Prognostic analysis of the bronchoplastic and broncho-arterioplastic lobectomy of non-small cell lung cancers—10-year experiences of 161 patients
Introduction
For central-type non-small cell lung cancers (NSCLCs), bronchoplasty are occasionally necessary to avoid pneumonectomy and spare lung functions. Technically, there are three types of bronchoplastic procedures: sleeve lobectomy, wedge bronchoplasty and flap bronchoplasty (1). Those different techniques can be utilized according to the scope of resection and reconstruction, which is based on the site and extent of tumor. Wedge bronchoplasty and flap bronchoplasty refers to condition in which the radial continuity of the bronchus is partly preserved and subsequent reconstruction does not involve the entire circumference. For sleeve reconstruction, it means that the conduit is completely transected and followed by end-to-end anastomosis. Moreover, double sleeve lobectomy is defined when both the bronchus and the pulmonary artery (PA) undergo sleeve resections and reconstructions (2).
The techniques of bronchoplastic and broncho-arterioplastic surgery have been gradually improved in recent decade. And 5-year survival rates have been estimated and found to range from 24% to 70% across different centers (3-9). However, few studies focused on bronchoplasty, and the prognostic risk factors of bronchoplasty and broncho-arterioplasty had not been adequately addressed. Therefore, present retrospective study aims to evaluate our single institutional database and summarize such prognostic factors of NSCLC based on the 10-year experiences.
Materials and methods
The Human Ethics Review Committee of the hospital approved the study.
Patients
From January 1, 2004 to December 31, 2013, 16,361 patients underwent lung resections for lung cancer, and 170 patients were subjected to lobectomy with bronchial and/or PA reconstruction with a systematic lymphadenectomy in Shanghai Pulmonary Hospital. After excluding four cases with pneumonectomy carried out as final decision and five cases with metastatic (M1) disease, a total of 161 patients were enrolled in the present study.
Preoperative evaluations
All patients were diagnosed with NSCLC by bronchoscopy. For preoperative staging, all patients underwent a contrast-enhanced chest computed tomography (CT). N2 disease with bulky mediastinal lymphadenopathy (lymph nodes larger than 25 mm in short axis) was contraindicated for surgery (10). In cases with contra-lateral mediastinal lymphadenopathy, endobronchial ultrasound (EBUS) biopsy has been utilized to assess mediastinal nodal status since July 2010; prior to that, mediastinoscopy was adopted to exclude N3 cases. Patients with confirmed N2 disease were recommended for neo-adjuvant chemotherapy with a cisplatin-based regimen and then underwent re-evaluations for surgical treatment. Remote metastases were also excluded using head magnetic resonance (MR), abdominal MR or sonography, bone scintigraphy or positron emission tomography-computed tomography (PET/CT) scans before operations. Physical examination, laboratory testing, electrocardiograms, and lung function tests were performed in all patients. Patients with forced expiratory volume in 1 second (FEV1) <70% were considered as decreased lung function.
Surgical technique
Operations were performed under general anesthesia with double-lumen endotracheal intubation. Thoracic cavity was accessed with a standard posterolateral thoracotomy through the 4th/5th intercostal space or in later cases, using video-assisted thoracoscopies. Upon hilum exposure, position and range of tumor involvement were first assessed for resectability and range of resection. Occasionally, when longitudinal tumor involvement had reached either segmental artery branch of the innocent lung or pericardial entrance of the PA trunk, over-length end-to-end artery anastomosis was considered risky and the operation was converted to pneumonectomy.
Anatomical dissection and en-bloc removal of the target lobe were performed first. Proximal control of the main PA is usually obtained extrapericardially and distal control of the PA is usually obtained in the pulmonary fissure. A tangential transection was preferred when there was only tumor extension onto the root of a PA branch. For tumor that involved more than 1/4 perimeter of PA wall, circumferential (sleeve) PA resection was performed with subsequent end-to-end reconstruction. The anastomosis was performed with 5-0 prolene running suture. Generally, local administration of 1:500 heparin solution was utilized to flush the blocked vessel lumen; thus, no systemic heparin administration was applied.
