular imaging at and below the tissue surface (14-16). It was developed in the 1990’s for ophthalmic applications but has since been used to assess vessel structure, atherosclerotic plaque, and more recently, bronchial wall structure.
Light is emitted by an imaging catheter and the interference pattern between reflected light and light backscattered from the tissue at different depths is collected and analyzed by an interferometer. These patterns are then recombined and decoded, forming a high resolution cross-sectional image. Contact between instrument and tissue is unnecessary and intravenous contrast, dyes or radiation are not needed. OCT resolution is 20 times higher than ultrasound and can be displayed on a monitor in real time. OCT imaging depth is 2-3 mm, and axial and lateral resolution varies between 5-30 micrometers depending on the scanning conditions.
Tsuboi et al. compared OCT images to histological findings of bronchial lesions (14) and found that on OCT, normal bronchial mucosa appears homogeneous whereas the submucosal layer is reflective due to extracellular matrix; A gap is visible between the submucosa and smooth muscle layer, and underlying cartilage shows much scattering. Alveoli have a uniform bronchial wall appearance and air-containing alveoli can be clearly differentiated. Infiltrating cancers, on the other hand, show unevenly distributed high backscattering areas and loss of layer structure and glandular tissue. Lam et al. demonstrated that quantitative measurement of epithelial thickness could differentiate between invasive carcinoma and CIS (P=0.004), and also between dysplasia and metaplasia or hyperplasia (P=0.002). Basement membrane remained intact with CIS, but became disrupted with invasive cancer (15). Certain OCT characteristics can potentially differentiate SqCC from adenocarcinoma without the need for biopsy (17).
Despite these promising pilot studies, it remains to be seen if and how OCT will add value to our current diagnostic approaches. Possible uses for OCT include: distinguishing benign from malignant central and peripheral lesions, differentiating CIS from mincr-invasive cancer; and improving bronchoscopic sampling of PPLs. The performance characteristics and limitations of OCT need to be defined before the technique becomes mainstream.
PPLs
EBUS radial probe (RP-EBUS)
Advances in electronic miniaturization allow a 360-degree viewing ultrasound to fit into a 1.4 mm probe that can be passed through the working channel of a standard bronchoscope. This so-called RP-EBUS allows localization of peripheral lesions and depth assessment of endobronchial lesions. At standard frequency of 20 MHz, the spatial resolution is less than 1 mm and penetration depth is 4-5 cm.
RP-EBUS has two main uses.
Localisation of PPLs
By placing the ultrasound probe into lung periphery one can characterize the tissue densities surrounding the probe. Normal air-filled alveoli have a homogeneous “snow-storm” appearance. If the probe is within a solid lesion however the interface between the mass and surrounding aerated lung is represented by a bright line, confirming the probe is within the target.
The use of a guide sheath (GS) in combination with RP-EBUS was introduced in 2004 (18). The GS is a catheter that fits over the RP-EBUS, leaving only the distal ultrasound probe exposed. The RP-EBUS/GS are advanced together through the working channel into the target subsegment until the lesion of interest is localized. Advancing and retracting the RP-EBUS/GS defines the lesion’s proximal and distal extents. Once the desired biopsy site is established, the GS is left in situ and the RP-EBUS is removed and replaced by pre-measured biopsy tools, ensuring samples are taken from the desired location. An additional theoretical benefit of GS is tamponade of biopsy-related bleeding (Figure 1).
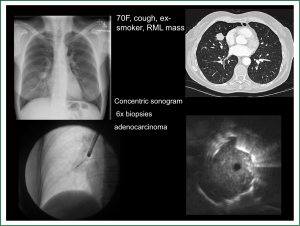
Perhaps the best evidence favouring RP-EBUS/GS over fluoroscopy guided transbronchial lung biopsy (TBLBx) for PPLs was published in 2005 by Paone (19). 221 patients with PPLs were randomly assigned to either procedure (97 RP-EBUS/GS, 124 TBLBx) and those without a diagnosis underwent more invasive investigation. Sensitivity for lung cancer was 0.79 and 0.55 (P=0.004), and accuracy was 0.85 and 0.69 (P=0.007) in RP-EBUS/GS and TBLBx groups respectively, with the majority of the benefit evident in lesions <3 cm [sensitivity of RP-EBUS/GS vs. TBLBx for <3 cm, 71% (47-95%) vs. 23% (3-43%), P value <0.001].
A systematic review and meta-analysis of RP-EBUS/GS for PPL diagnosis (16 studies n=1,420 patients) showed point sensitivity for diagnosis of lung cancer was 0.73, however there was significant study heterogeneity. Complication rates varied between 0-7.4%, with the most common being minor bleeding. Pooled pneumothorax rate was 1.0%, and rate of intercostal drainage was only 0.4% (20).
Most RP-EBUS/GS data originate from centres with bronchoscopic expertise and hence may not reflect “real-world” results. Roth et al. performed a prospective randomised cohort study of RP-EBUS/GS for PPLs by 29 different physicians practicing at community hospitals in Western Norway between June 2005 and January 2008 (21). With a cancer prevalence of 71.5%, the sensitivity for malignancy in the RP-EBUS/GS group was 36% compared with 43.7% in the non-EBUS group. If there was a bronchus leading directly to the lesion, the diagnostic sensitivity was considerably higher (62.2%). This study may provide a more realistic view of what is achievable with RP-EBUS/GS in a community hospital and demonstrates that EBUS is useful in confirming lesion location, but not in guiding the bronchoscopist to the lesion.
The only consistent predictor of success is probe location in relation to the lesion; If the probe is surrounded by tumour (concentric ultrasound image) the diagnostic yield is much higher than if the probe is adjacent to (eccentric image) or not associated with the target lesion (18,22-24). Although Kurimoto found yield was independent of lesion size (18), subsequent studies recognise that higher diagnostic yields are achieved from larger lesions (20).
Few studies have compared RP-EBUS/GS to the non-invasive gold standard of CT-TTNA. In 2008 Fielding described a prospective series of RP-EBUS/GS and compared this to a retrospective review of CT-TTNA during the same period. Diagnostic sensitivity for RP-EBUS/GS was only 35% for lesions touching the visceral pleura, compared to 74% for lesions not touching the visceral pleural. While overall pneumothorax rates were 1% and 28% in EBUS GS and CT groups respectively, the CT-TTNA pneumothorax rate was only 2.6% for lesions in contact with the visceral pleura (25). These results suggest that CT-TTNA rather than RP-EBUS/GS should be the first line investigation for pleural-based lesions.
