Cardiac regeneration: current therapies—future concepts
Introduction
In 2009 cardiovascular disease (CVD) still accounted for 32.3% of all deaths in the United States and therefore continues to be one of the main causes of death (1). From 1999 to 2009, the rate of death due to CVD has declined, but nevertheless the burden of disease remains high. Although improved medical care and acute management of myocardial infarction have led to a considerable reduction of early mortality rate survivors are susceptible to an increased prevalence of chronic heart failure as they develop scarring followed by ventricular remodeling despite optimum medical care (2,3).
Interestingly, cardiovascular operations and interventional procedures increased by 28% from 2000 to 2010 implicating an enormous cost factor for the healthcare system (1). For 2009, it was estimated that the direct and indirect costs of CVD and stroke add up to about $312.6 billion in the United States, which was more than for any other diagnostic group (1).
The main issue of current pharmacological, interventional or operative therapies is their disability to compensate the irreversible loss of functional cardiomyocytes (4). Hence, the future challenge of cardiovascular therapies will be the functional regeneration of myocardial contractility by novel concepts, like cell based therapy, tissue engineering or reprogramming of scar fibroblasts (5,6).
Current therapies—adult stem and precursor cells
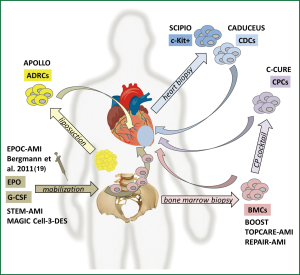
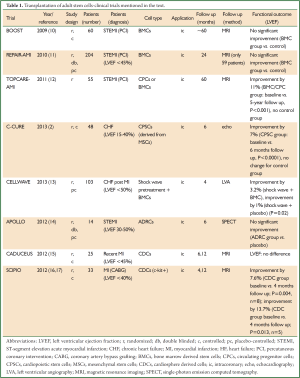
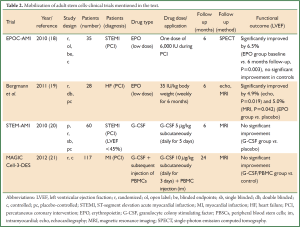
After promising preclinical results using adult stem and precursor cells for cardiac regeneration a rapid clinical translation using autologous bone marrow cells (BMCs) in patients was initiated (7,8). In the last few years numerous clinical trials addressing the transplantation of various adult stem cell populations for cardiac regeneration have been performed. Essential characteristics for the selected adult stem cell populations are the potential to proliferate, migrate and the ability to transdifferentiate into various mature cell types (9). Today, different adult stem cell sources like BMCs, myocardium or adipose tissue derived cells were already used in clinical trials. Beside direct intracoronary or intramyocardial transplantation of adult stem cells into the heart mobilization of autologous progenitor cells by administration of different cytokines [i.e., erythropoietin (EPO) or granulocyte colony stimulating factor (G-CSF)] were also evaluated in first clinical trials (summarized in Figure 1 and Tables 1,2).
Full Table
Full Table
Bone marrow derived stem cells (BMCs)
The bone marrow as an autologous source for adult stem cells is easily accessible and renewable. Significant improvement in left ventricular function after transplantation of BMCs has been reported in animal models of myocardial infarction (22,23). The BMCs consist of different subpopulations like BM-hematopoietic stem cells (BM-HSCs, ~2-4%), mesenchymal stem cells (also termed BM-mesenchymal stromal cells, BM-MSCs, ~0.1%), BM-endothelial progenitor cells (BM-EPCs) or side population cells (9). BM-HSCs give rise to lymphoid and myeloid lineages during embryonic development, whereas BM-MSCs develop into osteoblasts, chondrocytes and skeletal myocytes (24). BM-MSCs are physiologically situated in the stromal adherent fraction of the bone marrow where they maintain homeostatic turnover of BM-HSCs (25). Both multipotent precursor cell populations are self-renewing, grow clonogenic, and can be induced to differentiate into a cardiomyocyte phenotype in vitro (26-28). BM-HSCs are typically characterized by surface receptors like CD133, CD34 or CD117 (c-KIT) whereas BM-MSCs express CD105, CD73 and CD90 but lack CD34, CD45 and others (28). BM-EPCs have embryonic angioblastic properties and are able to promote the repair of damaged endothelium (29).
Multiple clinical trials with various results have been performed with BM-HSCs, BM-MSCs and BM-EPCs (9,30-33). In a recently published metastudy (3) combining the results of 50 studies from 2003 to 2011 BMC transplantation improved the left ventricular ejection fraction (LVEF) by an average of 3.96% and infarct size was reduced by 4.03% compared with the standard treatment groups (P=0.00001). These benefits were found in acute myocardial infarction as well as in chronic ischemic heart disease patients.
A few years ago first long term results of stem cell transplantation clinical trials were published (10-12). The key question was whether short-term improvements were persistent over the long term. Two trials analyzing BMC transplantation for cardiac repair, TOPCARE-AMI and BOOST, have published their five year follow-up results. For the BOOST trial no significant difference in LVEF between the control and the BMC-treated group was detectable (10). The five year results of TOPCARE-AMI, however, confirmed a persistence of the beneficial effects on LV function (12). LVEF was improved by 11% (P<0.001) at 5 years. Though, it has to be mentioned that the trial design lacks a placebo-controlled treatment group. The REPAIR-AMI trial, as the largest double-blind, placebo-controlled trial with 204 patients, presented its 2-year follow-up data and found no ameliorated LVEF in the BMC- compared to the placebo treated group (11).
Owing to inconsistent functional outcomes of BMC transplantation further methodological refinements to improve the efficiency of BMC delivery were undertaken. The C-CURE trial advanced the paradigm of lineage specification in stem cell therapy (2). BM-MSCs were harvested from the bone marrow and then driven into cardiopoietic stem cells (CPCs) under addition of a cardiopoietic cytokine cocktail during extra-corporal cultivation. The CPCs were then endomyocardially delivered into 48 patients with a history of myocardial infarction. After six months an increase of LVEF by 7% was observed in the cell therapy group as determined by echocardiography whereas the standard care group only showed an improvement of 0.2% (P<0.0001). The study is limited since it is neither blinded nor placebo-controlled. However, the paradigm of pretransplant lineage specification might proof as an interesting rationale for further clinical validation.
The CELLWAVE study combined the delivery of BM-MSCs with a cardiac shock wave pretreatment (low-energy shock wave under continuous electrocardiographic trigger) in order to improve homing of the delivered cells while increasing the local expression of chemoattractants such as stromal cell-derived factor 1 (SDF-1) and vascular endothelial growth factor (VEGF) (13). The shock wave BMC group showed an absolute change in LVEF of +3.2% after four months whereas the shock wave placebo infusion group only displayed a 1% improvement (P=0.02).
In summary, clinical trials have documented that BMC transplantation is safe and feasible but the ambitious goal of cardiac functional recovery after myocardial infarction remains elusive (3). Outcomes in terms of efficacy are inconsistent and in most series only transient (15). However, several clinical trials are announced and ongoing (34).