The bronchial stumps were initially trimmed according to the preoperative bronchoscopic findings, and the resected margins were sent immediately to intraoperative frozen sections to confirm R0 resection. Broncho-plastic procedures were performed when tumor growth was within the lobar orifice. The retained inner lateral bronchial wall was tailored into transverse “V” shape to diminish possible tensions. Subsequent end-to-end anastomosis was performed with 4-0 absorbable sutures in an interrupted flavor. When there was wider bronchial defect, complete transection and sleeve reconstruction were performed with 4-0 sutures. A sheet of adjacent pedunculated pleura or intercostal muscle flaps was interposed between the two anastomoses (bronchial and artery). Two chest tubes were then placed in routine flavor.
Postoperative management
The patients were encouraged to cough and expectorate to promote pulmonary inflation. Prophylactic intravenous antibiotics were administered during the first three postoperative days. Low-molecular heparin was administered only when the patient was at high risk of pulmonary embolism (11). Chest tubes were removed when there was no air-leak upon complete lung re-inflation and when pleural effusion was <200/day. Postoperative morbidity and mortality were defined as any complication or death during hospitalization or within 30 days after the surgery.
Adjuvant therapy and follow-up
Upon complete resections, all patients were recommended to undergo four cycles of platinum-based adjuvant chemotherapies. Radiotherapies were administered when there were >2 stations of N2 metastasis (12). Also, this decision in some cases was made by the referring medical oncologist.
The patients were asked to schedule the first re-visit at 4 weeks after operations. It was scheduled every 34 months in first 2 years and every 6–12 months thereafter. Chest CT scans were recommended every 6–12 months postoperatively. The patients were followed up by either mail, phone call, or outpatient clinic visits. In the present study, the median follow-up time was 31 (range, 8–121) months. The overall survival status was known for 143 (88.8%) patients and disease-free survival (DFS) status was known for 132 (82.0%) patients, respectively. Before final statistical analysis, all cases were re-staged according to the latest version of TNM staging system for lung cancer (13).
Statistical analysis
Quantitative variables were expressed as mean ± standard error (SD). Chi-square test was for categorical variables, and the student’s t-test was for continuous variables. Survival rates were calculated using life-table analysis. Survival curves were plotted using the Kaplan-Meier formula, with the date of surgery as the starting point. The log-rank test was performed for univariate analysis. Variables that achieved significance at P<0.05 were entered into the multivariable analyses via the Cox regression model. SPSS 20.0 data analysis software (IBM Inc., CA, USA) was used for statistical analysis. Statistical significance was considered at P<0.05.
Results
General information
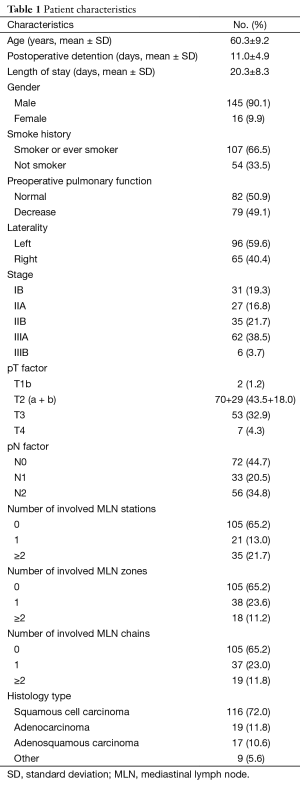
Overall, 161 patients were enrolled in the present study (Table 1), 145 males (90.1%) and 16 females (9.9%), with an average age of 60.3±9.2 (range, 19–80) years. Furthermore, 107 (66.5%) patients were smokers or ever smokers. Additionally, 62 (38.5%) patients had comorbidities: hypertension [37], coronary artery disease [3], and arrhythmia [5] while 18 patients had either PA hypertension, emphysema or history of pulmonary tuberculosis. Mean preoperative FEV1 was 74.5%±16.1%; 79 patients (49.1%) had a FEV1 less than 70%. Twenty-two (13.7%) patients received induction chemotherapy. Ninety-six (59.6%) procedures were performed on the left side and 65 (40.4%) on the right. Lastly, 104 (64.6%) patients had adjuvant chemotherapy, while 18 (11.2%) patients received sequential adjuvant radiotherapy.