Depth of invasion of endobronchial lesions
RP-EBUS with a surrounding inflatable balloon can clearly define tracheal and bronchial wall layers, making it an excellent tool to assess tumour invasion. Ultrasonographic assessment of tumour depth correlates well with histopathologic findings (26) and this measurement determines appropriate therapy; Tumours that invade through the cartilage layer require radiotherapy or surgery, whereas those with an intact cartilage layer can be treated endoscopically.
RP-EBUS can also determine whether centrally located tumours adjacent to the trachea are invading the trachea (clinical T4 stage) or simply adjacent to and compressing but not invading the trachea (clinical T1a-3 depending on lesion size). Herth et al. studied 131 consecutive patients with central thoracic malignancies potentially involving the central airways (27). All patients underwent chest CT followed by WLB and RP-EBUS, with subsequent surgical evaluation and radiology results blinded from the bronchoscopists and surgeons. CT reported 77% of lesions were invading airways, but RP-EBUS showed invasion in only 47% of cases. When using surgical assessment as the gold standard, RP-EBUS had a specificity of 100%, sensitivity of 89%, and accuracy of 94%, for assessing tumour invasion.
ENB
ENB is a relatively new bronchoscopic technique with both diagnostic and therapeutic applications. ENB is a two-stage process: pre-procedure planning, and the actual procedure itself. DICOM data is uploaded to a planning computer via network or compact disk. The planning screen consists of four windows, each of which can display axial, sagittal or coronal views, as well as a virtual bronchoscopic animation and three-dimensional bronchial tree. The bronchoscopist outlines the target and then places waypoints along bronchi that lead to this lesion. The planned path can then be viewed via virtual bronchoscopic animation, allowing the operator to see precisely which sequence of airways lead to the lesion.
Selective cannulation of bronchi is possible with a specialized cannula housed in an extended working channel (EWC), passed together through the working channel. The proximal end houses a “steering wheel” that allows deflection of the distal tip in one of eight directions; the distal end contains a “locatable guide” (LG), whose position is tracked through an electromagnetic field encompassing the patient’s chest with the assistance of three location pads placed on the patient’s chest. The computer provides instructions on how and when to turn the “steering wheel” and advance the catheter to reach each waypoint and finally the target lesion (Figure 2). Once the LG is in close proximity and aligned to the target lesion, the EWC is left in place and the LG is removed and replaced with biopsy instruments.
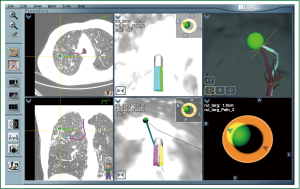
Most of the published ENB literature is case series of patients with PPLs. The overall diagnostic yield for ENB alone is highly variable and ranges from 59% to 77.3% (28-34). The only randomised controlled trial compared (31) EBUS RP, ENB, or a combined approach (ENB to navigate to the lesion and RP-EBUS to confirm lesion localization) to diagnose one hundred and twenty PPLs. Diagnostic yield was 69%, 59% and 88% for RP-EBUS, ENB, and combined ENB/RP-EBUS groups respectively, suggesting that highest diagnostic yield may be achieved via combined procedures that utilize the strengths of each modality.
The majority of ENB publications are non-consecutive cohorts that do not describe selection/inclusion criteria, and (apart from Eberhardt et al.) do not randomize patients to competing modalities. ENB has never been compared to CT-TTNA and thus it is uncertain where ENB fits into the diagnostic algorithm. Due to its high cost and considerable pre-procedure planning (both to obtain DICOM images of recommended parameters, as well as pathway planning) ENB is only likely to become mainstream if consumable prices fall and high level evidence demonstrates diagnostic equivalence to CT-TTNA and/or additional benefit above RP-EBUS. Based on Eberhardt’s study, ENB and RP-EBUS may have complimentary roles, however this combined approach would likely increase cost and procedure time.
Virtual bronchoscopy (VB)/ultrathin bronchoscopy (UB)
VB aims to address the inability of RP-EBUS to guide the bronchoscopist to the target lesion. Traditionally, the bronchoscopist views two-dimensional axial, coronal, and sagittal CT views, and mentally reconstructs a three-dimensional image of the bronchial tree before plotting a path to the target lesion. VB allows CT reconstruction of the bronchial tree allowing “virtual” bronchoscopic animation enabling more accurate procedure planning. An example of this was shown in Figure 3.
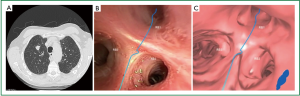
For maximum utility, VB should be coupled with pathway planning software (for example, “Lungpoint”, Broncus Medical Inc, CA, USA) and UB; Newer scopes have external diameters of only 2.8 mm (35), and allow direct visualization up to the 9th generation bronchus. A direct consequence of UB however is a smaller working channel and hence smaller biopsy samples.
UB was initially used by Asano to perform barium marking before thoracoscopic surgery for PPLs (36). The UB could be guided under direct vision to a median sixth generation bronchi (range 4th-9th generation) and markers could be placed to a median distance of 4 mm from the lesion (within 10 mm in 27 of 31 lesions). A separate study whereby patients underwent both WLB and UB in the same procedure demonstrated diagnostic rates of 54.3% and 60% respectively, and when both were combined the yield increased further to 62.8% (37). UB was able to obtain diagnostic material in 59.3% of the patients who had negative rapid cytology on WLB.
Asahina et al. assessed the utility of combining VB with RP-EBUS/GS in 29 patients with PPLs ≤30 mm. 80% of lesions were visualized ultrasonographically and diagnostic sensitivities were 44.4% for lesions <20 mm, and 91.7% for lesions ≥20 mm (38). In a randomised trial of 199 patients with PPLs ≤30 mm undergoing RP-EBUS/GS with and without VB (VBNA vs. non-VBNA groups) the VBNA group demonstrated higher diagnostic yield (80.4% vs. 67%, P=0.032), shorter procedure time (24.0 vs. 26.2 mins, P=0.016), and shorter navigation time (8.1 vs. 9.8 mins, P=0.045) (39).
Recently though a randomised controlled multicentre trial of UB with and without VB for PPLs found no difference in diagnostic yield. 350 patients were randomised and yields were 67.1% vs. 59.9% for VNBA vs. non-VBNA groups respectively. Subgroup analysis showed improved yield in the VBNA group for right upper lobe lesions, lesions invisible on CXR, and lesions in the peripheral third of the lung field. It could be argued that VB is of little benefit to highly experienced operators, however it still may be of significant assistance in those less familiar with bronchial anatomy (40).
UB/VB has three major shortcomings. Firstly, the ability to obtain sufficient tissue for molecular analysis with UB is unknown but presumably reduced as biopsy forceps size may affect biopsy size and quality. Secondly, VB quality is dependent on CT source data and recommended DICOM parameters may be unavailable. Thirdly, VB systems rely on a skilled second operator to manipulate the VB image to the same orientation as the real-time bronchoscopic image; without this, the risk of disorientation is high (41).
