|
Brief Technique Reports
Surgical Management Of 3 Cases With Huge Tracheoesophageal Fistula With Esophagus Segment in situ As Replacement Of The Posterior Membranous Wall Of The Trachea
Jianxing He, Manyin Chen, Wenlong Shao, Shuben Li, Weiqiang Yin, Yingying Gu, Daoyuan Wang, Steven Tucker
From Department of Cardiothoracic Surgery (Drs He, Shao, Li, Yin, and Wang), Department of Pathology (Dr Gu), the First A ffiliated Hospital of Guangzhou
Medical College, Guangzhou Research Institute of Respiratory Disease & China State Key Laboratory of Respiratory Disease (Drs He, Chen, Shao, Li, Y in,
and Wang), Centro Hospital Conde De S. Januario (Dr Chen), Pacific Cancer Centre Singapore (Dr Tucker)
Corresponding to: Prof. Dr. Jianxing He, MD, PhD, FACS. Department of Cardiothoracic
Surgery, The First Affiliated Hospital of Guangzhou Medical College; Guangzhou Research
Institute of Respiratory Disease & China State Key Laboratory of Respiratory Disease. No.
151,Yanjiang Rd,Guangzhou 510120,PR China.Tel:+86-20-83337792,Fax:+86-20-83350363, Email: hejx@vip.163.com
Submitted July 11, 2009. Accepted for publication August 15, 2009.
Available at www.jthoracdis.com
|
|
Abstract
Objective:
Tracheoesophageal fistula (TEF) is an uncommon and potentially life-threatening complication of blunt chest trauma. The objectives of this report are to describe our surgical experience in three patients with huge TEF and to evaluate the short-term results of surgical management in this potentially life-threatening complication of blunt chest trauma.
Methods:
Three patients with huge TEF (5.2-7.0 cm in diameter) after blunt chest trauma were kept in supine position, then the neck was
excided and esophagus was free, double breasted suture and clench and mutilation were performed successively over the same level of superior aperture of thorax. The thoracic esophagus was then located in situ at the membrane portion where the trachea was incomplete. Meanwhile, the stomach was then freed through a middle abdominal incision and pulled through the posterior tunnel of the sternum to the neck.
Last, an anastomosis between esophagus and tubular stomach was performed over left neck.
Results:
All three patients recovered well after the operation. They were able to take liquids and then solids beginning ten days after the
procedure. One year post-operation, they were able to resume normal activity.
Conclusion:
The surgical management of patients with huge TEF by esophageal exclusion (cervical gastroesophagostomy) and use of
esophagus segment in situ as replacement of the posterior membranous wall of the trachea is feasible.
Key words
huge tracheoesophageal fistula; Surgical management
J Thorac Dis 2009;1:39-45. DOI: 10.3978/j.issn.2072-1439.2009.12.01.014
|
|
Introduction
Tracheoesophageal fistula (TEF) is a very uncommonand potentially life-threatening complication of blunt chest trauma. The first
reported case of TEF caused by blunt chest injury was published in
1936 by Vinson ( 1). Beal et al. have estimated the incidence of
blunt esophageal perforation in major trauma to be as low as
0.001% (double check, if is true, then delete this sentence since the
morbidity is too low) ( 2). Clinically, coughing and choking after
swallowing, the so-called “swallow-cough complex”or Ono’s
sign, is considered a classic sign for the presence of TEF.
Confirmation and extent of TEF can be accurately diagnosed on esophagography and CT imaging ( 3, 4), while supplemental information
may be acquired with esophagoscopy and bronchoscopy ( 5).
After diagnosis is confirmed, surgical repair must be carried out
as soon as possible. Non-operative mortality is as high as 80%
while operative mortality is estimated at 9.3% ( 6). Closure of the
membranous trachea in one layer and closure of the oesophagus in
two layers, which are membrane and muscle layer, is commonly
undertaken ( 7). Structural interposition between the trachea and oesophagus is advised to decrease the recurrence of TEF and can be
achieved either with a muscle (intercostals, sternothyroid, stern-
ocleidomastoid, strap) or pericardial/pleural flap to separate the lumen of trachea and esophagus ( 6, 8- 12) as well as with fibrin glue
being applied together ( 13). However for patients with a huge
TEF, that is TEF greater than 5 cm, surgical repair is difficult to
perform as the huge defect and necrosis of local tissues after the
blunt chest trauma may be accelerated with concurrent infection
and chemical erosion.