Full table
Operations
Overall, 28 broncho-arterioplastic and 133 bronchoplastic procedures were conducted. There were 97 sleeve and 64 wedge bronchoplasties, respectively; specifically, PA sleeve resections and reconstructions were performed in 26 patients while the remained 135 patients received partial resection. Complete double sleeve reconstructions were performed in 24 cases. Eighty-six patients (52.8%) had left upper lobectomy while nine (5.6%) had left lower lobectomy. Forty-eight (29.8%) patients had right upper lobectomies while one had right middle lobectomy and three had right lower lobectomies. Eight patients had right mid-lower bilobectomies. Eight patients had right upper-middle bilobectomies. R0 resection was achieved in all patients. Sixteen (9.9%) cases had blood loss >1,000 mL because of pleural adhesion or vascular accidents. Operations were completed within 4 hours in 81 cases.
Pathology
There were 116 (72.0%) squamous cell carcinomas, 19 (11.8%) adenocarcinomas, 17 (10.6%) adenosquamous carcinomas, and others (n=9). Overall, 2,475 lymph nodes (15 nodes/patient) were removed, and 275 nodes were positive. Among 1,670 resected mediastinal nodes, 172 (10.3%) were positive and 47 out of 365 hilar lymph nodes were M1. The pathological staging information is shown in Table 1.
Post-operative complications
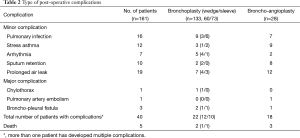
Table 2 shows the incidence of postoperative complications within 90 days. No intraoperative death was reported. The 90-day post-operative mortality rate was 3.1% (5/161). One patient had ischemic stroke on the 2nd post-operative day and died 1 month later. Another patient had acute massive pulmonary embolism and died on the first postoperative day. The rest three patients died of respiratory failure subsequent to pneumonia aggravation 10 to 40 days postoperatively.
Full table
The 90-day post-operative morbidity rate was 24.8% (n=40). The morbidity rate for bronchoplasty was 16.5% (22/133), which is lower than broncho-arterioplasty (64.3%, 18/28). Major complications, such as chylothorax (n=1), broncho-pleural fistula (n=3) and PA embolism (n=1, lead to mortality) both occurred in bronchoplasty and broncho-arterioplasty groups. Three patients developed late bronchopleural fistulas, and two #22 chest tubes were immediately placed. One tube was inserted to the chest apex area for air exhaust, while the other was placed at the lowest position of the cavity for complete liquid discharge. And we performed the lavage of the cavity in order to clean the cavity and to decrease bacteria number to the greatest degree. Lavage was performed twice or 3 times a day until the pleural fluid revealed a normal white blood cell (WBC) count. The patients were educated to practice postural drainage after chest tube drainage. While the cavity was completely drained and tube drainages revealed no effusions, the chest tube was removed. Other complications, including pulmonary infection (n=16), asthma exacerbation (n=12), arrhythmia (n=7), sputum retention with bronchoscopic intervention (n=10), and prolonged air leak (n=19) were successfully managed conservatively.
Site of recurrence
No recurrence was observed along the PA suture line and the bronchial anastomotic site. Twenty-one patients had local recurrence (17 pulmonary and 4 at lymph nodes). Extrathoracic distant metastasis was proved in 35 patients at a median time of 7 months. Systemic chemotherapies were administered after the diagnosis. Adrenal metastectomy was performed in one patient with isolated adrenal metastasis. The median survival of these patients with disease relapse was 17.8 months (95% CI: 11–48 months) after operation.
Survival analysis
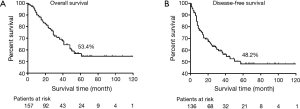
The overall 5-year survival (OS) rate was 53.4%, and 5-year DFS rate was 48.2% (Figure 1A,B). DFS was determined upon the event of local recurrence (n=12), systemic metastases (n=13), N3 lymph nodes involvement (n=4), metastases to bone (n=11), liver (n=2), brain (n=3), and lung metastasis at remaining ipsilateral lobes (n=1), or contralateral lobes (n=4). Upon the end of follow-up date (July 2014), 81 (50.3%) patients maintained DFS and 19 (11.8%) experienced tumor-recurrence.