Complications of guided bronchoscopic techniques
The biggest advantage of bronchoscopic methods over CT-TTNA for biopsy of PPLs is the lower complication rate. In a meta-analysis of RP-EBUS (16 studies, n=1,420), complication rates varied between 0-7.4%. The pooled pneumothorax rate was 1.0% and the pooled rate of intercostal catheter drainage was 0.4%. No patients had bleeding requiring intervention, and no deaths were reported.
In contrast, data from 15,865 adults who underwent CT-TTNA from the 2006 Healthcare Cost and Utilisation Project’s State Ambulatory Surgery Databases and State Inpatient Databases for California, Florida, Michigan and New York, demonstrated significantly higher complication rates 5: pneumothorax rate was 15% (95% CI: 14.0-16.0%) and 6.6% of all biopsies (95% CI: 6-7.2%) required chest tube insertion. Furthermore, the population most likely to have a PPL requiring investigation (60-69 years old smokers, those with COPD) was also the most likely to suffer from procedural complications. 1% of procedures were associated with hemorrhage with 17.8% of this required blood transfusion.
Conclusion: PPLs
Guided bronchoscopic methods (EBUS GS/RP, ENB, VB/UB) have higher diagnostic sensitivity than TBLBx, but slightly lower sensitivity than CT-TTNA. The biggest advantage with a bronchoscopic approach is the lower complication rate; The diagnostic yield of each guided bronchoscopic technique is similar (42). Each technique has advantages and disadvantages and will ultimately depend on availability, local expertise, and lesion location. Only a large scale multicentre randomised trial directly comparing guided bronchoscopy to CT-TTNA will verify the merits of each procedure and determine when each procedure should be used. The weakness of contemporary data is that they are case series of non-consecutive patients where the selection criteria are not explicitly outlined and comparator groups are not used. Even when comparator studies are performed, the results are dependent on operator expertise so that data are not necessarily generalisable.
Peribronchial/peritracheal/mediastinal lesions
Convex probe EBUS guided transbronchial needle aspiration (CP-EBUS TBNA)
Peribronchial and mediastinal lesions are accessible by conventional TBNA (cTBNA) however intimate anatomical knowledge is required to ensure safety and adequate diagnostic yield. Accurate assessment of an abnormal mediastinum is vital in lung cancer staging to guide best treatment. For several years, the gold standard for mediastinal staging has been surgical mediastinoscopy however this requires significant cost including hospital admission, general anaesthesia, and has associated morbidity and mortality (43). CP-EBUS TBNA addresses many of the shortcomings of cTBNA and surgical mediastinoscopy. Its rapid widespread adoption is due to its excellent utility, ease of use, ability to perform as a day case under light anaesthesia, and excellent patient satisfaction (44). Furthermore, the most recent American College of Chest Physician guidelines regarding staging of lung cancer recommend using EBUS TBNA, EUS FNA, or a combined approach over surgical staging as the best first test for investigating radiologically suspicious mediastinal lymph nodes (grade 1C) (45).
An integrated CP-EBUS TBNA scope/aspirating needle combination allows ultrasonic, real-time visualization of the needle inside the target lesion. The dedicated scope has a 6.9 mm outer diameter and 2 mm instrument channel whose distal end houses a CP-EBUS with flex of 120 degrees upward to 90 degrees downward. An inflatable balloon is sometimes applied over the probe to improve ultrasound signal, particularly in regions where the probe cannot be flexed against the bronchus wall. Vision is through a 30 degree oblique forward viewing fibre-optic lens with an 80 degree viewing angle. The dedicated 21 or 22 G needle is advanced to the distal end of the working channel and secured proximally onto the bronchoscope. Once the target is identified ultrasonically and doppler excludes overlying vessels, the needle is plunged into the lesion. The central stylet is moved back and forth to clear bronchial debris, and 8 to 10 aspirations are taken.
Several studies have proven the utility of CP-EBUS TBNA for mediastinal staging of lung cancer. In one study 163 lymph nodes were sampled in 105 patients. CP-EBUS TBNA correctly predicted lymph node stage with a sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and diagnostic accuracy of 94.6%, 100%, 100%, 89.5%, and 96.3% respectively (46). A series of CP-EBUS TBNA of 572 lymph nodes from 502 patients demonstrated a sensitivity, specificity, and PPV were 94%, 100%, and 100% respectively with surgical staging as the gold standard (47). The same group performed CP-EBUS TBNA on NSCLC patients with a radiologically normal mediastinum, before undergoing surgical staging. For the detection of malignancy, sensitivity, specificity, and NPV was 92.3%, 100%, and 96.3% respectively, with no complications; performing CP-EBUS TBNA on NSCLC patients with a radiologically normal mediastinum avoided unnecessary surgical exploration in one of six patients (48).
Two large systematic reviews/meta-analyses confirm these findings and cement the utility of CP-EBUS TBNA in the staging of lung cancer. In the first (20 studies), sensitivity ranged between 85-100% and NPV ranged from 11-97.4%, with no serious complications reported (49). In the second (11 studies, P=1,299), sensitivity and specificity was 0.93 and 1.00 respectively; For the subgroup selected based on CT or PET findings sensitivity increased to 0.94 compared to only 0.76 without CT or PET. Only two patients had complications (0.15%) (50).
Yasufuku et al. performed one of the few prospective studies directly comparing CP-EBUS TBNA to mediastinoscopy for staging of lung cancer (51). Patients with confirmed or suspected NSCLC requiring mediastinoscopy as part of their staging investigations were eligible for inclusion. 153 patients underwent CP-EBUS TBNA followed by mediastinoscopy, with the operator blinded to the rapid on-site examination results; If lymph node involvement was present, patients proceeded onto surgical resection and these results were used as gold standard. Sensitivity, NPV and diagnostic accuracy for CP-EBUS TBNA and mediastinoscopy was 81%, 91%, 93% and 79%, 90%, 93% respectively, with no significant differences between the two in yielding true pathologic N stage (P=0.78).
Combined mediastinal staging with CP-EBUS TBNA and EUS-FNA (so called “medical staging”) provides more complete staging allowing additional access to stations 8 and 9. Medical staging seems superior to either staging method alone (52-54). A randomized controlled study showed that medical staging had a higher sensitivity for nodal metastases and was associated with fewer unnecessary thoracotomies when compared to mediastinoscopy (55). Both EUS-FNA and CP-EBUS TBNA can be performed in the one sitting using a single bronchoscope without compromising efficacy, and presumably saving time and money (56,57).
