Reclassification of early stage pulmonary adenocarcinoma and its consequences
Introduction
Lung cancer is still the leading cause of death in the United States compared with other types of malignancies. Over the years, a change in the frequency of the two most common histological subtypes—adenocarcinoma and squamous cell carcinoma—has seen adenocarcinoma emerge as the most common form of lung cancer (1). The relatively poor prognosis for these tumors reflected in a 5-year survival rate estimated at 15.6% makes early detection and the development of new treatment strategies even more crucial (2). In this context it is not only becoming increasingly important to recognize tumors early in their course but also to separate tumors that are likely to behave more indolently from those with more aggressive behavior.
The term “bronchioloalveolar carcinoma (BAC)” is commonly used to describe a pulmonary adenocarcinoma that is well differentiated and is believed to be associated with a better outcome. A flaw with this term has always been the discrepancy in its use not just among pathologists but also among clinicians resulting in confusion about what this entity really means. In order to clarify this issue, a new classification of lung adenocarcinoma has recently been proposed (3). In this publication, one of the main recommendations was to discontinue the use of the term “BAC” and replace it with the new categories of “adenocarcinoma in situ (AIS)” for tumors with a pure bronchioloalveolar growth pattern and 100% survival and “minimally invasive adenocarcinoma (MIA)” for tumors with a predominant bronchioloalveolar growth pattern, small foci of invasion measuring ≤5 mm or less and near 100% survival (3). Although this change was no doubt well intended, these recommendations were primarily based on a selected review of the literature and focused on tumors categorized as BAC thereby failing to provide a justification for the new nomenclature based on an actual case series that would conform to the definitions outlined. In addition, there is no comparison—either by clinical parameters or statistical analysis—to other types of lung adenocarcinomas that are not classified as BAC yet present at a similar clinical stage. This leads to the question of what the clinical implications of such a new proposal would be. The consequence of calling a tumor “in situ” or “minimally invasive” may create a false sense of security for patients and their clinicians and may lead to suboptimal treatment. On the other hand, one has to wonder about the necessity of creating these new categories if one argues that the overall survival of patients with early stage adenocarcinoma is statistically not significantly different regardless of histological growth pattern. The same applies to the treatment of these tumors, usually complete surgical resection and staging. If there is no significant treatment difference—irrespective of histological pattern—then what is the clinical value of such histological separation? Unfortunately, these issues have not been addressed in the proposed new classification system.
BAC—historical context and current definition
BAC has long been recognized in the literature. The first descriptions of this type of tumor date back to the end of the 19th and early 20th centuries when Malassez (4) and Musser (5) referred to pulmonary tumors with features of BAC as “epithelioma” and “primary cancer of the lung”, respectively. Histologically, these tumors were characterized by a prominent bronchioloalveolar pattern but differed in their extension into the surrounding tissues. Following these initial descriptions, countless publications emerged in the literature describing similar features, albeit under a variety of different names such as “alveolar cell tumor of lung”, “pulmonary adenomatosis” or “mucocellular papillary adenocarcinoma of the lung” (6-14). Despite these different names and an uncertainty of how many of these tumors were single nodules, multifocal lesions or diffuse tumors, it appears that stromal invasion was consistently absent although some of the cases had documented metastatic disease. In those early descriptions the main contention was the cell of origin and whether the tumor originated from the alveolar lining cells or the cells lining the terminal airways. In 1960, Liebow (15) used the term BAC to define a well differentiated adenocarcinoma and unified the different growth patterns described for this tumor: (I) the single nodular pattern; (II) the disseminated nodular pattern; (III) the diffuse pattern. Of note, as per Liebow’s definition, these tumors were also capable of invading lymph nodes, pleura and extrathoracic organs but were characterized by long dormancy and slow growth, particularly those presenting as a single nodule. This original definition by Liebow subsequently underwent different interpretation and in the latest WHO publications, BAC is defined as a tumor that shows growth of neoplastic cells along pre-existing alveolar structures without evidence of stromal, pleural or vascular invasion de facto implying that BAC is a form of AIS even though this was not explicitly