Survivals were subsequently calculated according to respective T and N classifications. There were only two T1 cases and seven T4 cases; thus, the 5-year survival rates were not available for T1 and T4 categories. The 5-year OS were 54.4% for T2 case and 54.8% for T3 (P=0.447). The 5-year DFS for T2 and T3 cases were 52.6% and 55.9%, respectively (P=0.338) (Figure 2A,B). For different N scales, the 5-year OS were 66.3% (N0), 49.0% (N1), and 39.2% (N2) (P=0.05). Five-year DFS were 60.2% (N0), 43.9% (N1), and 36.8% (N2). Significant difference was observed in 5-year DFS (P=0.012) (Figure 2C,D).
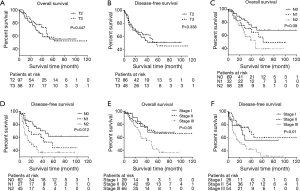
For stage I (n=31) and stage II (n=62) patients, the 5-year OS were 67.7% and 64.4% while 5-year DFS were 55.7% and 58.9%, respectively. The 5-year OS and 5-year DFS were 36.9% and 31.9% for stage III disease, respectively. Statistically significant differences in 5-year survival rates emerged between stage I, II, and III diseases (OS, P=0.027; DFS, P=0.007) (Figure 2E,F).
Univariate analysis revealed that age >65 and higher grade of pN were associated with poor overall survival while right side tumor, non-squamous histology type, higher grade of pT, and pN were associated with worse DFS (Table 3). Multivariate analysis showed that pN is an independent indicator of worse outcome (Table 4).

Full table
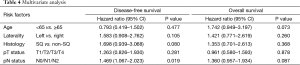
Full table
Considering the pN2 stage was the only prognostic factor and pN2 was a heterogeneous disease course with an extremely wide range of survival, we further made a detailed analysis. To investigate the association between outcomes and lymph node station or lymphatic drainage, we divided the mediastinal lymph node (MLN) according to zones, chains as our group reported previously (14). According to the proposal from the International Association for the Study of Lung Cancer (IASLC) staging project (15), four intra-thoracic mediastinal nodal zones have been classified: upper zone/superior mediastinal nodes (station-2, -3 and -4); AP zone/aortic nodes (station-5 and -6); subcarinal zone (station-7) and lower zone (station-8 and -9). As suggested by Riquet et al. (16), five intra-thoracic mediastinal lymphatic chains were defined: Stations-2 and -4; Stations-3A and -3P; Stations-5 and -6; Station-7; Stations-8 and -9. Upper zone/superior mediastinal nodes (station-2, -3 and -4) took up the largest part in the nodal zones while chain constituted by stations-2 and -4 was the most frequently being invaded. Multiple stations, zones and chains metastasis were found showing a trend toward worse prognosis than single stations, zones and chains involvement, although not statistically significant (Figure 3A-F).

Discussion
In 1947, Sir Clement Price Thomas (17) first reported a successful case underwent a circumferential resection of the right main bronchus for an adenoma. In 1952, he further reported that two ends of a bronchus could be united well in the way as the intestine (18). Few years later, Allison (19) performed a first sleeve resection for a lung cancer patient and Paulson et al. (20) made the first comprehensive report on bronchoplasty. And the mortality, morbidity and 5-year survival rates in patients with bronchoplasty and broncho-arterioplasty for lung cancer have gradually improved during the past decades (Table 5).
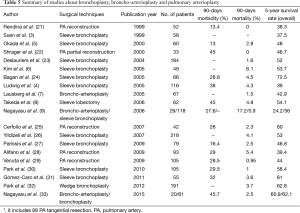
Full table
Past studies focused mostly on comparing clinical significance of bronchoplasties and broncho-arterioplasties with pneumonectomies (2,9,26). A meta-analysis by Ma et al. (34) suggested that bronchial sleeve lobectomy combined with pulmonary angioplasty has more favorable survivals compared to pneumonectomy, resulting in approximately 30% lower death risk. The weighted mean operative mortality was 3.3% and complication rate was 32.4% for the double plastic surgery. The 5-year overall survival was 30.6% for the pneumonectomies and 38.7% for the broncho-arterial reconstructions. Gómez-Caro et al. (31) showed that the mean survival times of sleeve lobectomy (n=55, including 10 bronchoangioplasty cases) versus pneumonectomy (n=21) were 46 and 34 months (P=0.03). In Ludwig’s case series (4), the OS were 39% after sleeve lobectomy (n=116, 30 broncho-angioplasty cases included) and 27% after pneumonectomy (n=194). Therefore, past studies validated the satisfactory outcomes of bronchoplasty and broncho-arterioplasty. However, the prognostic risk factors for this specific patients group had not been well discussed in the previous studies.