Adipose tissue derived regenerative cells (ADRCs)
More than a decade ago, it was shown that human lipoaspirates contain multipotent cells with a differentiation potential beyond that of the adipocytic lineage (26). These ADRCs share many properties with BM-MSCs (35). Adipose tissue as an autologous source for therapeutic regenerative cells is particularly advantageous since it can be harvested in relatively large quantities by liposuction (35) and contains up to 2,500 times more MSC-like cells than freshly isolated bone marrow (14). The considerably higher amount of MSC-like cells in adipose tissue would obviate a time consuming expansion prior to application for therapeutic assignment as it is necessary for BM-MSCs. A preclinical study in pig analyzing the effects of ADRC administration after acute myocardial infarction demonstrated functional improvement (36). The APOLLO trial is a randomized, double-blind and placebo-controlled study to investigate the safety and feasibility of intracoronary infusion of ADRCs (20 million cells) in 14 acute phase patients with large ST-segment elevation acute myocardial infarction (STEMI) (14). SPECT analysis demonstrated a trend but no significant improvement of LVEF compared to the placebo group. Nevertheless, a randomized, placebo-controlled phase IIb/III trial (ADVANCE) with an intracoronary infusion of two doses of ADRCs in up to 375 patients with STEMI is planned (14).
Mobilization of progenitor cells
A different strategy for cardiac regeneration by adult stem cells is the mobilization of progenitor cells from the bone marrow by administration of growth factors or cytokines such as fibroblast growth factor (FGF), VEGF, EPO or G-CSF (37).
EPO is a glycoprotein hormone that controls erythropoiesis and is known to increase the oxygen-carrying capacity of the blood (38). Hypoxic ischemic cardiomyocytes and vascular endothelial cells have surface EPO receptors which make them potential targets for EPO treatment (39). In animal models high-dose EPO administration led to reduced infarct size and functional preservation (40,41). However, clinical studies of single high doses of EPO following acute myocardial infarction were disappointing (42). Current clinical trials, like EPOC-AMI or the study of Bergmann et al., were sought to investigate the low-dose application of EPO (either short- or long-term) (18,19). The EPOC-AMI trial (18) showed a significantly improved LVEF in the short-term low-dose EPO group of 6.5% (P=0.003). Bergmann et al. (19) reported an enhanced LVEF for the low-dose long-term EPO group [echo: (5.2±2.0)%, P=0.013; MRI: (3.1±1.6)%, P=0.124]. No significant improvements were found for the placebo groups.
G-CSF is secreted by monocytes, fibroblasts, and endothelial cells and is involved in myeloid development and neutrophil maturation (43). In animal models improved haemodynamic function and infarct remodeling could be shown (44,45). Two meta-analyses regarding the role of G-CSF in cardiac repair after myocardial infarction demonstrated that G-CSF failed to improve LV function (46,47). The STEM-AMI trial also failed to show LVEF improvement six months after G-CSF administration (20).
The ability of BMC mobilization either by EPO or G-CSF to promote cardiac repair remains controversial. Alternative approaches like the MAGIC Cell-3-DES trial combined G-CSF stimulation with an intramyocardial application of peripheral blood stem cells (PBSCs) (21). After two years LVEF was improved by 2.8% (P=0.045) in the G-CSF/cell group but not in the control group. Future efforts are made by combining G-CSF with DPP-IV (CD26/dipeptidylpeptidase IV) inhibitors to increase the homing of stem cells by inhibiting the degradation of SDF1alpha, an important homing related surface receptor (48). Several DPP-IV inhibitors have already been chemically approved for anti-diabetic treatment (49,50). The SITAGRAMI-Trial intends to analyze the impact of G-CSF and Sitagliptin (DPP-IV inhibitor) in patients suffering from acute myocardial infarction (48).
Other cytokines, such as FGF, VEGF, growth hormone (GH) or insulin-like growth factor (IGF) have been applied in various randomized, controlled clinical trials, unfortunately without reproducing the efficacy observed in pre-clinical investigations [summarized in (37)].
Mechanisms of stem cell action in the diseased heart
Different regenerative mechanisms of transplanted cells within the injured myocardium have been described (9). (I) Transplanted stem cells could directly transdifferentiate into cardiomyocytes; (II) Paracrine effects could induce growth of resident cardiomyocytes; (III) Resident endogenous myocardial stem cells may be stimulated; (IV) Cell fusion between transplanted cells and resident cardiomyocytes could be induced. Currently, it is understood that the predominant working mechanism of BMC or ADRC therapy is mediated through a paracrine release of antiapoptotic, immunomodulatory, and proangiogenic host- and cell-derived factors (51). Hence, the moderate positive effects of stem cell delivery to impaired myocardium are more likely due to enhanced neovascularization rather than a formation of new cardiac muscle. A direct transdifferentiation into cardiomyocytes or a cell fusion between the transplanted stem cells and resident myocytes were considered unlikely.
Cardiac derived stem cells
Current clinical studies also use different populations of autologous cardiac progenitor cells, directly derived from the human adult heart (52). To yield cardiosphere derived cells (CDCs) percutaneous endomyocardial biopsy specimens are grown in primary culture where they develop multicellular clusters known as cardiospheres (53). CDCs are a natural mixture of stromal, mesenchymal, and progenitor cells expressing distinct surface receptors (e.g., c-Kit) (53). In vitro CDCs grow clonogenic and have multilineage potential. Safety and efficacy of CDC transplantation has been demonstrated in preclinical studies (53,54). In a murine model of myocardial infarction the functional outcome of different stem cell type transplantations, including BM-MSCs, ADRCs and CDCs were compared (55). CDCs were clearly superior in terms of ischemic tissue preservation, anti-remodelling effects and functional benefits (55).
The results of the CADUCEUS trial, a phase I randomized trial, were recently published (15). CDCs were derived from right ventricular endomyocardial biopsies and expanded for about five weeks before retransplantation. The CDCs were delivered by intracoronary infusion into the infarct related artery in 17 patients with recent myocardial infarction (3-5 months). Outcomes were compared with eight patients who received conventional medical therapy. No difference with respect to LVEF-change could be detected after six months but infarct size was significantly reduced in the cell treated group (by 7.7%, after 6 months and by 12.3% after 12 months, P=0.001) (15). Unfortunately the study could not be performed in a blinded manner because of ethical concerns regarding right ventricular biopsy on the controls.
Cardiac stem cells, positive for the tyrosine kinase receptor c-KIT, were first described in 2003 (56) as a resident population of stem cells in adult mammalian myocardium. These stem cells are capable of differentiating into cardiomyocytes, vascular smooth muscle cells and endothelial cells, respectively (57). As these cells are furthermore self-renewing and grow clonogenic, they fulfill all criteria of a tissue-specific stem cell (57). Several preclinical studies could demonstrate functional improvements after transplantation of c-Kit positive stem cells into ischemic hearts (56,58,59).
As a milestone in the history of cardiac stem cell therapy the first-in-man trial, the randomized, open-label SCIPIO trial (16,17) intended to analyze the feasibility, safety and efficacy of an intracoronary infusion of c-KIT+/(LIN-) CDCs into patients with sustained myocardial infarction. After one year LVEF was analyzed by MRI in only five treated patients and was found to be improved by 12.3% (compared to baseline, P=0.013). Mortality or major adverse cardiac events (MACE) following cell therapy did not emerge in this study.
The benefits of CDC therapy may be mediated through activation of endogenous regenerative pathways or even through a direct transdifferentiation of delivered cells into cardiomyocytes (15) whereas other studies claim that even CDCs exert most of their beneficial effects via indirect paracrine mechanisms (59,60).