CP-EBUS TBNA equipment, technique and specimen preparation are critical. Diagnostic yield does not differ between 21 and 22 G aspirating needles, but the former results in fewer needle passes (58), better preserves histological structure, with the trade-off being more blood contamination (59). Although suction is traditionally applied to aspirate samples, a randomised controlled trial found no difference in specimen adequacy, quality or diagnosis between specimens with and without suction (60). Three aspiration passes per lymph node is diagnostically optimal with additional passes offering minimal benefit when no rapid onsite evaluation (ROSE) is available (61). The “tissue coagulum clot” method, which involves pushing the specimen onto a pre-cut piece of filter paper with the needle moved in a circular motion to build a cone shaped coagulum of clot and tissue, may increase amounts of diagnostic material when compared to conventional saline needle rinse (62). The use of mini-forceps-transbronchial needle forceps with a beveled end to facilitate penetration through the bronchus wall and jaws that can be opened under ultrasound guidance-is an alternate method to achieve larger volume samples and pilot studies confirm its safety and efficacy (63).
Histological subtyping from CP-EBUS TBNA reflects true histology. In one retrospective study, 88 patients who underwent CP-EBUS TBNA had these results compared to core biopsies and/or follow up surgery. Sensitivity, specificity, PPV, and NPV for CP-EBUS TBNA were 85%, 100%, 100%, and 89.7% respectively (64). A more recent study of 92 patients with NSCLC demonstrated a 76% agreement between needle aspirate and biopsy for subtyping (kappa =0.52), with agreement increasing if cell blocks were available (96%, kappa 0.91 vs. 69%, kappa 0.39 respectively) (65).
Mutational analysis is also possible from CP-EBUS TBNA samples. 154 out of 156 cases were successfully analysed (98.7%) for EGFR mutations using the PCR clamp technique on cell-pellets derived from needle-washed solution (66). Garcia-Olive and colleagues showed EGFR analysis was possible in 72.2% of patients undergoing CP-EBUS TBNA with metastatic nodal specimens (67). In a different study analysis for EGFR and KRAS sequences using COLD-PCR was achieved in 95.5% and 98.4% respectively of samples (68). A UK group found that 88% of their CP-EBUS TBNA samples were adequate for mutational analysis using the Scorpion ARMS kit (69).
The actual false negative rate for CP-EBUS TBNA is a matter of debate; Whilst specificity is unequivocally acceptable, sensitivity and NPV is more important when staging cancer. In 109 patients who underwent CP-EBUS TBNA of PET-avid N2 and N3 lymph nodes, 32 patients were tumour negative by CP-EBUS TBNA but subsequent biopsy in 19 showed malignancy in 7, four due to sampling error, and three due to detection error (70). Thus, in the setting of a high pre-test probability of nodal metastasis and no malignant cells on CP-EBUS TBNA, surgical biopsy should be used for confirmation (71).
Aside from mediastinal staging for NSCLC, CP-EBUS TBNA can diagnose central parenchymal lesions not visible on WLB, avoiding procedures associated with higher complication rates such as CT guided TTNA or mediastinoscopy (72-74).
Sarcoidosis can be reliably detected on CP-EBUS TBNA specimens. The combination of CP-EBUS TBNA and ROSE has high diagnostic accuracy, good interobserver agreement, and can inform the bronchoscopist of whether additional passes are necessary (75). Diagnostic accuracy of CP-EBUS TBNA is significantly higher than transbronchial biopsy or bronchoalveolar lavage (91.4% CP-EBUS TBNA, 65.7% BAL, 40% TBLBx) in Stage I sarcoid disease, although the three modalities have equivalent diagnostic rates in Stage II sarcoid disease (76). A systematic review and meta-analysis of CP-EBUS TBNA for sarcoidosis (15 studies, n=533) found a pooled diagnostic accuracy of 79% with only five minor complications reported, however significant study heterogeneity and publication bias were identified (77).
The diagnosis of sarcoidosis on CP-EBUS TBNA, however, should be tempered by the patient’s pre-test probability of having this condition; in a case series of 1,275 patients undergoing CP-EBUS TBNA, granulomatous inflammation was found in 154 (12.1%) patients of whom 12 (7.8%) had a concurrent diagnosis of cancer, although no patient had both granulomatous inflammation and malignancy within the same lymph node (78). Patients with a high pre-test probability of malignancy but only granulomatous inflammation from CP-EBUS TBNA samples should be considered for additional sampling or close radiological follow up to ensure a benignity (79).
Cost effectiveness
Economic analyses validate the economic viability of CP-EBUS TBNA compared to cTBNA and mediastinoscopic staging. In a retrospective cohort of 294 patients with thoracic lymphadenopathy from a University Hospital, 37 patients underwent cTBNA and 257 had CP-EBUS TBNA. 90% of the CP-EBUS TBNA group was diagnostic compared to 62.2% of the cTBNA group; a higher proportion in the cTBNA group needed additional surgical procedures such as mediastinoscopy, video-assisted thoracic surgery (VATS), or an open thoracotomy. The mean savings with CP-EBUS TBNA was $1,071.09 per patient (80). Improved cost efficacy was also found in a health technology assessment involving hospitals from the United Kingdom, Belgium, and the Netherlands (81). Study patients were randomized to either surgical staging alone, or CP-EBUS TBNA/EUS-FNA followed by surgical staging if negative. The 6-month cost of the former group was £10,459 per patient compared to £9,713 per patient with the latter approach, a saving of £746 per patient mainly through reducing mediastinoscopies and unnecessary thoracotomies.
Learning curve/training
Proficiency in CP-EBUS TBNA improves with experience, however the number of procedures required for proficiency is uncertain (82,83). A cusum (cumulative sum control chart) analysis determined that learning curve duration was highly variable, even for experienced bronchoscopists, with one operator almost immediately gaining competence, whilst another still on the learning curve after 100 procedures (84).
Trainees performing CP-EBUS TBNA increase procedure time, amount of sedation used, and complication rates (85). Obtaining proficiency using an CP-EBUS TBNA simulator before performing real procedures may address some of these issues; In one study, simulator training was equivalent to 15-25 “on-the-job” procedures in terms of procedure time and percentage of lymph nodes successfully identified (86,87). With the number of trainees wanting to acquire this new skill, coupled with the increasing focus on efficiency and reduction in complications, the EBUS simulator may become an increasingly valuable asset.
ROSE
ROSE of needle aspirates is thought to be beneficial in CP-EBUS TBNA but results have been varied. Potential advantages of ROSE for CP-EBUS TBNA include: quicker diagnosis, shorter procedure time, fewer needle passes per lymph node, and as a consequence reduction in complications.