From present 161 cases, we proved that pN was an independent prognostic factor of patients with bronchoplastic and broncho-arterioplastic procedures. Meanwhile, tumor position, histology type, pT, and pN were associated with worse DFS. Some related articles proposed similar results on single aspect of either bronchoplastic or angioplastic cases. Venuta et al. (29) had concluded that for 65 double sleeve reconstructions (in a case series of 105 angioplasties), induction therapy, N2 status, and adenocarcinoma were all negative prognostic factors. Overall, 5-year survivals were 52.6% for N0+ N1 and 20% for N2 cases. Kojima et al. (35) showed that the 5-year overall and DFS rates were 76.0% and 62.9% for pN0 cases (n=14), 45.7% and 43.5% for pN1 cases (n=27), and 28.9% and only 20.0% for pN2+ pN3 cases (n=20) (P=0.014 and 0.036), respectively. In addition, for pN2 patients with multiple stations, zones and chains metastasis, they may need closer follow-up and more aggressive post-operative adjuvant therapy, as this group showed a worse prognosis (36).
Interestingly, when comparing outcomes of stage I (T1b/T2aN0M0) and stage II (T2b/T3N0M0, T1b/T2aN1M0) cases, difference in survival rates was non-significant. After excluding N1 cases (in order to eliminate the prognostic impact of lymph node metastasis), the difference between stage I and stage II remained non-significant (P=0.979, data not shown). Since this is a highly selected patient group with locally advanced tumor extension at hilum region, it is reasonable to hypothesize that all these cases (T1b/T2a/T2b/T3N0M0) share the same T status. Considering the 5-year survival at 64.4–67.7%, T3N0M0 (stage II) categorization might be equally acceptable. Supporting evidence comes from other studies. The 5-year survival rates of stage I and stage II disease were 57.3% and 69.5%, respectively, in Yildizeli’s study (26), and 75% and 53%, respectively, in Venuta’s study (29). Similarly, no statistically significant differences emerged between these two stages in 5-year survivals in both studies.
Moreover, several technical issues should be addressed. Apart from PA sleeve reconstruction by end-to-end anastomosis, sleeve reconstruction by a prosthetic conduit could be performed by indication. A prosthetic conduit could be synthetic or biologic material, such as autologous pericardial tissue or pulmonary vein portion. The main pitfall of using conduit interposition is sizing its length. Using autologous pericardial tissue or pulmonary vein may also compromise the integrity of pericardium and vessel. For bovine pericardial tissue, that may be less cost-effective and less biocompatible. We did not use conduit interposition in our institution, however, there were many advantages for conduit interposition and this technique is feasible (21,31). For example, autologous pericardial tissue or pulmonary vein conduits are fresh and unpreserved, cost-free, and biocompatible. Also, using conduits could elongate the restriction of the resection length of PA. Regarding wedge bronchoplasty and sleeve bronchoplasty, they carry different morbidity risk and outcomes. Wedge bronchoplasty has been considered as containing high risk of bronchial stenosis, kinking and angulation (1) and may not show satisfactory outcome with respect to tumor and nodal clearance (37). However, based on our result and recently published data, wedge bronchoplasty is an acceptable procedure compared with sleeve bronchoplasty. Operative death did not develop in this study. The mortality rate for sleeve lobectomy was reported to be 2.7% to 7.5%, similar to the wedge bronchoplasty which is 3.7% (32). For surgical morbidity, wedge bronchoplasty is also a safer procedure than sleeve lobectomy. For long-term survival, the wedge bronchoplasty were comparable with sleeve lobectomy (Table 5). And the local recurrence rate of wedge bronchoplasty was about 8.9% (32), comparable with sleeve lobectomy (5–24%) (6,24). These results imply that wedge bronchoplastic lobectomy can be performed by experienced surgeons without compromising oncologic principles.