While esophageal excision is useful in patients with simple
esophagus fistulas the reconstruction of the trachea will be more
difficult and the risk of failure in patients with TEF is increased
due to vascular damage to the tracheal tissue. Further, the use of
esophageal excision with anastomosis between tubular stomach
and the esophagus above the fistula, reported by Steiger et al., is
well described as a classical palliative surgery in patients with malignant TEF ( 14). Here we describe the successful treatment of
three patients with huge TEF after blunt chest trauma by
esophageal exclusion (cervical gastroesophagostomy) and use of
esophagus segment in situ as replacement of the posterior membranous wall of the trachea.
The objectives of this report are to describe our surgical experience in three patients with huge TEF and
to evaluate the short-term results of surgical management in this
potentially life-threatening complication of blunt chest trauma.
|
|
Patients and methods
We studied three patients with huge TEF after blunt chest trauma who were successful
treated by esophageal exclusion (cervical gastroesophagostomy) and use of esophagus segment in situ as
replacement of the posterior membranous wall of the trachea.
Case 1.
A 21-year-old male was referred to our department in February
2007 from a hospital in Macao, PR China, due to multiple traumas
sustained in an automobile accident thirteen days prior.
Patient was initially seen for extensive traumatic injuries in the
Emergency Department at a local hospital in Macao. Internal fixation of
bilateral lower extremities fracture as well suture and bandage to numerous
wounds were performed. After his surgical procedures, his extremities were
noted to move freely but he then developed shortness of breath, hypotension,
and vascular insufficiency. He then was intubated and placed on mechanical
ventilation to sustain his vital functions. Radiographic and clinical analy sis
demonstrated bilateral tension pneumothorax, hempthorax andmediastinal emphysema.
After performing bilateral thoracic and mediastinal cavity drainage the circulatory system became stable but his
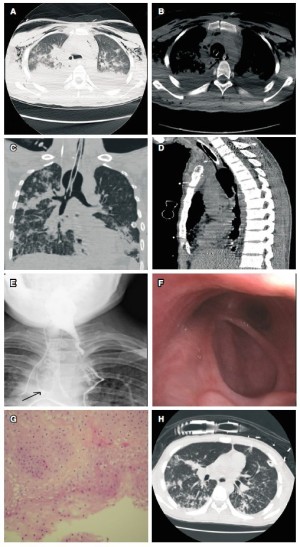
ventilation remained unstable. Further investigation with bronchoscopy and CT imaging showed an approximate 1cm TEF as
well as extensive chemical burn of lungs caused by erosive gastric
reflux ( Fig 1A and 1B). A subsequent jejunostomy and gastrostomy was performed to
provide enteral alimentation as well as decompression of the stomach. However, erosive reflux continued
and the patient developed a secondary pulmonary infection due to
aspiration of gastric contents. Further, as the initial TEF was proximal to the carina the patient
continued to suffer unstable ventilation. Ultimately, a left double-lumen tracheal catheter was inserted
into left bronchus to obtain left-lung unilateral pulmonary ventilation ( Fig 1C).
There after the patient was referred to our facility for
further evaluation and treatment.
Initail evaluation by bronchoscopy and esophagoscopy in our
department demonstrated a huge TEF. A biliary fistula was confirmed with erosion of the trachea and tunica muscularis identified.
The size of fistulae enlarged from 1cm to 7cm over 72 hours. Complete loss of the posterior wall of thoracic segments of pars mem
branacea of trachea and esophagus was noted along with the plane
encompassing the superior aperture of thorax to 1 cm above carina
was absent and the region fused into a huge fistulae. The case was
further complicated at this point with extensive bilateral pneumonia with
urulent sputum noted in the trachea ( Fig 1C).