Owing the different study designs and patient populations a direct comparison of most clinical studies is generally difficult. However, the functional outcome of CDC-trials appears slightly more encouraging when compared with BMC/ADRC trials.
Pending issues of current stem cell trials
Most clinical trials using different adult stem cell transplantation or mobilization strategies have proofed safety and feasibility. In order to optimize therapy protocols further efforts have to be made to identify underlying mechanisms of stem cell migration, differentiation and myocardial regeneration. Other important issues are the optimal stem cell type and number, timing of their delivery and application route, respectively.
Above all, the most effective stem cell type for cardiac regeneration has to be defined. BMCs and ADRCs both seem to act in a paracrine way to improve cardiac function whereas CDCs are believed to have some capacity to transdifferentiate into cardiomyocytes. Furthermore, it remains open whether the use of allogeneic cells can be of clinical relevance in the future. Not only that autologous cells have to be expanded for extensive time periods excluding their use in the acute setting, the expansion is furthermore expensive and their quality will vary depending on the patient (5).
The optimal timing of cell delivery after myocardial infarction is potentially one of the main issues in terms of cell homing and survival. Myocardial infarction is an ischemic event directly followed by a significant inflammatory reaction accompanied by the secretion of cytokines and additional growth factors by macrophages or other immune cells (61). A transplantation of unprotected cells into this environment results in significant cell death (62). It was speculated that delivered stem cells even take part in the inflammation cascade than in the formation of new myocardium and vessels (28). At the moment the optimal time point for stem cell application seems to be within the first month after myocardial infarction (28).
The optimal form and route of cell delivery into the ischemic heart remains another major challenge. Intramyocardial delivery with several injections could be a risk factor for triggering ventricular arrhythmias. Preferring an intracoronary cell administration might reduce the risk for arrhythmias but may foster cell loss.
All these issues motivate future research for improving delivery methods, find more potent cell types and better strategies to boost cell engraftment (63).
Future concepts
Since studies with adult stem/precursor cells and mobilization therapies did by now not lead to the desired results in terms of a full functional restoration further strategies are under investigation—but so far have not been translated into clinical reality. Transplantation of alternative cell sources with “true” regenerative properties, tissue engineering with various scaffold materials and different cells for colonization, stimulation of resident stem or progenitor cell sources or a reprogramming of scar tissue back into functional myocardium are promising approaches.
Novel cell sources for cardiac regeneration with “true” regenerative properties
Embryonic stem cells (ESCs) are undifferentiated cells obtained from the inner cell mass of blastocysts revealing unlimited self-renewal capacity and pluripotency. They have the potential to develop into derivatives of all three germ layers: endoderm, mesoderm and ectoderm (64). These properties made ESCs particularly interesting for cardiac regeneration (33).
A few years ago, a spectacular discovery overcame ethical and presumably immune rejection issues of ESCs. In 2012 the Nobel Prize in Physiology or Medicine was awarded to Sir John B. Gurdon and Shinya Yamanaka for the discovery that mature somatic cells can be reprogrammed to a pluripotent stage (65,66). Takahashi and Yamanaka (65,67) generated so called induced pluripotent stem cells (iPSCs) by retroviral delivery and subsequent overexpression of a four-gene-set (Klf4, c-Myc, Oct4 and Sox2) from murine and human dermal fibroblasts (Figure 2). IPSCs exhibit similar pluripotency features and differentiation capacity like ESCs (64). Although teratoma formation is still of concern for iPSCs this milestone in cell biology opened a new era in regenerative and personalized medicine (68). The risk of teratoma formation can be reduced by predifferentiating pluripotent cells (ESCs, iPSCs) into cardiomyocytes or cardiac progenitor cells before transplantation (Figure 2). Usually only a small fraction of pluripotent stem cells spontaneously differentiates into cardiomyocytes (5-15%) with relative mature structural and functional properties (64). However, the yield can be improved by directing the differentiation into the cardiac lineage by choosing appropriate culture conditions (69).
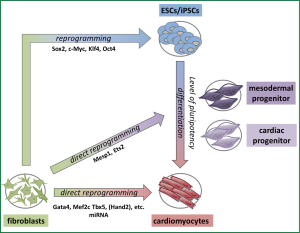
Finally, questions remain concerning the ideal maturity level for cells to be implanted. In the course of differentiation from the level of pluripotency to maturity a cell traverses several progenitor stages (Figure 2). Different cardiac progenitor cell populations have been characterized by the expression of specific transcription factors like Flk1, Isl1 or Nkx2.5 (70-72). All of them have the potential to differentiate into various cardiac lineages like endothelial cells, smooth muscle cells or myocytes. Current studies try to induce cardiac progenitor cells from fibroblasts for regenerative purposes. It has been shown that a lentiviral overexpression of only two transcription factors, Ets2 and Mesp1, is sufficient to reprogram fibroblasts into cardiac progenitor-like cells (73) (Figure 2). Induced cardiac progenitor cells (iCPCs) would be advantageous for transplantational medicine since they are still able to proliferate. However, they do not show the same risk of teratoma formation like pluripotent cells.
Transplantation of cells with “true” regenerative potential
Several of the above mentioned novel cell sources, like ESCs, iPSCs or their derivatives have been transplanted after cardiac myocardial ischemia in different animal models (Figure 3).
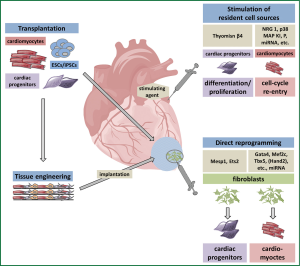
The efficacy of ESC transplantation after myocardial infarction has early been demonstrated in animal studies (74,75). So far, ESCs have not been used clinically since, aside from ethical and political concerns, ESCs would be immunologically incompatible to the patient and entail the risk of teratoma formation (32).
The use of iPSCs in infarct repair has also been shown in different preclinical models (76-78). First of all therapeutic benefits of iPSC-based treatment have been established in small-animal infarction models (76,77). LVEF was improved two weeks after intramyocardial delivery of murine iPSCs compared to fibroblast transplantation (76) or PBS injection (77) after induction of myocardial infarction in mouse models. Functional improvement could also be achieved by intramyocardial transplantation of undifferentiated pig iPSCs in a porcine infarction model (78). Furthermore, infarct size could be decreased by iPSC transplantation in the mentioned animal studies. In contrast to ESCs iPSCs represent a potential autologous source of cells with an ESC-like phenotype and thus would be particularly useful for transplantation medicine (79).
For transplantation of pluripotent stem cell derived cardiomyocytes it is important to efficiently purify and select the developed cardiomyocytes to get a homogenous cell population (64). Cardiac function in infarcted rat hearts could be ameliorated without teratoma formation by transplanting predifferentiated cardiomyocytes (80,81). However, efficient engraftment and survival continue to be an issue in preclinical studies (82). Furthermore there is growing evidence that pluripotent stem cell derived cardiomyocytes differentiated from ESCs or iPSCs are rather immature. Whether they can undergo further maturation within the host tissue remains to be determined (83).