As shown in the literature, present study demonstrated again inspiring long-term outcomes of bronchoplasty and broncho-angioplasty in lung cancer surgery. Similarly, with technical improvement, the mortality and morbidity rates of bronchoplasties and broncho-arterioplasties have been acceptably low. The documented 5-year overall survival ranged from 42.9% to 47.0% and 5-year DFS ranged from 40.0% to 43.1% (4,28,38). Nagayasu et al. (9) reported that among broncho-arterioplasty patients with stage I or II, the 5-year survival rate was 51.9%. For patients with stage III or IV, 5-year survival rate was 7.9%. Venuta et al. (29) found that for combined bronchoplasty and broncho-arterioplasty group, survival rates were 75% for stage IB, 53% for stage II, 31% for stage IIIA, and 19% for IIIB. The results in present study were also encouraging, indicating that the OS was 53.4% and 5-year DFS was 48.2%. Moreover, the 90-day post-operative complication rate was 24.8% in the present study, which is obviously comparable to prior results ranging from 23% to 40.4% (3,35,38,39). Considering such low mortality and morbidity, the discussed maneuvers were both safe, effective, and inspiring.
This study had several important limitations. First, this was a retrospective single-institution study. A retrospective study is susceptible to various sources of bias, which may not have been identified and controlled. Second, the median follow-up time is 31 months. The explanation of the follow-up time is that the patients in our institution were increasing in recent years and near-term patients therefore make up the large proportion of our cohort. Admittedly, the follow-up time is not long enough and may cause an overestimation of long-term prognosis. However, the large number of patients in our study could make the prognostic results reliable. And we assessed the postoperative outcomes and compared the prognostic impact of surgical techniques and clinical pathological variables, those results are all convincing as well. Furthermore, there was a high rate (42.2%) of pathologic stage III, and the majority (34.8% in all patients) of stage III is N2 disease. It should be noted that invasive mediastinal staging may not be performed for patients without showing lymph node enlargement in early years. And EBUS and mediastinoscopy could classify correctly almost 90% of cases, but some patients were still found to have unexpected macro or microscopically evidence of mediastinal spread (40-42). That may cause unexpected N2 disease. Also, not every patient could be respond to neoadjuvant therapy and then be downstaged. In addition, bronchoplasty and broncho-arterioplasty is mostly performed for locally advanced tumor extension at hilum region. Therefore, for the cohort itself, it may include many advanced NSCLC patients, so the stage III is reasonably high. In addition, the rate of stage III patients is comparable to other related studies: Park et al. (32) showed 37.2% stage III patients. Nagayasu et al. (33) reported about 30% to 35% stage III patients. Galetta et al. (43) showed that stage IIIA and stage IIIB were 36.2% and 14.9%, respectively.
In conclusion, as a lung parenchyma preserving surgery, bronchoplastic and broncho-arterioplastic lobectomy are both safe and effective for NSCLC patients. Pathological lymph node status (pN) was closely related to long-term survival in this disease category of patients with NSCLC.
Acknowledgements
Funding: This study was supported by the Shanghai Committee of Science and Technology (grants 15411968400 and 14411962600).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Khargi K, Duurkens VA, Versteegh MM, et al. Pulmonary function and postoperative complications after wedge and flap reconstructions of the main bronchus. J Thorac Cardiovasc Surg 1996;112:117-23. [PubMed]
- Tronc F, Gregoire J, Rouleau J, et al. Long-term results of sleeve lobectomy for lung cancer. Eur J Cardiothorac Surg 2000;17:550-6. [PubMed]
- Suen HC, Meyers BF, Guthrie T, et al. Favorable results after sleeve lobectomy or bronchoplasty for bronchial malignancies. Ann Thorac Surg 1999;67:1557-62. [PubMed]
- Ludwig C, Stoelben E, Olschewski M, et al. Comparison of morbidity, 30-day mortality, and long-term survival after pneumonectomy and sleeve lobectomy for non-small cell lung carcinoma. Ann Thorac Surg 2005;79:968-73. [PubMed]
- Okada M, Yamagishi H, Satake S, et al. Survival related to lymph node involvement in lung cancer after sleeve lobectomy compared with pneumonectomy. J Thorac Cardiovasc Surg 2000;119:814-9. [PubMed]
- Kim YT, Kang CH, Sung SW, et al. Local control of disease related to lymph node involvement in non-small cell lung cancer after sleeve lobectomy compared with pneumonectomy. Ann Thorac Surg 2005;79:1153-61; discussion 1153-61. [PubMed]
- Lausberg HF, Graeter TP, Tscholl D, et al. Bronchovascular versus bronchial sleeve resection for central lung tumors. Ann Thorac Surg 2005;79:1147-52; discussion 1147-52. [PubMed]
- Takeda S, Maeda H, Koma M, et al. Comparison of surgical results after pneumonectomy and sleeve lobectomy for non-small cell lung cancer: trends over time and 20-year institutional experience. Eur J Cardiothorac Surg 2006;29:276-80. [PubMed]
- Nagayasu T, Matsumoto K, Tagawa T, et al. Factors affecting survival after bronchoplasty and broncho-angioplasty for lung cancer: single institutional review of 147 patients. Eur J Cardiothorac Surg 2006;29:585-90. [PubMed]
- Silvestri GA, Gonzalez AV, Jantz MA, et al. Methods for staging non-small cell lung cancer: Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 2013;143:e211S-50S.
