Primary malignant pericardial mesothelioma—a rare cause of superior vena cava thrombosis and constrictive pericarditis
Introduction
Primary malignant pericardial mesothelioma (PMPM) morbidity is rare, with high lethality. The treatment options of the tumor are limited, and the prognosis is poor.
Case report
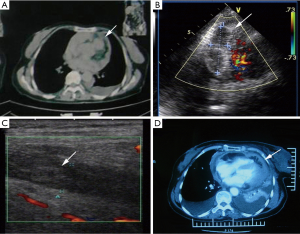
A 60-year-old woman who was admitted to our hospital in April 2009 complained of repeatedly dry cough and shortness of breath. Transthoracic echocardiography demonstrated profuse pleural effusion and pericardial effusion. The symptoms were markedly relieved by 2 times of pericardiocentesis and 3 times of thoracentesis. Cytological analysis of the clear fluid showed no malignant cells, except for a small increase in eosinophilic cells, and PCR for tuberculosis was negative. The woman had no apparent history of occupational or incidental exposure to asbestos or history of smoking or tuberculosis. The patient was then examined by an F-18 fluorodeoxyglucose positron emission tomography/computed tomography (FDG- PET/CT) scan in another hospital, showing a recurrent large pericardial effusion and pleural effusion with a thickened pericardium (Figure 1A). In July 2009, the patient was re-admitted to our hospital, and transthoracic echocardiography on July 22 demonstrated copious pericardial effusion with a thickened pericardium (Figure 1B). The patient was misdiagnosed for tuberculosis clinically, but an experimental anti-TB treatment was ineffective. On August 14, transthoracic echocardiography and inferior vena cava ultrasound showed thrombosis of the proximal end of the right internal jugular vein and left subclavian vein (Figure 1C). On August 15, a chest CT scan demonstrated that she suffered from pericardial tumors (suspected malignant), bilateral pleural effusion, lung inflammation, and breast swelling (Figure 1D).
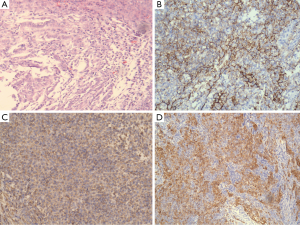
Digital subtraction angiography (DSA) was performed to insert a thrombosis filter in the superior vena cava but failed. On the same day, the patient underwent a partial pericardiectomy, revealing a tumor 11 cm × 9 cm × 2 cm in size. However, a complete resection could not be performed due to tumor adherence to the right ventricular, right atrium and superior vena cava, and part of the inferior vena cava. Histological examination revealed diffuse infiltration of the pericardium by epithelial cells (Figure 2A). An evaluation of immunohistochemical markers showed a strongly positive reaction to epithelial membrane antigen (EMA) (Figure 2B), vimentin (Figure 2C) and cytokeratin (CK) (Figure 2D).
Some of the patients’ symptoms, i.e., dyspnea and chest congestion, had been relieved after surgical resection of the mesothelioma because the compression of the tumor was reduced to a certain extent. Subsequently, two cycles of chemotherapy with cisplatin and etoposide (dosage according to recently published trials) were administered during four months (1-4), but little remission was achieved. The patient remained symptomatic and unfortunately died in December 2009 because of multiorgan dysfunction syndrome.
Discussion
Malignant mesothelioma is a malignant tumor derived from mesenchymal tissue, usually occurring in the peritoneum or pleura and rarely occurring in the pericardium. The incidence of PMPM was found to be below 0.0022% in a large autopsy study (1). Malignant pericardial mesothelioma is characterized by unrepresentative solid growth of the mesothelium, with a structure of atypical cavities environed by fibrous stroma (2).
Although PMPM is infrequent, it is the third most common tumor around the heart/pericardium, after angiosarcoma (33%) and rhabdomyosarcoma (20%) (3). A review of 29 cases presented in the recent literature indicates a higher incidence of malignant pericardial mesothelioma among men than women, with a male-female ratio of 3:1. PMPM occurs more commonly in the elderly, and the median age is 46 (range, 19-76). No obvious relationship between asbestos exposure and the development of pericardial mesothelioma has been established (4).
PMPM is often misdiagnosed after a patient’s first clinical signs because of its asymptomatic symptoms in the early course. The symptoms of PMPM usually range from dyspnea, cough and dysphagia to chest pain (5) and even superior vena cava thrombosis, as in this case. The clinical misdiagnosis rate is extremely high because the symptoms of PMPM are atypical and often confused with conditions such as tuberculous pericarditis, coronary heart disease, atrial myxoma, heart failure, and cardiomyopathy. In this case, the patient was tentatively treated with an antituberculotic due to misdiagnosis by low-grade fever and thickened pericardium.