In a prospective study, 120 patients suspected of having lung cancer with mediastinal adenopathy ≥10 mm were randomized to CP-EBUS TBNA with or without ROSE. In the ROSE group, the decision to make additional passes/procedures was based on ROSE findings at the operator’s discretion; In the non-ROSE group, the target lesion underwent a minimum of three punctures, and additional punctures or bronchoscopic procedures were performed if the examiner deemed it necessary. There were significantly fewer punctures of the target lesion in the ROSE group (mean 2.2 punctures vs. 3.1 punctures, P<0.001) and significantly greater additional procedures in the non-ROSE group (57% non-ROSE vs. 11% ROSE). The mean bronchoscopy time, sensitivity and diagnostic accuracy did not differ between the groups.
ROSE results, however, need to be interpreted with caution as false negatives can occur (88) and concordance between staging and final pathological diagnosis is not perfect. On-site adequacy criteria have been proposed to reduce the risk of false negative specimens (89) but have not been prospectively validated.
CP-EBUS TBNA complications
CP-EBUS TBNA is a very safe procedure. Data from the prospectively enrolled American College of Chest Physicians Quality Improvement Registry, Evaluation, and Education (AQuIRE database) included 1,317 patients from 6 hospitals who underwent CP-EBUS TBNA for lymph node sampling (90). 19 patients (1.44%) had a complication with one patient dying from bleeding. Only TBLBx was associated with increased risk on multivariate analysis. Pneumothorax occurred in seven patients and TBLBx was the only variable associated with increased risk; [2.7% TBLBx had pneumothorax vs. 0.2% of those who did not (P=0.001)]. Factors associated with escalation in care included age >70, deep sedation or general anaesthesia, and inpatient status. There were no differences in complication rates between hospitals and outcomes were not associated with procedural volume. ROSE reduced the rate of subsequent TBLBx (P=0.006).
A questionnaire about CP-EBUS TBNA sent to 520 Japan Society for Respiratory Endoscopy-accredited facilities aimed to determine the rate of complications in Japan (40). Of 7,345 CP-EBUS TBNA performed in 210 facilities, 90 complications occurred (1.23%) with hemorrhage being the most frequent complication (0.68%), followed by infection (0.19%) and pneumothorax (0.03%). Only one death occurred (0.01%), the cause of which was cerebral infarction. Equipment related complications were common, with breakage of the ultrasound bronchoscope and puncture needle in 1.33% and 0.2% respectively. These rates are slightly greater than those reported in two meta-analyses (0% and 0.15%) (49,50). Individual case reports detail infectious complications (91-96), needle breakage (97), intramural hematoma (98), and pneumothorax.
Conclusion: peribronchial/peritracheal lesions including mediastinal lymphadenopathy
CP-EBUS TBNA has revolutionised mediastinal staging of lung cancer with sensitivity approaching mediastinoscopy associated with few complications. The widespread adoption of CP-EBUS TBNA internationally by surgeons and physicians is a tribute to its utility, usefulness, simplicity and safety; It is one of the few diagnostic techniques that has truly revolutionized lung cancer diagnostics.
Conclusions
Innovative bronchoscopic techniques are allowing lung cancer to be diagnosed earlier and more accurately in an increasingly non-invasive fashion. Peripheral lesions are targeted by RP-EBUS/GS, ENB, and UB/VB; Mucosal lesions can be identified with NBI and AFB; OCT allows cellular analysis without the need for biopsy; and central peribronchial lesions can be accurately localized and sampled with EBUS TBNA. Whilst the role of established techniques like RP-EBUS/GS, CP-EBUS TBNA, NBI and AFB are well established, data regarding emerging techniques such as ENB and OCT are immature and require further study to establish their utility.
The field of interventional diagnostic pulmonology is rapidly advancing, with the aim of safer, earlier, less invasive and more accurate modalities to identify and diagnose lung cancer earlier. Coupled with newer therapeutics such as stereotactic body radiation and targeted therapeutic agents, it is hoped that lung cancer mortality will no longer be the most common cause of cancer related mortality worldwide.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Jemal A, Thun MJ, Ries LA, et al. Annual report to the nation on the status of cancer, 1975-2005, featuring trends in lung cancer, tobacco use, and tobacco control. J Natl Cancer Inst 2008;100:1672-94. [PubMed]
- Sun J, Garfield DH, Lam B, et al. The value of autofluorescence bronchoscopy combined with white light bronchoscopy compared with white light alone in the diagnosis of intraepithelial neoplasia and invasive lung cancer: a meta-analysis. J Thorac Oncol 2011;6:1336-44. [PubMed]
- Holty JE, Kuschner WG, Gould MK. Accuracy of transbronchial needle aspiration for mediastinal staging of non-small cell lung cancer: a meta-analysis. Thorax 2005;60:949-55. [PubMed]
- Rivera MP, Mehta AC, Wahidi MM. Establishing the diagnosis of lung cancer: diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 2013;143:e142S-65S.