- Geerts WH, Heit JA, Clagett GP, et al. Prevention of venous thromboembolism. Chest 2001;119:132S-75S. [PubMed]
- Rodrigues G, Choy H, Bradley J, et al. Adjuvant radiation therapy in locally advanced non-small cell lung cancer: Executive summary of an American Society for Radiation Oncology (ASTRO) evidence-based clinical practice guideline. Pract Radiat Oncol 2015;5:149-55. [PubMed]
- Goldstraw P, Crowley J, Chansky K, et al. The IASLC Lung Cancer Staging Project: proposals for the revision of the TNM stage groupings in the forthcoming (seventh) edition of the TNM Classification of malignant tumours. J Thorac Oncol 2007;2:706-14. [PubMed]
- Zheng H, Wang LM, Bao F, et al. Re-appraisal of N2 disease by lymphatic drainage pattern for non-small-cell lung cancers: by terms of nodal stations, zones, chains, and a composite. Lung Cancer 2011;74:497-503. [PubMed]
- Rusch VW, Asamura H, Watanabe H, et al. The IASLC lung cancer staging project: a proposal for a new international lymph node map in the forthcoming seventh edition of the TNM classification for lung cancer. J Thorac Oncol 2009;4:568-77.
- Riquet M, Arame A, Foucault C, et al. Prognostic classifications of lymph node involvement in lung cancer and current International Association for the Study of Lung Cancer descriptive classification in zones. Interact Cardiovasc Thorac Surg 2010;11:260-4. [PubMed]
- Thomas CP. Conservative resection of the bronchial tree. J R Coll Surg Edinb 1956;1:169-86. [PubMed]
- Johnston JB, Jones PH. The treatment of bronchial carcinoma by lobectomy and sleeve resection of the main bronchus. Thorax 1959;14:48-54. [PubMed]
- Allison PR. Course of thoracic surgery in Groningen. (Quoted by Jones PH. Lobectomy and bronchial anastomosis in the surgery of bronchial carcinoma). Available online: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2413903/pdf/annrcse00351-0026.pdf
- Paulson DL, Shaw RR. Preservation of lung tissue by means of bronchoplastic procedures. Am J Surg 1955;89:347-55. [PubMed]
- Rendina EA, Venuta F, De Giacomo T, et al. Sleeve resection and prosthetic reconstruction of the pulmonary artery for lung cancer. Ann Thorac Surg 1999;68:995-1001; discussion 1001-2. [PubMed]
- Shrager JB, Lambright ES, McGrath CM, et al. Lobectomy with tangential pulmonary artery resection without regard to pulmonary function. Ann Thorac Surg 2000;70:234-9. [PubMed]
- Deslauriers J, Gregoire J, Jacques LF, et al. Sleeve lobectomy versus pneumonectomy for lung cancer: a comparative analysis of survival and sites or recurrences. Ann Thorac Surg 2004;77:1152-6; discussion 6. [PubMed]
- Bagan P, Berna P, Pereira JC, et al. Sleeve lobectomy versus pneumonectomy: tumor characteristics and comparative analysis of feasibility and results. Ann Thorac Surg 2005;80:2046-50. [PubMed]
- Cerfolio RJ, Bryant AS. Surgical techniques and results for partial or circumferential sleeve resection of the pulmonary artery for patients with non-small cell lung cancer. Ann Thorac Surg 2007;83:1971-6; discussion 1976-7.