A couple of challenges remain before these technologies can be translated successfully from bench to bedside. First of all the high risk of insertional mutagenesis using viral vectors for reprogramming has to be considered (84). Nonintegrating reprogramming techniques by direct protein delivery, miRNA or episomal vectors were already developed but often with lower efficacy (85-87). Moreover, epigenetic features (methylation patterns of the genomic DNA) were observed to be reminiscent of the original somatic cell and therefore indicate incomplete reprogramming (88). Another obstacle is the time needed to derive patient specific iPSCs so that they cannot be used under urgent circumstances (64).
Direct reprogramming-new myocardium from scar tissue
The iPSC technology builds the scientific basis for alternative reprogramming strategies. In 2010 Ieda and colleagues first published the direct reprogramming of fibroblasts into cardiomyocytes without an intermediate pluripotent stage by Gata4, Mef2c, Tbx5 (GMT) (89) (Figure 2). This raises the possibility to directly generate functional myocardium by reprogramming postinfarct scar tissue, which largely consists of fibroblasts (Figure 3). Genetic lineage tracing models were used to demonstrate that resident non-myocytes in the infarcted murine heart could be reprogrammed into induced cardiomyocyte-like cells (iCMs) by retroviral, intramyocardial GMT delivery in vivo (90). After three month a reduced infarct size and a modestly attenuated cardiac dysfunction could be documented. In parallel a second group around Eric Olson came up with a more efficient reprogramming strategy combining GMT with Hand2 (91). They succeeded in improving cardiac function and reducing adverse ventricular remodeling after myocardial infarction by local delivery of GMTH in a mouse model. However, a recent publication (92) indicated that the process of reprogramming fibroblasts to cardiomyocytes by GMT is insufficient. Cardiac and tail tip fibroblasts of multiple transgenic reporter mice were reprogrammed by GMT with varying success. It could be clearly shown that fibroblasts, especially cardiac fibroblasts, were pushed to a cardiac fate (high expression levels of, e.g., Tbx20 and Tnnt2). However, no electrophysiological competent and molecular mature cardiomyocytes could be generated. The authors concluded that GMT induced only a partial cardiac gene program leading to an insufficient reprogramming.
Recently, a combination of microRNAs (miRNAs: 1, 133, 208 and 499) was identified to be capable to reprogram murine fibroblasts to cardiomyocyte-like cells (93) albeit with a low efficiency (1.5-7.7% conversion rate). The effects of miRNA on gene expression can be powerful as a single miRNA may target multiple pathways simultaneously. The most recent study combined a subset of four transcription factors (Gata4, Hand1, Tbx5 and myocardin) with two miRNAs [1, 133] to successfully reprogram human fibroblasts to cardiomyocyte-like cells (94).
The success of direct reprogramming approaches may vary due to the different choice of mouse models and evaluation methods of cardiac phenotypes. Hence, all approaches had significant issues with efficacy that have to be overcome before a therapeutical application becomes feasible.
Tissue engineering-concepts for generation of cardiac tissue
Tissue engineering is actually an extension of cell transplantation in combination with a variety of scaffolds as one is more and more aware that the three-dimensional microenvironment plays an important role in cell differentiation and especially maturation (95) (Figure 3). As single cell approaches are limited in terms of high cellular wash out and low integration rates tissue engineering approaches may become increasingly important. Zhang and co-workers (95) compared the maturation of human embryonic stem cell-derived cardiomyocytes (hESC-CMs) in 2D monolayers and 3D fibrin-based patch cultures. In 3D patches hESC-CMs exhibited significantly higher conduction velocities, longer sarcomeres and an enhanced expression of genes associated with contractile function (like cTNT, αMHC, CASQ2, SERCA2) when compared to 2D cell culture. So far, a variety of scaffolds and cell sources have been used to generate functional cardiac tissue with diverging efficiency in vitro [for detailed review see (61,96)].
Furthermore, mechanical and electrical stimulation were found to markedly improve the structural organization and contractile properties of engineered cardiac constructs and resulted in a more mature cardiac muscle structure (61,97,98). Zimmermann and associates have demonstrated that the transplantation of large force-generating engineered heart tissues formed by mechanical pretreatment from neonatal rat heart cells, liquid collagen and matrigel led to undelayed electrical coupling and structural improvement in infarcted rat hearts (99).
For thicker engineered heart tissue constructs in vitro it is also necessary to ensure oxygen supply to keep settling cells viable. A study about the penetration depth of oxygen in non-perfused cardiac constructs demonstrated that myocytes could only survive within the first 100 µm of the construct thickness.
Current tissue engineering techniques have mainly been tested for functional improvements in rat models, but whether these approaches can be transferred to large animal models or even to human patients remains elusive (100).
Stimulation of endogenous cardiac repair
It has been demonstrated that the neonatal murine heart holds a remarkable regenerative capacity. Apical excision resulted in a total recovery of the removed tissue by myocyte proliferation without scarring (101). Bergmann and colleagues found that the human adult heart also generates new cardiomyocytes during a life-span with an approximate turnover rate of 1% per year (age 25), declining to 0.45% by the age of 75 (102). The Cold War nuclear bomb tests caused a significant increase of 14C-concentration in the atmosphere that gradually dropped after termination of the tests. Since the genomic DNA of every cell reflects the atmospheric 14C-concentration at the time point of its generation the investigators were able to date the age of cardiac myocytes by the amount of 14C in their nuclei.
Hence, the paradigm of terminally differentiated quiescent myocytes is obsolete since the mammalian adult heart seems to conserve a certain capacity for cardiomyocyte turnover (4). Evidence exists that new cardiomyocytes could be generated by one of the following mechanisms: a division of pre-existing mature cardiomyocytes, a dedifferentiation of cardiomyocytes followed by amplification, or a differentiation of resident progenitor cells (4).
In a transgenic mouse model combined with stable isotope labelling it was recently shown that the genesis of new cardiomyocytes in the adult heart occurs by the division of pre-existing cardiomyocytes and that the same mechanism is engaged in the regenerative processes after myocardial infarction (103). The stimulation of cell-cycle reentry of preexisting cardiomyocytes might therefore be an approach for cardiac regeneration. Pharmacological agents like a p38 MAP kinase inhibitor, the growth factor neuregulin-1 or the extracellular matrix signaling protein periostin showed encouraging results in vitro but they failed to replicate their beneficial effects to the full extent in vivo (104-106). Slight improvements in cardiac function after infarction could merely be achieved by neuregulin-1 (105) or by combining the inhibition of p38 MAP kinase with FGF1 (104) (Figure 3).
As regulatory pathways that govern heart development and growth are modulated, amongst others, by numerous miRNAs (107), the delivery of miRNAs into the injured heart could be another therapeutic approach to stimulate cardiomyocyte proliferation (Figure 3). Eulalio and coworkers identified several miRNAs inducing cardiac regeneration by a functional screening using a whole genome miRNA library (108). They showed that injection of synthetic miRNAs miR-590-3p and miR-199a-3p into the neonatal mouse heart led to increased cardiomyocyte, but not cardiac fibroblast, proliferation. The adenoviral delivery of these miRNAs into the adult murine heart after myocardial infarction reduced fibrotic scar size and improved cardiac function (108).
Several publications about the regenerative capacity of the hearts of newts, zebrafish or neonatal mice showed that pre-existing cardiomyocytes adjacent to the site of injury undergo a process of dedifferentiation characterized by molecular and/or cytoskeletal changes (101,109,110). Dedifferentiated myocytes can re-express gene markers of immaturity, like Gata4, and disassemble sarcomeric structures (101,110,111). Additionally they pass a proliferative phase evident by the expression of DNA synthesis markers before re-differentiating into mature myocytes.