A left-lung unilateral respirator with left branch of double tubes bronchial
catheter was maintained for ventilation. Now a common cavity between the esophagus and
trachea measuring 7cm in length was detected through thoracic CT scans ( Fig 1D).
After sufficient preoperative preparations, surgical repair was
performed under general anesthesia. Surgical exploration indicated
“frozen mediastinum” due to inflammation and the plane from the
superior aperture of thorax to the carina could not be separated.
Extensive collateral vascularization was noted over the esophagus
and trachea all associated with superior mediastinal inflammation.
We noted the dislocated oesophagus for blood supply over internal
segment posterior wall of thorax. We then took the dislocated
esophagus over internal segment posterior wall of thorax and replaced with the membrane of trachea.
The esophagus was severed from 2 cm below the carina, then using double breasted suture and
clench, the thoracic cavity was closed.
The patient was kept in supine position, then the neck was excided and esophagus was free,
double breasted suture and clench
and mutilation were performed successively over the same level of
superior aperture of thorax (Ethicon, Johnson & Jonhnson, Pennsylvania, USA).
The thoracic esophagus was then located in situ at
the membrane portion where the trachea was incomplete. Meanwhile, the stomach was then freed through a middle abdominal incision and pulled through the posterior tunnel of the sternum to the
neck. Last, an anastomosis between esophagus and tubular stomach was performed over left neck.
Case 2.
A 6-year-old boy was referred to our department from a hospital
in Shanghai, PR China, due to choking after taking food. Six
months prior to referral the child was severely injured in an automobile accident
and during recovery he required a tracheotomy
and gastrotomy for five and a half months. He was initially evaluated and diagnosed
with a traumatic TEF by esophagoscopy and
bronchoscopy in an Emergency Department Anhui province, PR
China. He was then referred to a children’s hospital in Shanghai,
PR China where tracheotomy and gastrostomosis were performed
to stabilize his ventilation and nutrition. After stabilizing and receiving symptomatic treatment his condition im proved. He was
then referred to our department for further evaluation and treatment.
On admission we noted the child to be thin and small, with a
weight of 15 kg. On examination, the tracheostomy cannula was
unobstructed, the inferior extremity of which reached at the level of
carina; on auscultation moist and coarse rales in the lungs were
noted; the gastric stoma duct over abdomen was unobstructed, and
fluid nutritional diets could successfully pass through gastric stoma
duct. After bronchoscopy and three-dimensional reconstruction examination of chest CT scan,
a diagnosis of huge TEF was confirmed with the fused commont cavity between esophagus and trachea
measuring 5.2 cm in length. The child’s case was further
complicated with bilateral lower lobe pneumonia associated with
gastric reflux and aspiration.
Prior to definitive repair a successful jejunostomy was performed to maximize nutrition and
prevent further aspiration. Enteral nutrition was administered via a jejunal fistula.
The gastric fistula was then connected via an external vacuum extractor to reduce
pulmonary inflammation caused by chronic gastric reflux into
lungs. The patient’s condition significantly improved with supportive care and his weight began to increase. After a month of supportive care and with careful preparation, replacement of the membranous trachea with dislocated esophagus in situ and
esophageal-gastric anastomosis over the left neck via a meta-sternal tunnel were performed through the two incisions at neck and
upper central abdomen.
Case 3.
A 46-year-old woman was referred to our department on August
20, 2008, from a hospital in Shenzhen, PR China, because of multiple traumas over the whole body and bucking for a month, caused
by traffic accident. Four months before, patient was admitted to a
hospital in Shenzhen because of multiple traumas caused by traffic
accident. She was found to be with bilateral pulmonary contusion,
fracture of left ribs from the third to seventh, right ribs from second
to eleventh, left collar bone, and transverse process in the right side
of C7. Bilateral thoracic close drainage was performed in the emergent salvage. After the operation, she was sent to ICU and given
respirator to assist ventilation because of hemorrhagic shock.