- Yildizeli B, Fadel E, Mussot S, et al. Morbidity, mortality, and long-term survival after sleeve lobectomy for non-small cell lung cancer. Eur J Cardiothorac Surg 2007;31:95-102. [PubMed]
- Parissis H, Leotsinidis M, Hughes A, et al. Comparative analysis and outcomes of sleeve resection versus pneumonectomy. Asian Cardiovasc Thorac Ann 2009;17:175-82. [PubMed]
- Alifano M, Cusumano G, Strano S, et al. Lobectomy with pulmonary artery resection: morbidity, mortality, and long-term survival. J Thorac Cardiovasc Surg 2009;137:1400-5. [PubMed]
- Venuta F, Ciccone AM, Anile M, et al. Reconstruction of the pulmonary artery for lung cancer: long-term results. J Thorac Cardiovasc Surg 2009;138:1185-91. [PubMed]
- Park JS, Yang HC, Kim HK, et al. Sleeve lobectomy as an alternative procedure to pneumonectomy for non-small cell lung cancer. J Thorac Oncol 2010;5:517-20. [PubMed]
- Gómez-Caro A, Garcia S, Reguart N, et al. Determining the appropriate sleeve lobectomy versus pneumonectomy ratio in ce ntral non-small cell lung cancer patients: an audit of an aggressive policy of pneumonectomy avoidance. Eur J Cardiothorac Surg 2011;39:352-9. [PubMed]
- Park SY, Lee HS, Jang HJ, et al. Wedge bronchoplastic lobectomy for non-small cell lung cancer as an alternative to sleeve lobectomy. J Thorac Cardiovasc Surg 2012;143:825-831.e3.
- Nagayasu T, Yamasaki N, Tsuchiya T, et al. The evolution of bronchoplasty and broncho-angioplasty as treatments for lung cancer: evaluation of 30 years of data from a single institution. Eur J Cardiothorac Surg 2016;49:300-6. [PubMed]
- Ma Z, Dong A, Fan J, et al. Does sleeve lobectomy concomitant with or without pulmonary artery reconstruction (double sleeve) have favorable results for non-small cell lung cancer compared with pneumonectomy? A meta-analysis. Eur J Cardiothorac Surg 2007;32:20-8. [PubMed]
- Kojima F, Yamamoto K, Matsuoka K, et al. Factors affecting survival after lobectomy with pulmonary artery resection for primary lung cancer. Eur J Cardiothorac Surg 2011;40:e13-20. [PubMed]
- Casali C, Stefani A, Natali P, et al. Prognostic factors in surgically resected N2 non-small cell lung cancer: the importance of patterns of mediastinal lymph nodes metastases. Eur J Cardiothorac Surg 2005;28:33-8. [PubMed]
- Deslauriers J, Tronc F, Gregoire J. History and current status of bronchoplastic surgery for lung cancer. Gen Thorac Cardiovasc Surg 2009;57:3-9. [PubMed]
- Ghiribelli C, Voltolini L, Luzzi L, et al. Survival after bronchoplastic lobectomy for non small cell lung cancer compared with pneumonectomy according to nodal status. J Cardiovasc Surg (Torino) 2002;43:103-8. [PubMed]
- Lausberg HF, Graeter TP, Wendler O, et al. Bronchial and bronchovascular sleeve resection for treatment of central lung tumors. Ann Thorac Surg 2000;70:367-71; discussion 71-2. [PubMed]
- Varela G, Thomas PA. Surgical management of advanced non-small cell lung cancer. J Thorac Dis 2014;6 Suppl 2:S217-23. [PubMed]
- Lardinois D, De Leyn P, Van Schil P, et al. ESTS guidelines for intraoperative lymph node staging in non-small cell lung cancer. Eur J Cardiothorac Surg 2006;30:787-92. [PubMed]
- De Leyn P, Dooms C, Kuzdzal J, et al. Revised ESTS guidelines for preoperative mediastinal lymph node staging for non-small-cell lung cancer. Eur J Cardiothorac Surg 2014;45:787-98. [PubMed]
- Galetta D, Solli P, Borri A, et al. Bronchovascular reconstruction for lung cancer: does induction chemotherapy influence the outcomes? Ann Thorac Surg 2012;94:907-13; discussion 13. [PubMed]