Furthermore, various populations of endogenous cardiac stem or progenitor cells with the potential to create cardiomyocytes have been identified in the postnatal mammalian heart. They are characterized by different surface receptors like c-Kit (56), Sca-1 (112), the transcription factor Isl1 (113) or by their ability to efflux the fluorescent vital dye Hoechst 33342 (114). Recently, the epicardium, a mesothelial cell layer encapsulating the heart, was presented as another source of progenitor and paracrine cells contributing to heart development and maybe also to regeneration after cardiac injury (115,116). Stimulation of endogenous regeneration may become feasible by using small molecules or growth factors (29) (Figure 3). An activation of local signaling pathways could further stimulate vasculogenesis and angiogenesis and by this improve local blood flow (29). Thymosin β4 is implicated in the reorganization of the actin cytoskeleton, a process fundamentally required for cell migration and cytokinesis and is regarded as a potential candidate for stimulating resident cardiac progenitors, especially of epicardial origin (117) (Figure 3). Smart and colleagues stimulated adult mice with peritoneal Thymosin β4 injections before induction of myocardial infarction resulting in a significant increased WT1+ epicardial progenitor population two days after the ischemic event (118). Thymosin β4 treatment following myocardial infarction in a mouse model did exhibit overall cardioprotective effects but failed to show differentiation of epicardial cells into cardiomyocytes (119).
However, the question whether myocyte proliferation or resident cardiac progenitor cell stimulation, especially in elderly patients, is sufficient for efficient myocardial regeneration remains open. Further research is needed to identify paracrine signaling pathways involved in activation and recruitment of endogenous cardiomyocyte progenitors (4). A more detailed characterization of the molecular, phenotypic and functional identity of various endogenous cardiac progenitor cell populations in conjunction with the mapping of cell fate by implementing transgene lineage tracing models both in vitro and in vivo is required to pave the way for “true” myocardial regenerative medicine.
Summary and perspective
In summary, different cell types have been studied in clinical trials with good safety data but heterogeneous results in terms of efficacy. Several short- and long-term challenges like the optimal stem cell type, the optimal timing of stem cell delivery and the optimal application route remain open. Furthermore, integration of the grafts including control of vascularization have to be improved in the future (5). Finally it has to be taken into account, that several legal and practical issues need to be resolved before patients can be treated by stem cell technologies in a consistent and cost-effective manner (68).
Currently, various new cell sources with “true” regenerative potential are on trial. Multipotent ESCs and iPSCs can differentiate into any cell-type but risks of teratoma formation in case of direct transplantation have to be considered. ESCs additionally implicate ethical and immunological concerns. Pluripotent stem cell derived cardiac progenitor cells and cardiomyocytes offer versatile possibilities for regenerative strategies but entail risks concerning sufficient purity or maturity level since it remains to be determined whether further maturation takes place within the host tissue. Functional engraftment and survival of transplanted cells is still an issue that may be overcome by tissue-engineering techniques. A significant obstacle of all these therapies is the time needed to derive sufficient cells or tissue grafts since they cannot be used under urgent circumstances, such as an acute myocardial infarction or a rapidly progressive heart failure (64). Further approaches try to reprogram scar tissue into new functional myocardium by the administration of various transcription factors or miRNA. Unfortunately most of the current reprogramming strategies are realized by viral delivery of the respective factors entailing the risk of severely changing original genomics of the targeted cell. But even by using nonintegrating methods high levels of mutational changes were still observed (84). Finally, scientists intended to stimulate endogenous cell sources like existing cardiomyocytes or resident cardiac progenitor cells by growth factors or small molecules to initiate cardiac regeneration after injury. Unfortunately, significant issues remain with current approaches in regenerative strategies before the shift from bench to bedside can succeed.
In general, all described current and future strategies for cardiac regeneration are restricted by the limited understanding of basic principles, pathways and transcription factor networks that contribute to vertebrate heart development and cardiomyocyte cell differentiation as well as maturation. Since the heart is a most complex organ that requires uninterrupted function and therefore seamless integration of transplanted cells, the creation of functional heart tissue will very likely necessitate a combination of approaches (107). Despite a decade of numerous adult stem cell clinical trials we are still at the beginning of a promising journey in the field of regenerative cardiac medicine.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Go AS, Mozaffarian D, Roger VL, et al. Heart disease and stroke statistics--2013 update: a report from the American Heart Association. Circulation 2013;127:e6-e245. [PubMed]
- Bartunek J, Behfar A, Dolatabadi D, et al. Cardiopoietic stem cell therapy in heart failure: the C-CURE (Cardiopoietic stem Cell therapy in heart failURE) multicenter randomized trial with lineage-specified biologics. J Am Coll Cardiol 2013;61:2329-38. [PubMed]
- Jeevanantham V, Butler M, Saad A, et al. Adult bone marrow cell therapy improves survival and induces long-term improvement in cardiac parameters: a systematic review and meta-analysis. Circulation 2012;126:551-68. [PubMed]
- Steinhauser ML, Lee RT. Regeneration of the heart. EMBO Mol Med 2011;3:701-12. [PubMed]
- Laflamme MA, Murry CE. Heart regeneration. Nature 2011;473:326-35. [PubMed]
- Assmus B, Zeiher AM. Early cardiac retention of administered stem cells determines clinical efficacy of cell therapy in patients with dilated cardiomyopathy. Circ Res 2013;112:6-8. [PubMed]
- Strauer BE, Brehm M, Zeus T, et al. Intracoronary, human autologous stem cell transplantation for myocardial regeneration following myocardial infarction. Dtsch Med Wochenschr 2001;126:932-8. [PubMed]
- Stamm C, Westphal B, Kleine HD, et al. Autologous bone-marrow stem-cell transplantation for myocardial regeneration. Lancet 2003;361:45-6. [PubMed]
- Strauer BE, Steinhoff G. 10 years of intracoronary and intramyocardial bone marrow stem cell therapy of the heart: from the methodological origin to clinical practice. J Am Coll Cardiol 2011;58:1095-104. [PubMed]
- Meyer GP, Wollert KC, Lotz J, et al. Intracoronary bone marrow cell transfer after myocardial infarction: 5-year follow-up from the randomized-controlled BOOST trial. Eur Heart J 2009;30:2978-84. [PubMed]
- Assmus B, Rolf A, Erbs S, et al. Clinical outcome 2 years after intracoronary administration of bone marrow-derived progenitor cells in acute myocardial infarction. Circ Heart Fail 2010;3:89-96. [PubMed]