- Wiener RS, Schwartz LM, Woloshin S, et al. Population-based risk for complications after transthoracic needle lung biopsy of a pulmonary nodule: an analysis of discharge records. Ann Intern Med 2011;155:137-44. [PubMed]
- Chen W, Gao X, Tian Q, et al. A comparison of autofluorescence bronchoscopy and white light bronchoscopy in detection of lung cancer and preneoplastic lesions: a meta-analysis. Lung Cancer 2011;73:183-8. [PubMed]
- Lee P, van den Berg RM, Lam S, et al. Color fluorescence ratio for detection of bronchial dysplasia and carcinoma in situ. Clin Cancer Res 2009;15:4700-5. [PubMed]
- Shibuya K, Hoshino H, Chiyo M, et al. High magnification bronchovideoscopy combined with narrow band imaging could detect capillary loops of angiogenic squamous dysplasia in heavy smokers at high risk for lung cancer. Thorax 2003;58:989-95. [PubMed]
- Shibuya K, Nakajima T, Fujiwara T, et al. Narrow band imaging with high-resolution bronchovideoscopy: a new approach for visualizing angiogenesis in squamous cell carcinoma of the lung. Lung Cancer 2010;69:194-202. [PubMed]
- Herth FJ, Eberhardt R, Anantham D, et al. Narrow-band imaging bronchoscopy increases the specificity of bronchoscopic early lung cancer detection. J Thorac Oncol 2009;4:1060-5. [PubMed]
- Zaric B, Perin B, Becker HD, et al. Combination of narrow band imaging (NBI) and autofluorescence imaging (AFI) videobronchoscopy in endoscopic assessment of lung cancer extension. Med Oncol 2012;29:1638-42. [PubMed]
- Vincent BD, Fraig M, Silvestri GA. A pilot study of narrow-band imaging compared to white light bronchoscopy for evaluation of normal airways and premalignant and malignant airways disease. Chest 2007;131:1794-9. [PubMed]
- Zaric B, Becker HD, Perin B, et al. Narrow band imaging videobronchoscopy improves assessment of lung cancer extension and influences therapeutic strategy. Jpn J Clin Oncol 2009;39:657-63. [PubMed]
- Tsuboi M, Hayashi A, Ikeda N, et al. Optical coherence tomography in the diagnosis of bronchial lesions. Lung Cancer 2005;49:387-94. [PubMed]
- Lam S, Standish B, Baldwin C, et al. In vivo optical coherence tomography imaging of preinvasive bronchial lesions. Clin Cancer Res 2008;14:2006-11. [PubMed]
- Tearney GJ, Brezinski ME, Bouma BE, et al. In vivo endoscopic optical biopsy with optical coherence tomography. Science 1997;276:2037-9. [PubMed]
- Hariri LP, Applegate MB, Mino-Kenudson M, et al. Volumetric optical frequency domain imaging of pulmonary pathology with precise correlation to histopathology. Chest 2013;143:64-74. [PubMed]
- Kurimoto N, Miyazawa T, Okimasa S, et al. Endobronchial ultrasonography using a guide sheath increases the ability to diagnose peripheral pulmonary lesions endoscopically. Chest 2004;126:959-65. [PubMed]
- Paone G, Nicastri E, Lucantoni G, et al. Endobronchial ultrasound-driven biopsy in the diagnosis of peripheral lung lesions. Chest 2005;128:3551-7. [PubMed]
- Steinfort DP, Khor YH, Manser RL, et al. Radial probe endobronchial ultrasound for the diagnosis of peripheral lung cancer: systematic review and meta-analysis. Eur Respir J 2011;37:902-10. [PubMed]
- Roth K, Eagan TM, Andreassen AH, et al. A randomised trial of endobronchial ultrasound guided sampling in peripheral lung lesions. Lung Cancer 2011;74:219-25. [PubMed]
- Yamada N, Yamazaki K, Kurimoto N, et al. Factors related to diagnostic yield of transbronchial biopsy using endobronchial ultrasonography with a guide sheath in small peripheral pulmonary lesions. Chest 2007;132:603-8. [PubMed]
- Huang CT, Ho CC, Tsai YJ, et al. Factors influencing visibility and diagnostic yield of transbronchial biopsy using endobronchial ultrasound in peripheral pulmonary lesions. Respirology 2009;14:859-64. [PubMed]
- Shirakawa T, Imamura F, Hamamoto J, et al. Usefulness of endobronchial ultrasonography for transbronchial lung biopsies of peripheral lung lesions. Respiration 2004;71:260-8. [PubMed]
- Fielding DI, Robinson PJ, Kurimoto N. Biopsy site selection for endobronchial ultrasound guide-sheath transbronchial biopsy of peripheral lung lesions. Intern Med J 2008;38:77-84. [PubMed]
- Kurimoto N, Murayama M, Yoshioka S, et al. Assessment of usefulness of endobronchial ultrasonography in determination of depth of tracheobronchial tumor invasion. Chest 1999;115:1500-6. [PubMed]
- Herth F, Ernst A, Schulz M, et al. Endobronchial ultrasound reliably differentiates between airway infiltration and compression by tumor. Chest 2003;123:458-62. [PubMed]
- Makris D, Scherpereel A, Leroy S, et al. Electromagnetic navigation diagnostic bronchoscopy for small peripheral lung lesions. Eur Respir J 2007;29:1187-92. [PubMed]
- Lamprecht B, Porsch P, Pirich C, et al. Electromagnetic navigation bronchoscopy in combination with PET-CT and rapid on-site cytopathologic examination for diagnosis of peripheral lung lesions. Lung 2009;187:55-9. [PubMed]
- Seijo LM, de Torres JP, Lozano MD, et al. Diagnostic yield of electromagnetic navigation bronchoscopy is highly dependent on the presence of a Bronchus sign on CT imaging: results from a prospective study. Chest 2010;138:1316-21. [PubMed]
- Eberhardt R, Anantham D, Ernst A, et al. Multimodality bronchoscopic diagnosis of peripheral lung lesions: a randomized controlled trial. Am J Respir Crit Care Med 2007;176:36-41. [PubMed]
- Eberhardt R, Morgan RK, Ernst A, et al. Comparison of suction catheter versus forceps biopsy for sampling of solitary pulmonary nodules guided by electromagnetic navigational bronchoscopy. Respiration 2010;79:54-60. [PubMed]
- Mahajan AK, Patel SB, Hogarth DK. Electromagnetic navigational bronchoscopy: an effective and safe approach to diagnosing peripheral lung lesions unreachable by conventional bronchscopy. Chest Meeting Abstracts 2008;134:98002.
- Wilson DS, Bartlett RJ. Improved diagnostic yield of bronchoscopy in a community practice: combination of electromagnetic navigation system and rapid on-site evaluation. J Bronchol 2007;14:227-32.
- Dolina MY, Cornish DC, Merritt SA, et al. Interbronchoscopist variability in endobronchial path selection: a simulation study. Chest 2008;133:897-905. [PubMed]
- Asano F, Shindoh J, Shigemitsu K, et al. Ultrathin bronchoscopic barium marking with virtual bronchoscopic navigation for fluoroscopy-assisted thoracoscopic surgery. Chest 2004;126:1687-93. [PubMed]
- Yamamoto S, Ueno K, Imamura F, et al. Usefulness of ultrathin bronchoscopy in diagnosis of lung cancer. Lung Cancer 2004;46:43-8. [PubMed]
- Asahina H, Yamazaki K, Onodera Y, et al. Transbronchial biopsy using endobronchial ultrasonography with a guide sheath and virtual bronchoscopic navigation. Chest 2005;128:1761-5. [PubMed]
- Ishida T, Asano F, Yamazaki K, et al. Virtual bronchoscopic navigation combined with endobronchial ultrasound to diagnose small peripheral pulmonary lesions: a randomised trial. Thorax 2011;66:1072-7. [PubMed]
- Asano F, Aoe M, Ohsaki Y, et al. Complications associated with endobronchial ultrasound-guided transbronchial needle aspiration: a nationwide survey by the Japan Society for Respiratory Endoscopy. Respir Res 2013;14:50. [PubMed]
- Asano F, Matsuno Y, Tsuzuku A, et al. Diagnosis of peripheral pulmonary lesions using a bronchoscope insertion guidance system combined with endobronchial ultrasonography with a guide sheath. Lung Cancer 2008;60:366-73. [PubMed]
- Wang Memoli JS, Nietert PJ, Silvestri GA. Meta-analysis of guided bronchoscopy for the evaluation of the pulmonary nodule. Chest 2012;142:385-93. [PubMed]
- Lemaire A, Nikolic I, Petersen T, et al. Nine-year single center experience with cervical mediastinoscopy: complications and false negative rate. Ann Thorac Surg 2006;82:1185-9. [PubMed]
- Steinfort DP, Irving LB. Patient satisfaction during endobronchial ultrasound-guided transbronchial needle aspiration performed under conscious sedation. Respir Care 2010;55:702-6. [PubMed]
- Silvestri GA, Gonzalez AV, Jantz MA, et al. Methods for staging non-small cell lung cancer: Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 2013;143:e211S-50S.