Single or partial pericardial mesothelioma is rare, and a precise diagnosis is made prior to the patient’s death in only 10-20% of all cases (6). Indeed, the challenging diagnosis of pericardial diseases often requires a combination of multimodal imaging methods including echocardiography, CT, magnetic resonance imaging and FDG-PET scans (7). Cytologic examination of pericardial fluid is seldom conclusive (malignant cells demonstrated in 4/17 cases) (4). Although clinical data and imaging are very helpful for the diagnosis of pericardial mesothelioma, a definite diagnosis still relies on pericardial biopsy or pathology (8). Immunohistochemical studies could provide a diagnosis of mesothelioma and rule out the diagnosis of other tumors, and we used the same diagnosing methods as previously used (4,6,9). The patient was finally diagnosed with PMPM because there was no evidence of primary cancer origin in other organs.
At present, the optimal therapeutic tool is surgical resection in the early clinical course combined with radiotherapy and chemotherapy (9). Partial resection of the tumor in this case merely resulted in a short period of partial symptom reduction. The newer medical treatments, i.e., genetic testing therapies, cytokines therapies, and neoadjuvant chemotherapy, are popular therapies for most mesotheliomas (8-11). In malignant pleural mesothelioma (PMP), researchers found that low EMX2 expression was a negative prognostic factor for mesothelioma, which may serve as an important prognostic and predictive molecular biomarker of a progression-free survival (10). Komiya et al. indicated that CD26-expressing MPM cells upregulate the production of periostin, which enhances the migration and invasion of MPM cells (11). However, these approaches have not yet been applied for pericardial mesothelioma, perhaps due to the low incidence of PMPM and the relatively small number of cases.
The prognosis depends on tumor stage, pathology and genetics. Some 60% of patients die within 6 months after diagnosis due to complications that include heart failure or pericardial constriction, sudden invasion of the tumor into coronary thrombosis and myocardial infarction (12). Prognosis of this disease is dismal, and the median survival of patients with pericardial mesothelioma is approximately 6 months (1,4,12).
Conclusions
Primary pericardial mesotheliomas are extremely rare tumors. PMPM should be considered and appropriately managed in repeated pericardial effusion or pericardial constriction, non-responders to pericardiocentesis and even superior vena cava thrombosis of unknown etiology. Our case report shows that treatment options are limited and that the prognosis is poor.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- De Rosa AF, Cecchin GV, Kujaruk MR, et al. Malignant mesothelioma of the pericardium. Medicina (B Aires) 1994;54:49-52. [PubMed]
- Butz T, Faber L, Langer C, et al. Primary malignant pericardial mesothelioma - a rare cause of pericardial effusion and consecutive constrictive pericarditis: a case report. J Med Case Rep 2009;3:9256. [PubMed]
- Karadzic R, Kostic-Banovic L, Antovic A, et al. Primary pericardial mesothelioma presenting as constrictive pericarditis. Arch Oncol 2005;13:150-2.
- Nilsson A, Rasmuson T. Primary Pericardial Mesothelioma: Report of a Patient and Literature Review. Case Rep Oncol 2009;2:125-32. [PubMed]
- Reardon KA, Reardon MA, Moskaluk CA, et al. Primary pericardial malignant mesothelioma and response to radiation therapy. Rare Tumors 2010;2:e51. [PubMed]
- Papi M, Genestreti G, Tassinari D, et al. Malignant pericardial mesothelioma. Report of two cases, review of the literature and differential diagnosis. Tumori 2005;91:276-9. [PubMed]
- Langer C, Butz T, Horstkotte D. Multimodality in imaging calcific constrictive pericarditis. Heart 2006;92:1289. [PubMed]
- Sardar MR, Kuntz C, Patel T, et al. Primary pericardial mesothelioma unique case and literature review. Tex Heart Inst J 2012;39:261-4. [PubMed]
- Maruyama R, Sakai M, Nakamura T, et al. Triplet chemotherapy for malignant pericardial mesothelioma: a case report. Jpn J Clin Oncol 2006;36:245-8. [PubMed]
- Giroux Leprieur E, Hirata T, Mo M, et al. The homeobox gene EMX2 is a prognostic and predictive marker in malignant pleural mesothelioma. Lung Cancer 2014;85:465-71. [PubMed]
- Komiya E, Ohnuma K, Yamazaki H, et al. CD26-mediated regulation of periostin expression contributes to migration and invasion of malignant pleural mesothelioma cells. Biochem Biophys Res Commun 2014;447:609-15. [PubMed]
- Luk A, Ahn E, Vaideeswar P, et al. Pericardial tumors. Semin Diagn Pathol 2008;25:47-53. [PubMed]