- Leistner DM, Fischer-Rasokat U, Honold J, et al. Transplantation of progenitor cells and regeneration enhancement in acute myocardial infarction (TOPCARE-AMI): final 5-year results suggest long-term safety and efficacy. Clin Res Cardiol 2011;100:925-34. [PubMed]
- Assmus B, Walter DH, Seeger FH, et al. Effect of shock wave-facilitated intracoronary cell therapy on LVEF in patients with chronic heart failure: the CELLWAVE randomized clinical trial. JAMA 2013;309:1622-31. [PubMed]
- Houtgraaf JH, den Dekker WK, van Dalen BM, et al. First experience in humans using adipose tissue-derived regenerative cells in the treatment of patients with ST-segment elevation myocardial infarction. J Am Coll Cardiol 2012;59:539-40. [PubMed]
- Makkar RR, Smith RR, Cheng K, et al. Intracoronary cardiosphere-derived cells for heart regeneration after myocardial infarction (CADUCEUS): a prospective, randomised phase 1 trial. Lancet 2012;379:895-904. [PubMed]
- Bolli R, Chugh AR, D’Amario D, et al. Cardiac stem cells in patients with ischaemic cardiomyopathy (SCIPIO): initial results of a randomised phase 1 trial. Lancet 2011;378:1847-57. [PubMed]
- Chugh AR, Beache GM, Loughran JH, et al. Administration of cardiac stem cells in patients with ischemic cardiomyopathy: the SCIPIO trial: surgical aspects and interim analysis of myocardial function and viability by magnetic resonance. Circulation 2012;126:S54-64. [PubMed]
- Taniguchi N, Nakamura T, Sawada T, et al. Erythropoietin prevention trial of coronary restenosis and cardiac remodeling after ST-elevated acute myocardial infarction (EPOC-AMI): a pilot, randomized, placebo-controlled study. Circ J 2010;74:2365-71. [PubMed]
- Bergmann MW, Haufe S, von Knobelsdorff-Brenkenhoff F, et al. A pilot study of chronic, low-dose epoetin-{beta} following percutaneous coronary intervention suggests safety, feasibility, and efficacy in patients with symptomatic ischaemic heart failure. Eur J Heart Fail 2011;13:560-8. [PubMed]
- Achilli F, Malafronte C, Lenatti L, et al. Granulocyte colony-stimulating factor attenuates left ventricular remodelling after acute anterior STEMI: results of the single-blind, randomized, placebo-controlled multicentre STem cEll Mobilization in Acute Myocardial Infarction (STEM-AMI) Trial. Eur J Heart Fail 2010;12:1111-21. [PubMed]
- Kang HJ, Kim MK, Lee HY, et al. Five-year results of intracoronary infusion of the mobilized peripheral blood stem cells by granulocyte colony-stimulating factor in patients with myocardial infarction. Eur Heart J 2012;33:3062-9. [PubMed]
- Orlic D, Kajstura J, Chimenti S, et al. Bone marrow cells regenerate infarcted myocardium. Nature 2001;410:701-5. [PubMed]
- Jackson KA, Majka SM, Wang H, et al. Regeneration of ischemic cardiac muscle and vascular endothelium by adult stem cells. J Clin Invest 2001;107:1395-402. [PubMed]
- Franco D, Moreno N, Ruiz-Lozano P. Non-resident stem cell populations in regenerative cardiac medicine. Cell Mol Life Sci 2007;64:683-91. [PubMed]
- Nombela-Arrieta C, Ritz J, Silberstein LE. The elusive nature and function of mesenchymal stem cells. Nat Rev Mol Cell Biol 2011;12:126-31. [PubMed]
- Zuk PA, Zhu M, Mizuno H, et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng 2001;7:211-28. [PubMed]
- Mathiasen AB, Haack-Sorensen M, Kastrup J. Mesenchymal stromal cells for cardiovascular repair: current status and future challenges. Future Cardiol 2009;5:605-17. [PubMed]
- Donndorf P, Strauer BE, Haverich A, et al. Stem cell therapy for the treatment of acute myocardial infarction and chronic ischemic heart disease. Curr Pharm Biotechnol 2013;14:12-9. [PubMed]
- Teng M, Zhao X, Huang Y. Regenerating cardiac cells: insights from the bench and the clinic. Cell Tissue Res 2012;350:189-97. [PubMed]
- Krane M, Wernet O, Wu SM. Promises and pitfalls in cell replacement therapy for heart failure. Drug Discov Today Dis Mech 2010;7:e109-e115. [PubMed]
- Mathiasen AB, Jorgensen E, Qayyum AA, et al. Rationale and design of the first randomized, double-blind, placebo-controlled trial of intramyocardial injection of autologous bone-marrow derived mesenchymal stromal cells in chronic ischemic heart failure (MSC-HF Trial). Am Heart J 2012;164:285-91. [PubMed]
- Puliafico SB, Penn MS, Silver KH. Stem cell therapy for heart disease. J Gen Intern Med 2013;28:1353-63. [PubMed]
- Templin C, Luscher TF, Landmesser U. Cell-based cardiovascular repair and regeneration in acute myocardial infarction and chronic ischemic cardiomyopathy-current status and future developments. Int J Dev Biol 2011;55:407-17. [PubMed]
- Donndorf P, Kaminski A, Tiedemann G, et al. Validating intramyocardial bone marrow stem cell therapy in combination with coronary artery bypass grafting, the PERFECT Phase III randomized multicenter trial: study protocol for a randomized controlled trial. Trials 2012;13:99. [PubMed]
- Fraser JK, Schreiber R, Strem B, et al. Plasticity of human adipose stem cells toward endothelial cells and cardiomyocytes. Nat Clin Pract Cardiovasc Med 2006;3:S33-7. [PubMed]
- Valina C, Pinkernell K, Song YH, et al. Intracoronary administration of autologous adipose tissue-derived stem cells improves left ventricular function, perfusion, and remodelling after acute myocardial infarction. Eur Heart J 2007;28:2667-77. [PubMed]
- Beohar N, Rapp J, Pandya S, et al. Rebuilding the damaged heart: the potential of cytokines and growth factors in the treatment of ischemic heart disease. J Am Coll Cardiol 2010;56:1287-97. [PubMed]
- Opie LH. Erythropoietin as a cardioprotective agent: down but not out. Heart 2011;97:1537-9. [PubMed]
- Lipsic E, Schoemaker RG, van der Meer P, et al. Protective effects of erythropoietin in cardiac ischemia: from bench to bedside. J Am Coll Cardiol 2006;48:2161-7. [PubMed]
- Westenbrink BD, Lipsic E, van der Meer P, et al. Erythropoietin improves cardiac function through endothelial progenitor cell and vascular endothelial growth factor mediated neovascularization. Eur Heart J 2007;28:2018-27. [PubMed]
- Moon C, Krawczyk M, Ahn D, et al. Erythropoietin reduces myocardial infarction and left ventricular functional decline after coronary artery ligation in rats. Proc Natl Acad Sci U S A 2003;100:11612-7. [PubMed]
- Gao D, Ning N, Niu X, et al. Erythropoietin treatment in patients with acute myocardial infarction: a meta-analysis of randomized controlled trials. Am Heart J 2012;164:715-727.e1.
- Janssens SP. Granulocyte colony-stimulating factor in cardiac repair: time for a reappraisal? Eur J Heart Fail 2010;12:1021-3. [PubMed]
- Ohtsuka M, Takano H, Zou Y, et al. Cytokine therapy prevents left ventricular remodeling and dysfunction after myocardial infarction through neovascularization. FASEB J 2004;18:851-3. [PubMed]
- Ohki Y, Heissig B, Sato Y, et al. Granulocyte colony-stimulating factor promotes neovascularization by releasing vascular endothelial growth factor from neutrophils. FASEB J 2005;19:2005-7. [PubMed]
- Ince H, Valgimigli M, Petzsch M, et al. Cardiovascular events and re-stenosis following administration of G-CSF in acute myocardial infarction: systematic review and meta-analysis. Heart 2008;94:610-6. [PubMed]
- Abdel-Latif A, Bolli R, Zuba-Surma EK, et al. Granulocyte colony-stimulating factor therapy for cardiac repair after acute myocardial infarction: a systematic review and meta-analysis of randomized controlled trials. Am Heart J 2008;156:216-226.e9.