stated (16,17). More recently, a new classification of lung adenocarcinoma was proposed in order to provide more uniform terminology and diagnostic criteria (3). One of the main objectives of this proposal was the discontinuation of the use of the term BAC and its replacement with new terminology: the term “AIS” was introduced to describe a localized small (≤3 cm) adenocarcinoma with growth restricted to neoplastic cells along preexisting alveolar structures, lacking stromal, vascular, or pleural invasion and the term “MIA” was proposed for a small, solitary adenocarcinoma (≤3 cm), with a predominantly bronchioloalveolar pattern and ≤5 mm invasion in greatest dimension in any one focus. These new concepts were developed with the aim to identify tumors that according to the authors have 100% (AIS) or near 100% (MIA) survival if completely resected. Of note, this new classification has provoked several questions. Firstly, it introduces size criteria—tumors have to be smaller than 3 cm in order to qualify for the new categories and may not show more than 5 mm of invasion in cases of MIA. Setting such arbitrary cut-off points goes very much against the natural history of tumors that usually undergo continuous development from pre-invasive lesions to more extensive disease. Such criteria would also imply that a tumor otherwise compliant with the proposed criteria but a tumor size exceeding 3 cm falls short of a diagnosis of AIS and would have to be categorized elsewhere. The fact that stromal, lymphatic or pleural invasion has to be excluded or limited in order to qualify for the new categories also implies that such a diagnosis can only ever be made on resected material precluding definitive diagnosis on biopsy or cytologic preparations. From a clinical point of view criticism has been raised as to the fact that terms such as “carcinoma in situ” or “minimally invasive carcinoma” have a potential for misguidance and may simulate less indolent behavior than should be expected.
Pathological features
Traditionally, three different macroscopic types of BAC have been described:
- The localized form. This type of tumor is usually present in the periphery of the lung and relatively well circumscribed but unencapsulated. Areas of necrosis or hemorrhage are absent.
- The multinodular pattern. Here, the tumor involves the lung parenchyma in a miliary pattern with individual nodules usually less than 1 cm in size and with involvement of one or more lobes.
- The diffuse form. The appearance is that of a pneumonic process. No discrete masses or nodules are identified and a non-neoplastic process is often initially suspected. One or more lobes may be involved. This type is invariably associated with the mucinous type of BAC.
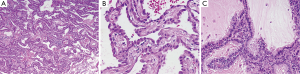
The morphological features of these tumors are very similar irrespective of the solitary, disseminated or diffuse growth pattern. Histologically, the tumor is characterized by growth along pre-existing alveolar walls which are partially or completely replaced by a low cuboidal or cylindrical epithelium (Figure 1A). The cytological features are bland and characterized by oval or round nuclei with variably conspicuous nucleoli (Figure 1B). Nuclear inclusions are not uncommon in the non-mucinous type of the tumor. Mucinous variants show more columnar cells with mucin production and extensive extracellular mucin in the alveolar lumina (Figure 1C). Even though mitotic activity is not prominent, rare mitotic figures can be seen in some cases. By definition the tumor are not allowed to show any stromal, pleural, lymphatic, or nodal invasion.
Discussion
Since the first descriptions of early well differentiated adenocarcinomas classified as BAC there has been active debate in the literature about this entity (6-15,18-24). The main contention with this tumor has always been its unclear definition and resultant subjective interpretation not only in pathology but also in clinical practice. A myriad of different opinions with regard to pathogenesis, diagnosis and prognosis have contributed to the controversy on this tumor (6-15,18-24). This starts with the question of whether this tumor should be regarded as a distinct clinicopathological entity or whether BAC represents a specific growth pattern. For instance, in a study of 30 cases, Bennett and Sasser (23) concluded that there is no histogenetical, morphological or clinical basis to separate BAC from the conventional type of adenocarcinoma. In a similar manner, Rosenblatt et al. (22) put forward their opinion, that extrathoracic tumors can metastasize to the lung in a manner indistinguishable from BAC supporting the opinion that BAC is a pattern rather than a specific entity. On the other hand, Delarue et al. (24) insisted on the validity of the entity and outlined some criteria for its diagnosis: (I) absence of primary adenocarcinoma elsewhere; (II) absence of intrinsic tumor of bronchogenic origin; (III) peripheral location involving alveolar ducts; and (IV) unaffected interstitium. Interestingly, the authors stated that metastatic disease and malignant pleural effusion may occur in these cases.