Further examination CT scan showed right pneumothorax, consolidation of right lower lung and contusion of left lower lung.
Patient’s inflammation and pulmonary contusion were improved after anti-inflammatory therapy and other symptomatic
treatments. Pleural canals in her thoracic cavities were pulled out 2
weeks later. One month later, the respirator and tracheal intubation
were moved away; and patient’s condition of spontaneous ventilation was good. But meanwhile, patient presented with edema of
right lower extremity. Colour Doppler Ultrasound examination indicated right femoral vein thrombosis; and her symptom was
reduced after receiving anticoagulant therapy. Two months later,
there was no abnormity of the pulling out of gastrointestinal decompression tube and lipid diet.
Forty days before, patient was moved to ICU because of aspiration pneumonitis caused by reflux of gastric contents, which was
improved a week later, and control of pulmonary infection was
continued to be performed. Vomiting and bucking reoccurred a
month before when he drank water. Posterior wall of the superior
segment of trachea and unclear boundary with esophagus were found through chest CT scan. He was admitted to our hospital for
further consultation because he was suspected to be with esophago-tracheal fistula.
When she was admitted: little thin and small figure, smooth respiration about 26/min, bondage of moist and coarse rales could be
heard over lungs; abdomen was flat and soft without palpable tumor or tenderness; myodynamia of both upper extremities was
about from grade II to III, and that of both lower extremities was
from grade IV to V. Nutrient canal of duodenum was unobstructed,
and tube feeding with fluid food was normal, but there was lots of
white spumy sputum. Esophago-tracheal fistula was accurately diagnosed through bronchoscopy and chest CT scan reconstruction.
Esophagus and trachea was contacted for about 5.3cm.
After admission, her nutritional condition and clinical symp
toms were improved through overall check up, nutritional support,
pressure sore prevention attendance, anti-inflammatory and anticoagulant therapy and other symptomatic treatments. Then, through
the two incisions at left neck and upper abdomen, replacement to
membranous part of trachea with dislocated esophagus, esophagus
and gaster anastomosis over left neck through the meta-sternum
tunnel was performed on October 9, 2008 under systemic anesthesia. The detail of this case was previously report ( 15).
|
|
Results
The case 1 recovered well after the operation. The tracheal
intubation tube was removed 24 hours after the operation and the
patient could expectorate on his own. He was able to take liquids
and then solids beginning ten days after the procedure. Broad
spectrum antibiotics were given and imaging showed resolution of
pneumonia. Esophagogram and tracheography were performed and
indicated a persistent cavity connecting with trachea over dislocated
oesophagus ( Fig 1E). He was discharged home on post-operative
day twenty. He was seen in follow-up regularly in our department.
The status of ventilation was normal, tracheoscopy indicated trachea
was unobstructed, and residual cavity of the dislocated oesophagus
was well healed ( Fig 1F). Pathology showed only scant
inflammatory cell infiltration over mucous membrane of the
esophagus six months post operation. One year post-operation, his
activity level was normal ( Fig 1G). At fifteen months post operation
his lung function indicated: FVC77.20%,
FEV1 84.58%, FEV1/FVC
92.1; airway resistance guideline total resistance of respiration is
136%,viscosity resistance under 5Hz was 132%, and there was no
evidence of gastric reflux into the esophagus using a 24-hour continuous PH monitor at the gastro-esophageal anastomostic stoma.
For the case 2, tracheal intubation was discontinued two hours
after the operation and the patient could breathe and expectorate on
his own. He was able to take an oral diet by post-operative day 10
and he was discharged home eighteen days later. Bronchoscopy
performed four months after the operation indicated mucous
membrane of esophagus replacing membra naceus tracheae satisfactorily.
The patient was able to resume normal activites including returning to school.
Nine months after the operation, his pulmonary function tests indicated: FVC55.00% , FEV1 60.76% ,
FEV1/FVC 90.52%; airway resistance guideline: total resistance of
respiration was 134%, viscosity resistance under 5Hz was 130%. A
24-hours PH monitor evaluation at the gastro-esophageal anastomostic stoma could not be performed as the young patient was unable to cooperate with the examination.