- Theiss HD, Gross L, Vallaster M, et al. Antidiabetic gliptins in combination with G-CSF enhances myocardial function and survival after acute myocardial infarction. Int J Cardiol 2013;168:3359-69. [PubMed]
- Drucker DJ, Nauck MA. The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet 2006;368:1696-705. [PubMed]
- Herman GA, Stevens C, Van Dyck K, et al. Pharmacokinetics and pharmacodynamics of sitagliptin, an inhibitor of dipeptidyl peptidase IV, in healthy subjects: results from two randomized, double-blind, placebo-controlled studies with single oral doses. Clin Pharmacol Ther 2005;78:675-88. [PubMed]
- Gnecchi M, Zhang Z, Ni A, et al. Paracrine mechanisms in adult stem cell signaling and therapy. Circ Res 2008;103:1204-19. [PubMed]
- Siu CW, Tse HF. Cardiac regeneration: messages from CADUCEUS. Lancet 2012;379:870-1. [PubMed]
- Smith RR, Barile L, Cho HC, et al. Regenerative potential of cardiosphere-derived cells expanded from percutaneous endomyocardial biopsy specimens. Circulation 2007;115:896-908. [PubMed]
- Johnston PV, Sasano T, Mills K, et al. Engraftment, differentiation, and functional benefits of autologous cardiosphere-derived cells in porcine ischemic cardiomyopathy. Circulation 2009;120:1075-83, 7 p following 1083.
- Li TS, Cheng K, Malliaras K, et al. Direct comparison of different stem cell types and subpopulations reveals superior paracrine potency and myocardial repair efficacy with cardiosphere-derived cells. J Am Coll Cardiol 2012;59:942-53. [PubMed]
- Beltrami AP, Barlucchi L, Torella D, et al. Adult cardiac stem cells are multipotent and support myocardial regeneration. Cell 2003;114:763-76. [PubMed]
- Anversa P, Kajstura J, Rota M, et al. Regenerating new heart with stem cells. J Clin Invest 2013;123:62-70. [PubMed]
- Bearzi C, Rota M, Hosoda T, et al. Human cardiac stem cells. Proc Natl Acad Sci U S A 2007;104:14068-73. [PubMed]
- Tang XL, Rokosh G, Sanganalmath SK, et al. Intracoronary administration of cardiac progenitor cells alleviates left ventricular dysfunction in rats with a 30-day-old infarction. Circulation 2010;121:293-305. [PubMed]
- Chimenti I, Smith RR, Li TS, et al. Relative roles of direct regeneration versus paracrine effects of human cardiosphere-derived cells transplanted into infarcted mice. Circ Res 2010;106:971-80. [PubMed]
- Vunjak-Novakovic G, Lui KO, Tandon N, et al. Bioengineering heart muscle: a paradigm for regenerative medicine. Annu Rev Biomed Eng 2011;13:245-67. [PubMed]
- Robey TE, Saiget MK, Reinecke H, et al. Systems approaches to preventing transplanted cell death in cardiac repair. J Mol Cell Cardiol 2008;45:567-81. [PubMed]
- Malliaras K, Marban E. Cardiac cell therapy: where we’ve been, where we are, and where we should be headed. Br Med Bull 2011;98:161-85. [PubMed]
- Bernstein HS, Srivastava D. Stem cell therapy for cardiac disease. Pediatr Res 2012;71:491-9. [PubMed]
- Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006;126:663-76. [PubMed]
- Gurdon JB. The developmental capacity of nuclei taken from intestinal epithelium cells of feeding tadpoles. J Embryol Exp Morphol 1962;10:622-40. [PubMed]
- Takahashi K, Tanabe K, Ohnuki M, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 2007;131:861-72. [PubMed]
- Ilic D, Polak J. Stem cell based therapy--where are we going? Lancet 2012;379:877-8. [PubMed]
- Burridge PW, Keller G, Gold JD, et al. Production of de novo cardiomyocytes: human pluripotent stem cell differentiation and direct reprogramming. Cell Stem Cell 2012;10:16-28. [PubMed]
- Kattman SJ, Huber TL, Keller GM. Multipotent flk-1+ cardiovascular progenitor cells give rise to the cardiomyocyte, endothelial, and vascular smooth muscle lineages. Dev Cell 2006;11:723-32. [PubMed]
- Moretti A, Caron L, Nakano A, et al. Multipotent embryonic isl1+ progenitor cells lead to cardiac, smooth muscle, and endothelial cell diversification. Cell 2006;127:1151-65. [PubMed]
- Wu SM, Fujiwara Y, Cibulsky SM, et al. Developmental origin of a bipotential myocardial and smooth muscle cell precursor in the mammalian heart. Cell 2006;127:1137-50. [PubMed]
- Islas JF, Liu Y, Weng KC, et al. Transcription factors ETS2 and MESP1 transdifferentiate human dermal fibroblasts into cardiac progenitors. Proc Natl Acad Sci U S A 2012;109:13016-21. [PubMed]
- Min JY, Yang Y, Sullivan MF, et al. Long-term improvement of cardiac function in rats after infarction by transplantation of embryonic stem cells. J Thorac Cardiovasc Surg 2003;125:361-9. [PubMed]
- Singla DK, Hacker TA, Ma L, et al. Transplantation of embryonic stem cells into the infarcted mouse heart: formation of multiple cell types. J Mol Cell Cardiol 2006;40:195-200. [PubMed]
- Nelson TJ, Martinez-Fernandez A, Yamada S, et al. Repair of acute myocardial infarction by human stemness factors induced pluripotent stem cells. Circulation 2009;120:408-16. [PubMed]
- Singla DK, Long X, Glass C, et al. Induced pluripotent stem (iPS) cells repair and regenerate infarcted myocardium. Mol Pharm 2011;8:1573-81. [PubMed]
- Li X, Zhang F, Song G, et al. Intramyocardial injection of pig pluripotent stem cells improves left ventricular function and perfusion: a study in a porcine model of acute myocardial infarction. PLoS One 2013;8:e66688. [PubMed]
- Shiba Y, Hauch KD, Laflamme MA. Cardiac applications for human pluripotent stem cells. Curr Pharm Des 2009;15:2791-806. [PubMed]
- Caspi O, Huber I, Kehat I, et al. Transplantation of human embryonic stem cell-derived cardiomyocytes improves myocardial performance in infarcted rat hearts. J Am Coll Cardiol 2007;50:1884-93. [PubMed]
- Laflamme MA, Chen KY, Naumova AV, et al. Cardiomyocytes derived from human embryonic stem cells in pro-survival factors enhance function of infarcted rat hearts. Nat Biotechnol 2007;25:1015-24. [PubMed]
- van Laake LW, van Donselaar EG, Monshouwer-Kloots J, et al. Extracellular matrix formation after transplantation of human embryonic stem cell-derived cardiomyocytes. Cell Mol Life Sci 2010;67:277-90. [PubMed]