Other authors have focused on the prognostic aspects of this tumor correlating the different types of BAC with patient survival. For instance, Manning et al. (25) studied 34 cases of BAC and separated the tumors into mucinous (multicentric) and non-mucinous (solitary) types and observed 5-year survival rates of 25% and 72%, respectively. In a study by Clayton (26) investigating 45 cases of BAC the author found that the 5-year survival dropped significantly for non-mucinous tumors that had aerogenous spread compared to those without stating that the prognosis depended not only on the size of the tumor but also on the presence or absence of alveolar spread.
Overall, it has to be said that although the literature is full of studies on this subject, the data derived from those is difficult to interpret. This is mainly due to the fact that different authors have included different pathological material including cytology preparations, small biopsies and resection specimens. In addition, many studies have not focused on a specific type but have variably included solitary tumors along with those showing a multinodular or diffuse growth pattern. Conflicting information about BAC has also been provided after the latest definition of this tumor by the WHO (16,17). For instance, in 2001 Breathnach et al. (27) found that the 5-year survival for stage I BAC was 83% compared to 67% for conventional adenocarcinoma of the same stage. However, in a study led by Ebright (28) it was finally concluded that the most important parameters in the evaluation of BAC are the clinical pattern and pathologic staging of these tumors rather than the degree of invasion on histological assessment. In 2006, Travis et al. (29) presented a review of BAC in which they defended the relevance of the existing WHO criteria by stating that there was existing evidence that patients with solitary small peripheral BAC have 100% survival at 5 years. This also served as the basis of the most recent proposal for the reclassification of adenocarcinoma. The study that most supported this claim was that by Noguchi et al. (30) investigating a series of 236 cases of small adenocarcinomas of the lung that measured up to 2 cm in greatest diameter. In this study, the authors separated different types of adenocarcinomas into categories ranging from A to F, with 28 of the ones designated as types A and B representing the localized form of BAC; however, on closer examination of the manuscript, the authors account for a total of 34 of these tumors. Interestingly, some of these tumors showed not only lymphatic but also pleural invasion, thus disqualifying these tumors as BAC according to the 1999/2004 WHO schemata. More importantly, by describing small tumors (≤2 cm) with a BAC growth pattern, the authors indirectly acknowledged that these tumors have the potential to show lymphatic or pleural invasion. Another study that was cited to support the current WHO criteria is the one by Zell et al. (31) who identified 1909 patients from the California Tumor Registry with a histological diagnosis of BAC and staging information. They found that stage I BAC had 1-year survival rate of 94% and 5-year survival rate of 65% while for BAC stage II these rates were 89% and 45%, respectively. It is obvious that these rates are nowhere near the 100% survival rate predicted by Noguchi et al. (30).
When the new classification of adenocarcinoma was proposed and the terms AIS and MIA were introduced in 2011, the basis of this classification was formed by a review of 312 selected references chosen from among 11,368 citations (3). Out of these, the core literature on which the proposed change in nomenclature was based consisted of about a dozen publications (32-41). However, on critical review of those publications the attentive reader will encounter a few inconsistencies. Firstly, a number of these publications included tumors that did not adhere to the current size criteria. For instance, several studies included lesions that were either smaller than 0.5 cm and therefore represented atypical adenomatous hyperplasia (AAH) or larger than 3 cm, which is the current cut off proposed for AIS and MIA (32,33,36-38,40). In a similar manner, there were several studies where the tumors clearly demonstrated vascular, pleural or lymph node invasion thereby failing the criteria for inclusion into the AIS and MIA categories (34,35). In addition, not all the studies cited actually stated whether the tumors were submitted and evaluated in their entirety, which is another important criterion for a diagnosis of AIS and MIA (39-41). Interestingly, one of the studies cited states that BAC are often small tumors (<1 cm) and are associated with a better prognosis because of size and not histology (39).