- Yasufuku K, Chiyo M, Koh E, et al. Endobronchial ultrasound guided transbronchial needle aspiration for staging of lung cancer. Lung Cancer 2005;50:347-54. [PubMed]
- Herth FJ, Eberhardt R, Vilmann P, et al. Real-time endobronchial ultrasound guided transbronchial needle aspiration for sampling mediastinal lymph nodes. Thorax 2006;61:795-8. [PubMed]
- Herth FJ, Ernst A, Eberhardt R, et al. Endobronchial ultrasound-guided transbronchial needle aspiration of lymph nodes in the radiologically normal mediastinum. Eur Respir J 2006;28:910-4. [PubMed]
- Varela-Lema L, Fernández-Villar A, Ruano-Ravina A. Effectiveness and safety of endobronchial ultrasound-transbronchial needle aspiration: a systematic review. Eur Respir J 2009;33:1156-64. [PubMed]
- Gu P, Zhao YZ, Jiang LY, et al. Endobronchial ultrasound-guided transbronchial needle aspiration for staging of lung cancer: a systematic review and meta-analysis. Eur J Cancer 2009;45:1389-96. [PubMed]
- Yasufuku K, Pierre A, Darling G, et al. A prospective controlled trial of endobronchial ultrasound-guided transbronchial needle aspiration compared with mediastinoscopy for mediastinal lymph node staging of lung cancer. J Thorac Cardiovasc Surg 2011;142:1393-400.e1.
- Rintoul RC, Skwarski KM, Murchison JT, et al. Endobronchial and endoscopic ultrasound-guided real-time fine-needle aspiration for mediastinal staging. Eur Respir J 2005;25:416-21. [PubMed]
- Wallace MB, Pascual JM, Raimondo M, et al. Minimally invasive endoscopic staging of suspected lung cancer. JAMA 2008;299:540-6. [PubMed]
- Zhang R, Ying K, Shi L, et al. Combined endobronchial and endoscopic ultrasound-guided fine needle aspiration for mediastinal lymph node staging of lung cancer: a meta-analysis. Eur J Cancer 2013;49:1860-7. [PubMed]
- Annema JT, van Meerbeeck JP, Rintoul RC, et al. Mediastinoscopy vs endosonography for mediastinal nodal staging of lung cancer: a randomized trial. JAMA 2010;304:2245-52. [PubMed]
- Hwangbo B, Lee GK, Lee HS, et al. Transbronchial and transesophageal fine-needle aspiration using an ultrasound bronchoscope in mediastinal staging of potentially operable lung cancer. Chest 2010;138:795-802. [PubMed]
- Herth FJ, Krasnik M, Kahn N, et al. Combined endoscopic-endobronchial ultrasound-guided fine-needle aspiration of mediastinal lymph nodes through a single bronchoscope in 150 patients with suspected lung cancer. Chest 2010;138:790-4. [PubMed]
- Yarmus LB, Akulian J, Lechtzin N, et al. Comparison of 21-gauge and 22-gauge aspiration needle in endobronchial ultrasound-guided transbronchial needle aspiration: results of the American College of Chest Physicians Quality Improvement Registry, Education, and Evaluation Registry. Chest 2013;143:1036-43. [PubMed]
- Nakajima T, Yasufuku K, Takahashi R, et al. Comparison of 21-gauge and 22-gauge aspiration needle during endobronchial ultrasound-guided transbronchial needle aspiration. Respirology 2011;16:90-4. [PubMed]
- Casal RF, Staerkel GA, Ost D, et al. Randomized clinical trial of endobronchial ultrasound needle biopsy with and without aspiration. Chest 2012;142:568-73. [PubMed]
- Lee HS, Lee GK, Lee HS, et al. Real-time endobronchial ultrasound-guided transbronchial needle aspiration in mediastinal staging of non-small cell lung cancer: how many aspirations per target lymph node station? Chest 2008;134:368-74. [PubMed]
- Yung RC, Otell S, Illei P, et al. Improvement of cellularity on cell block preparations using the so-called tissue coagulum clot method during endobronchial ultrasound-guided transbronchial fine-needle aspiration. Cancer Cytopathol 2012;120:185-95. [PubMed]
- Herth FJ, Schuler H, Gompelmann D, et al. Endobronchial ultrasound-guided lymph node biopsy with transbronchial needle forceps: a pilot study. Eur Respir J 2012;39:373-7. [PubMed]
- Feller-Kopman D, Yung RC, Burroughs F, et al. Cytology of endobronchial ultrasound-guided transbronchial needle aspiration: a retrospective study with histology correlation. Cancer 2009;117:482-90. [PubMed]
- Tournoy KG, Carprieaux M, Deschepper E, et al. Are EUS-FNA and EBUS-TBNA specimens reliable for subtyping non-small cell lung cancer? Lung Cancer 2012;76:46-50. [PubMed]
- Nakajima T, Yasufuku K, Nakagawara A, et al. Multigene mutation analysis of metastatic lymph nodes in non-small cell lung cancer diagnosed by endobronchial ultrasound-guided transbronchial needle aspiration. Chest 2011;140:1319-24. [PubMed]
- Garcia-Olivé I, Monsó E, Andreo F, et al. Endobronchial ultrasound-guided transbronchial needle aspiration for identifying EGFR mutations. Eur Respir J 2010;35:391-5. [PubMed]
- Santis G, Angell R, Nickless G, et al. Screening for EGFR and KRAS mutations in endobronchial ultrasound derived transbronchial needle aspirates in non-small cell lung cancer using COLD-PCR. PLoS One 2011;6:e25191. [PubMed]
- Esterbrook G, Anathhanam S, Plant PK. Adequacy of endobronchial ultrasound transbronchial needle aspiration samples in the subtyping of non-small cell lung cancer. Lung Cancer 2013;80:30-4. [PubMed]
- Rintoul RC, Tournoy KG, El Daly H, et al. EBUS-TBNA for the clarification of PET positive intra-thoracic lymph nodes-an international multi-centre experience. J Thorac Oncol 2009;4:44-8. [PubMed]
- Andrade RS, Odell DD, D'Cunha J, et al. Endobronchial ultrasonography (EBUS)--its role in staging of non-small cell lung cancer and who should do it? J Thorac Cardiovasc Surg 2012;144:S9-13. [PubMed]