- Wu SM, Hochedlinger K. Harnessing the potential of induced pluripotent stem cells for regenerative medicine. Nat Cell Biol 2011;13:497-505. [PubMed]
- Gore A, Li Z, Fung HL, et al. Somatic coding mutations in human induced pluripotent stem cells. Nature 2011;471:63-7. [PubMed]
- Kim D, Kim CH, Moon JI, et al. Generation of human induced pluripotent stem cells by direct delivery of reprogramming proteins. Cell Stem Cell 2009;4:472-6. [PubMed]
- Anokye-Danso F, Trivedi CM, Juhr D, et al. Highly efficient miRNA-mediated reprogramming of mouse and human somatic cells to pluripotency. Cell Stem Cell 2011;8:376-88. [PubMed]
- Okita K, Matsumura Y, Sato Y, et al. A more efficient method to generate integration-free human iPS cells. Nat Methods 2011;8:409-12. [PubMed]
- Lister R, Pelizzola M, Kida YS, et al. Hotspots of aberrant epigenomic reprogramming in human induced pluripotent stem cells. Nature 2011;471:68-73. [PubMed]
- Ieda M, Fu JD, Delgado-Olguin P, et al. Direct reprogramming of fibroblasts into functional cardiomyocytes by defined factors. Cell 2010;142:375-86. [PubMed]
- Qian L, Huang Y, Spencer CI, et al. In vivo reprogramming of murine cardiac fibroblasts into induced cardiomyocytes. Nature 2012;485:593-8. [PubMed]
- Song K, Nam YJ, Luo X, et al. Heart repair by reprogramming non-myocytes with cardiac transcription factors. Nature 2012;485:599-604. [PubMed]
- Chen JX, Krane M, Deutsch MA, et al. Inefficient reprogramming of fibroblasts into cardiomyocytes using Gata4, Mef2c, and Tbx5. Circ Res 2012;111:50-5. [PubMed]
- Jayawardena TM, Egemnazarov B, Finch EA, et al. MicroRNA-mediated in vitro and in vivo direct reprogramming of cardiac fibroblasts to cardiomyocytes. Circ Res 2012;110:1465-73. [PubMed]
- Nam YJ, Song K, Luo X, et al. Reprogramming of human fibroblasts toward a cardiac fate. Proc Natl Acad Sci U S A 2013;110:5588-93. [PubMed]
- Zhang D, Shadrin IY, Lam J, et al. Tissue-engineered cardiac patch for advanced functional maturation of human ESC-derived cardiomyocytes. Biomaterials 2013;34:5813-20. [PubMed]
- Tulloch NL, Murry CE. Trends in cardiovascular engineering: organizing the human heart. Trends Cardiovasc Med 2013;23:282-6. [PubMed]
- Zimmermann WH, Schneiderbanger K, Schubert P, et al. Tissue engineering of a differentiated cardiac muscle construct. Circ Res 2002;90:223-30. [PubMed]
- Radisic M, Park H, Shing H, et al. Functional assembly of engineered myocardium by electrical stimulation of cardiac myocytes cultured on scaffolds. Proc Natl Acad Sci U S A 2004;101:18129-34. [PubMed]
- Zimmermann WH, Melnychenko I, Wasmeier G, et al. Engineered heart tissue grafts improve systolic and diastolic function in infarcted rat hearts. Nat Med 2006;12:452-8. [PubMed]
- Eschenhagen T, Eder A, Vollert I, et al. Physiological aspects of cardiac tissue engineering. Am J Physiol Heart Circ Physiol 2012;303:H133-43. [PubMed]
- Porrello ER, Mahmoud AI, Simpson E, et al. Transient regenerative potential of the neonatal mouse heart. Science 2011;331:1078-80. [PubMed]
- Bergmann O, Bhardwaj RD, Bernard S, et al. Evidence for cardiomyocyte renewal in humans. Science 2009;324:98-102. [PubMed]
- Senyo SE, Steinhauser ML, Pizzimenti CL, et al. Mammalian heart renewal by pre-existing cardiomyocytes. Nature 2013;493:433-6. [PubMed]
- Engel FB, Schebesta M, Duong MT, et al. p38 MAP kinase inhibition enables proliferation of adult mammalian cardiomyocytes. Genes Dev 2005;19:1175-87. [PubMed]
- Bersell K, Arab S, Haring B, et al. Neuregulin1/ErbB4 signaling induces cardiomyocyte proliferation and repair of heart injury. Cell 2009;138:257-70. [PubMed]
- Kühn B, del Monte F, Hajjar RJ, et al. Periostin induces proliferation of differentiated cardiomyocytes and promotes cardiac repair. Nat Med 2007;13:962-9. [PubMed]
- Xin M, Olson EN, Bassel-Duby R. Mending broken hearts: cardiac development as a basis for adult heart regeneration and repair. Nat Rev Mol Cell Biol 2013;14:529-41. [PubMed]
- Eulalio A, Mano M, Dal Ferro M, et al. Functional screening identifies miRNAs inducing cardiac regeneration. Nature 2012;492:376-81. [PubMed]
- Laube F, Heister M, Scholz C, et al. Re-programming of newt cardiomyocytes is induced by tissue regeneration. J Cell Sci 2006;119:4719-29. [PubMed]
- Jopling C, Sleep E, Raya M, et al. Zebrafish heart regeneration occurs by cardiomyocyte dedifferentiation and proliferation. Nature 2010;464:606-9. [PubMed]
- Kikuchi K, Holdway JE, Werdich AA, et al. Primary contribution to zebrafish heart regeneration by gata4(+) cardiomyocytes. Nature 2010;464:601-5. [PubMed]
- Oh H, Bradfute SB, Gallardo TD, et al. Cardiac progenitor cells from adult myocardium: homing, differentiation, and fusion after infarction. Proc Natl Acad Sci U S A 2003;100:12313-8. [PubMed]
- Laugwitz KL, Moretti A, Lam J, et al. Postnatal isl1+ cardioblasts enter fully differentiated cardiomyocyte lineages. Nature 2005;433:647-53. [PubMed]
- Martin CM, Meeson AP, Robertson SM, et al. Persistent expression of the ATP-binding cassette transporter, Abcg2, identifies cardiac SP cells in the developing and adult heart. Dev Biol 2004;265:262-75. [PubMed]
- Sucov HM, Gu Y, Thomas S, et al. Epicardial control of myocardial proliferation and morphogenesis. Pediatr Cardiol 2009;30:617-25. [PubMed]
- Zhou B, Honor LB, He H, et al. Adult mouse epicardium modulates myocardial injury by secreting paracrine factors. J Clin Invest 2011;121:1894-904. [PubMed]
- Smart N, Risebro CA, Melville AA, et al. Thymosin beta4 induces adult epicardial progenitor mobilization and neovascularization. Nature 2007;445:177-82. [PubMed]
- Smart N, Bollini S, Dube KN, et al. De novo cardiomyocytes from within the activated adult heart after injury. Nature 2011;474:640-4. [PubMed]
- Zhou B, Honor LB, Ma Q, et al. Thymosin beta 4 treatment after myocardial infarction does not reprogram epicardial cells into cardiomyocytes. J Mol Cell Cardiol 2012;52:43-7. [PubMed]