It becomes clear that meaningful and unequivocal conclusions cannot be drawn from these publications and that reclassification of BAC based on a literature review rather than critically comparing all adenocarcinoma in terms of survival is not only misleading but can actually be dangerous. In addition, current surgical practice recommends surgical resection for all early stage adenocarcinomas irrespective of histologic grade therefore the main emphasis should be on tumor staging at the time of diagnosis. Staging is the single most important factor that determines the need for additional medical treatment leaving histological subtyping of adenocarcinomas secondary and not particularly critical in decisions regarding additional treatment.
In order to shed more light on this subject, a recent study of 104 cases of early stage pulmonary adenocarcinoma (pT1N0M0) from a single institution attempted to determine whether there was a statistically significant difference between tumors with pure BAC growth pattern and conventional adenocarcinomas (42). In this study some important observations were made: (I) associated malignancies had a negative impact on patient survival; (II) in patients without associated malignancies, no statistical differences in survival were found when tumors were graded into well, moderately and poorly differentiated tumors; (III) no statistical difference in survival was found when tumors were separated based on their BAC component (<25%; 25-50%; >50%) (Figure 2A), and once again, no statistically significant difference was noted when these were compared to conventional adenocarcinomas; (IV) when tumors were divided into pure BAC versus other types, again, the results failed to show any statistically significant survival difference (Figure 2B); there was merely a trend towards better outcome in tumors with a BAC component (Figure 2C).

The results of this study challenge the validity of the new classification of adenocarcinomas. If staging—not histology—remains the most important prognostic factor then the question arises of what it the purpose of a reclassification based on histological criteria? Another interesting fact is that in that same study none of the cases represented the newly proposed MIA casting further doubt on the validity of such a concept. In addition, the findings also suggest that what is currently labeled “atypical adenomatous hyperplasia” and which represents a lesion with bronchioloalveolar growth pattern but with a size of ≤0.5 cm in greatest dimension probably reflects the true AIS.
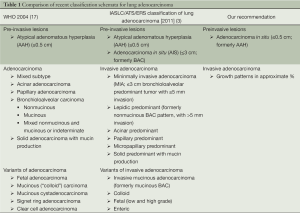
In summary, the current literature appears to challenge the new classification of adenocarcinoma with regard to the reclassification of BAC. The suggested change in terminology implying this to be a pre-invasive lesion may mislead clinicians and prevent full staging of the patients. Careful staging is all the more important as several studies have shown that small adenocarcinomas have documented metastatic disease to lymph nodes in as many as 22% of patients (43,44). Therefore treatment for patients with small adenocarcinomas should not be based on the histological features of the tumor but rather on the stage of the disease at the time of diagnosis. Nonetheless, the diagnosis of adenocarcinoma should include not only the grade of the tumor and the pattern of growth but also the presence or absence of tumor spread. Based on this we suggest the following terminology for adenocarcinomas (also see Table 1):
Full table
For biopsy specimens:
- Adenocarcinoma with BAC growth pattern (tumors ≥0.5 cm)
For surgical resections:
- AIS (≤0.5 cm; formerly AAH)
- Adenocarcinoma [growth pattern(s) in approximate %]
- Size of the tumor
- Pleural integrity
- Lymph node status
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Wahbah M, Boroumand N, Castro C, et al. Changing trends in the distribution of the histologic types of lung cancer: a review of 4,439 cases. Ann Diagn Pathol 2007;11:89-96. [PubMed]
- Surveillance, epidemiology and end results program (SEER): (Available online: http://www.cdc.gov/cancer/lung/statistics/index.html and http://www.seer.cancer.gov/csr/ 1975_2008)
- Travis WD, Brambilla E, Noguchi M, et al. International association for the study of lung cancer/american thoracic society/european respiratory society international multidisciplinary classification of lung adenocarcinoma. J Thorac Oncol 2011;6:244-85. [PubMed]
- Malassez L. Examen histologique d’un cas de cancer encephaloide du poumon (epithelioma). Arch Physiol Norm Pathol 1876;3:353-72.