- Tournoy KG, Rintoul RC, van Meerbeeck JP, et al. EBUS-TBNA for the diagnosis of central parenchymal lung lesions not visible at routine bronchoscopy. Lung Cancer 2009;63:45-9. [PubMed]
- Fielding D, Bashirzadeh F, Nguyen P, et al. Review of the role of EBUS-TBNA for the pulmonologist, including lung cancer staging. Zhongguo Fei Ai Za Zhi 2010;13:410-7. [PubMed]
- Bhatti HA, Bajwa A, Bhatti JA, et al. Diagnostic yield of EBUS-TBNA for the evaluation of centrally located peribronchial pulmonary lesions. J Bronchology Interv Pulmonol 2013;20:107-12. [PubMed]
- Plit ML, Havryk AP, Hodgson A, et al. Rapid cytological analysis of endobronchial ultrasound-guided aspirates in sarcoidosis. Eur Respir J 2012. [Epub ahead of print]. [PubMed]
- Nakajima T, Yasufuku K, Kurosu K, et al. The role of EBUS-TBNA for the diagnosis of sarcoidosis--comparisons with other bronchoscopic diagnostic modalities. Respir Med 2009;103:1796-800. [PubMed]
- Agarwal R, Srinivasan A, Aggarwal AN, et al. Efficacy and safety of convex probe EBUS-TBNA in sarcoidosis: a systematic review and meta-analysis. Respir Med 2012;106:883-92. [PubMed]
- DePew ZS, Gonsalves WI, Roden AC, et al. Granulomatous inflammation detected by endobronchial ultrasound-guided transbronchial needle aspiration in patients with a concurrent diagnosis of cancer a clinical conundrum. J Bronchology Interv Pulmonol 2012;19:176-81. [PubMed]
- Chee A, Tremblay A. EBUS, granulomas, and cancer: what does this represent? J Bronchology Interv Pulmonol 2012;19:171-3. [PubMed]
- Grove DA, Bechara RI, Josephs JS, et al. Comparative cost analysis of endobronchial ultrasound-guided and blind TBNA in the evaluation of hilar and mediastinal lymphadenopathy. J Bronchology Interv Pulmonol 2012;19:182-7. [PubMed]
- Sharples LD, Jackson C, Wheaton E, et al. Clinical effectiveness and cost-effectiveness of endobronchial and endoscopic ultrasound relative to surgical staging in potentially resectable lung cancer: results from the ASTER randomised controlled trial. Health Technol Assess 2012;16:1-75. [PubMed]
- Groth SS, Whitson BA, D’Cunha J, et al. Endobronchial ultrasound-guided fine-needle aspiration of mediastinal lymph nodes: a single institution’s early learning curve. Ann Thorac Surg 2008;86:1104-9. [PubMed]
- Bizekis CS, Santo TJ, Parker KL, et al. Initial experience with endobronchial ultrasound in an academic thoracic surgery program. Clin Lung Cancer 2010;11:25-9. [PubMed]
- Kemp SV, El Batrawy SH, Harrison RN, et al. Learning curves for endobronchial ultrasound using cusum analysis. Thorax 2010;65:534-8. [PubMed]
- Stather DR, Maceachern P, Chee A, et al. Trainee impact on advanced diagnostic bronchoscopy: an analysis of 607 consecutive procedures in an interventional pulmonary practice. Respirology 2013;18:179-84. [PubMed]
- Stather DR, MacEachern P, Chee A, et al. Evaluation of clinical endobronchial ultrasound skills following clinical versus simulation training. Respirology 2012;17:291-9. [PubMed]
- Stather DR, Maceachern P, Rimmer K, et al. Assessment and learning curve evaluation of endobronchial ultrasound skills following simulation and clinical training. Respirology 2011;16:698-704. [PubMed]
- Nakajima T, Yasufuku K, Saegusa F, et al. Rapid on-site cytologic evaluation during endobronchial ultrasound-guided transbronchial needle aspiration for nodal staging in patients with lung cancer. Ann Thorac Surg 2013;95:1695-9. [PubMed]
- Nayak A, Sugrue C, Koenig S, et al. Endobronchial ultrasound-guided transbronchial needle aspirate (EBUS-TBNA): a proposal for on-site adequacy criteria. Diagn Cytopathol 2012;40:128-37. [PubMed]
- Eapen GA, Shah AM, Lei X, et al. Complications, consequences, and practice patterns of endobronchial ultrasound-guided transbronchial needle aspiration: Results of the AQuIRE registry. Chest 2013;143:1044-53. [PubMed]
- Oguri T, Imai N, Imaizumi K, et al. Febrile complications after endobronchial ultrasound-guided transbronchial needle aspiration for intra-pulmonary mass lesions of lung cancer--a series of 3 cases. Respir Investig 2012;50:162-5. [PubMed]
- Huang CT, Chen CY, Ho CC, et al. A rare constellation of empyema, lung abscess, and mediastinal abscess as a complication of endobronchial ultrasound-guided transbronchial needle aspiration. Eur J Cardiothorac Surg 2011;40:264-5. [PubMed]
- Steinfort DP, Johnson DF, Irving LB. Incidence of bacteraemia following endobronchial ultrasound-guided transbronchial needle aspiration. Eur Respir J 2010;36:28-32. [PubMed]
- Parker KL, Bizekis CS, Zervos MD. Severe mediastinal infection with abscess formation after endobronchial ultrasound-guided transbrochial needle aspiration. Ann Thorac Surg 2010;89:1271-2. [PubMed]
- Steinfort DP, Johnson DF, Irving LB. Infective complications from endobronchial ultrasound-transbronchial needle aspiration. Eur Respir J 2009;34:524-5. [PubMed]
- Haas AR. Infectious complications from full extension endobronchial ultrasound transbronchial needle aspiration. Eur Respir J 2009;33:935-8. [PubMed]
- Ozgül MA, Cetinkaya E, Tutar N, et al. An unusual complication of Endobronchial Ultrasound-Guided Transbronchial Needle Aspiration (EBUS-TBNA): the needle breakage. Ann Thorac Cardiovasc Surg 2013. [Epub ahead of print]. [PubMed]
- Botana-Rial M, Núñez-Delgado M, Pallarés-Sanmartín A, et al. Intramural hematoma of the pulmonary artery and hemopneumomediastinum after endobronchial ultrasound-guided transbronchial needle aspiration. Respiration 2012;83:353-6. [PubMed]