- Musser JH. Primary cancer of the lung. U Penn Med Bull 1903;16:289-96.
- Sweany HC. A so-called alveolar cell cancer of the lung. Arch Pathol 1935;19:203-7.
- Casilli AR, White HJ. Rare forms of primary malignant lung tumors. Am J Clin Pathol 1940;10:623-41.
- Geever KT. Alveolar cell tumor of the human lung. Arch Pathol 1942;33:551-69.
- Herbut PA. Alveolar cell tumor of the lung; further evidence of its bronchiolar origin. Arch Pathol (Chic) 1946;41:175-84. [PubMed]
- Wood DA, Pierson PH. Pulmonary alveolar adenomatosis in man. Is this the same disease as jaagsiekte in sheep? Am Rev Tuberc 1945;51:205-24.
- Ikeda K. Alveolar cell carcinoma of the lung. Am J Clin Pathol 1945;15:50-63.
- Osserman KE, Neuhof H. Mucocellular papillary adenocarcinoma of the lung, lobectomy, five-year follow-up. J Thorac Surg 1950;19:875-6. [PubMed]
- Drymalski GW, Thompson JR, Sweany HC. Pulmonary adenomatosis; a report of three cases. Am J Pathol 1948;24:1083-93. [PubMed]
- Delarue NC, Graham EA. Alveolar cell carcinoma of the lung (pulmonary adenomatosis, Jagziekte?) a multicentric tumor of epithelial origin. J Thorac Surg 1949;18:237-51. [PubMed]
- Liebow AA. Bronchiolo-alveolar carcinoma. Adv Intern Med 1960;10:329-58. [PubMed]
- Travis WD, Colby TV, Corrin B, et al. eds. WHO Histological Classification of Tumours: Histological typing of lung and pleural tumours. 3rd edition. Berlin: Springer Verlag, 1999.
- Travis WD, Brambilla E, Müller-Hermelink HK, et al. eds. Pathology and Genetics of Tumours of the Lung, Pleura, Thymus and Heart. Lyon: IARC, 2004.
- Overholt RH, Meissner WA, Delmonico JE Jr. Favorable bronchiolar carcinoma. Dis Chest 1955;27:403-13. [PubMed]
- Watson WL, Farpour A. Terminal bronchiolar of "alveolar cell" cancer of the lung. Two hundred sixty-five cases. Cancer 1966;19:776-80. [PubMed]
- Belgrad R, Good CA, Woolner LB. Alveolar-cell carcinoma (terminal bronchiolar carcinoma). A study of surgically excised tumors with special emphasis on localized lesions. Radiology 1962;79:789-98. [PubMed]
- Munnell ER, Lawson RC, Keller DF. Solitary bronchiolar (alveolar cell) carcinoma of the lung. J Thorac Cardiovasc Surg 1966;52:261-70. [PubMed]
- Rosenblatt MB, Lisa JR, Collier F. Primary and metastatic bronciolo-alveolar carcinoma. Dis Chest 1967;52:147-52. [PubMed]
- Bennett DE, Sasser WF. Bronchiolar carcinoma: a valid clinicopathologic entity? A study of 30 cases. Cancer 1969;24:876-87. [PubMed]
- Delarue NC, Anderson W, Sanders D, et al. Bronchioloalveolar carcinoma: a reappraisal after 24 years. Cancer 1972;29:90-7. [PubMed]
- Manning JT, Spjut HJ, Tsechen JA. Bronchioloalveolar carcinoma: the significance of two histopathologic types. Cancer 1984;54:525-34. [PubMed]
- Clayton F. Bronchioloalveolar carcinomas. Cell types, patterns of growth, and prognostic correlates. Cancer 1986;57:1555-64. [PubMed]
- Breathnach OS, Kwiatkowski DJ, Finkelstein DM, et al. Bronchioloalveolar carcinoma of the lung: recurrences and survival in patients with stage I disease. J Thorac Cardiovasc Surg 2001;121:42-7. [PubMed]
- Ebright MI, Zakoski MF, Martin J, et al. Clinical pattern and pathologic stage but not histologic features predict outcome for bronchioloalveolar carcinoma. Ann Thorac Surg 2002;74:1640-6. [PubMed]
- Travis WD, Garg K, Franklin WA, et al. Bronchioloalveolar carcinoma and lung adenocarcinoma: the clinical importance and research relevance of the 2004 World Health Organization pathologic criteria. J Thorac Oncol 2006;1:S13-9. [PubMed]
- Noguchi M, Morikawa A, Kawasaki M, et al. Small adenocarcinoma of the lung: histologic characteristics and prognosis. Cancer 1995;75:2844-52. [PubMed]
- Zell JA, Ignatius Ou SH, Ziogas A, et al. Validation of the proposed International Association for the Study of Lung Cancer non-small cell lung cancer staging system revisions for advanced bronchioloalveolar carcinoma using data from the California Cancer Registry. J Thorac Oncol 2007;2:1078-85. [PubMed]
- Yamato Y, Tsuchida M, Watanabe T, et al. Early resection of a prospective study of limited resection for bronchioloalveolar adenocarcinoma of the lung. Ann Thorac Surg 2001;71:971-4. [PubMed]
- Watanabe S, Watanabe T, Arai K, et al. Results of wedge resection for focal bronchioloalveolar carcinoma showing pure ground-glass attenuation on computed tomography. Ann Thorac Surg 2002;73:1071-5. [PubMed]
- Suzuki K, Asamura H, Kusumoto M, et al. “Early” peripheral lung cancer: prognostic significance of ground glass opacity on thin-section computed tomography scan. Ann Thorac Surg 2002;74:1635-9. [PubMed]
- Sakurai H, Dobashi Y, Mizutani E, et al. Bronchioloalveolar carcinoma of the lung 3 centimeters or less in diameter: a retrospective assessment. Ann Thorac Surg 2004;78:1728-33. [PubMed]
- Yamada S, Kohno T. Video-assisted thoracic surgery for pure ground-glass opacities 2 cm or less in diameter. Ann Thorac Surg 2004;77:1911-15. [PubMed]
- Yoshida J, Nagai K, Yokose T, et al. Limited resection trial for pulmonary ground-glass opacity nodules: fifty-case experience. J Thorac Cardiovasc Surg 2005;129:991-6. [PubMed]
- Koike T, Togashi K, Shirato T, et al. Limited resection for non-invasive bronchioloalveolar carcinoma diagnosed by intraoperative pathologic examination. Ann Thorac Surg 2009;88:1106-11. [PubMed]
- Vazquez M, Carter D, Brambilla E, et al. Solitary and multiple resected adenocarcinomas after CT screening for lung cancer: Histopathologic features and their prognostic implications. Lung Cancer 2009;64:148-54. [PubMed]
- Yim J, Zhu LC, Chiriboga L, et al. Histological features are important prognostic indicators in early stages lung adenocarcinoma. Mod Pathol 2007;20:233-41. [PubMed]
- Borczuk AC, Qian F, Kazeros A, et al. Invasive size is an independent predictor of survival in pulmonary adenocarcinoma. Am J Surg Pathol 2009;33:462-9. [PubMed]
- Weissferdt A, Kalhor N, Marom E, et al. Early pulmonary adenocarcinoma (T1N0M0): a clinical, radiological, surgical, and pathological correlation of 104 cases. The MD Anderson Cancer Center Experience. Mod Pathol 2013;26:1065-75. [PubMed]
- Takizawa T, Terashima M, Koike T, et al. Lymph node metastasis in small peripheral adenocarcinoma of the lung. J Thorac Cardiovasc Surg 1998;116:276-80. [PubMed]
- Konaka C, Ikeda N, Hiyoshi T, et al. Peripheral non-small lung cancers 2.0 cm or less in diameter: Proposed criteria for limited pulmonary resection based upon clinicopathological presentation. Lung Cancer 1998;21:185-91. [PubMed]