And for the case 3, trachea cannula was removed an hour later
after the operation, and patient can breathe and expectorate on her
own. Neck incision became red and swollen on the sixth day after
the operation, which was improved after change of dressing and
drainage. On the 20th day after operation, her taking food became
normal and she was discharged 24 days later after operation.
|
|
Discussion
TEF complicates the course of disease in about 20% patients
with esophageal and tracheal cancer ( 16). It is estimated that only
10% of acquired TEF are due to benign disease ( 17). TEF is a rare
complication following blunt chest trauma. In a retrospective study
of 3,606 trauma patients reported by Beal and associates ( 2), 2,560
patients (71%) sustained blunt trauma and only 3 patients (0.001%)
developed esophageal perforations. One could postulate, therefore,
that the incidence of TEF probably constitutes less than 0.001% of
all blunt trauma cases. An extensive review of the literature by
Reed et al ( 6). includes 61 cases since the first reports by Vinson in
1936 ( 1). Accordingly, the commonest mode of injury is vehicular
accidents involving an unrestrained driver and the most common
site of fistula formation is in the posterior trachea at or just above
the carina ( 6).
The majority (80.4%) of TEFs became symptomatic within the
first 10 days of injury with immediate onset in 11.5% of the cases
( 1). Late and very-late onset of TEF after trauma has also been reported as late as 425 days after injury ( 18), 20 years ( 9), or even 38
years ( 19). Immediate onset is often associated with tracheal and
esophageal rupture with subsequent fistula formation. In late onset,
the mechanism may be slightly different with compression of the
trachea and esophagus between the sternum and the vertebral bodies
resulting in partial laceration of the posterior membranous trachea
with contusion and loss of vascularity to the adjacent anterior
esophageal wall. Subsequently, necrosis of the esophageal wall occurs over time resulting in a delayed TEF ( 6).
Several mechanisms also have been proposed to explain TEF
formation ( 20- 22). In Case 1, we noted the fistula enlarged from
1cm to 7cm within three days. We beleive the mechanism of TEF
formation in this case may be compression of the trachea and
esophagus between the sternum and vertebral bodies. We note the
reflex closure of the glottis together with thoracic compression can
produce a rapid increase in intraluminal pressure, which would be
greater in the larger airways. Subsequently, a partial laceration occurs in the mucosa of the posterior membranous trachea, resulting
in reflux bile erosion to the trachea, from the mucosa to submucosa, basement membrane, and muscle of trachea, which can’t be
protected by the mucosa due to the laceration in the mucosa. Concurrently, the anterior esophageal wall is damaged with impairment
of the mucosal blood supply. Esophageal necrosis then occurs
rapidly and followed by TEF formation. It is interesting to find the
trachea and esophagus merge with single huge cavity. ( Fig 1C,D)
The delayed formation of the TEF could account for the majority
(59.0%) of patients who exhibit symptoms 3 to 10 days after blunt
trauma ( 6, 23, 24). However, Stothert and associates propose an al-
ternate mechanism of TEF formation involving immediate tracheal
and esophageal rupture with subsequent fistula formation ( 21).
Coughing and choking after swallowing, the“swallow-cough
complex” or Ono's sign, is considered the classic sign of TEF.
Other signs and symptoms include crepitation over the neck, chest, or
abdominal pain, hemoptysis or hematemesis, dyspnea, dysphagia,
hoarseness, odynophagia, and abdominal distention. The most
commonly associated findings reported include subcutaneous air
(54.1%) and pneumothorax or pneumomediastinum (41.0%). Rib
fractures (31.1%) and hemoptysis (26.2%) are also commonly reported findings ( 6), as well as the
“breathing-bag sign”( 25). Once
the diagnosis of TEF is suspected, mediastinal contamination
should be minimized by placing a large nasogastric tube in the patient's stomach and another in the proximal esophagus to remove
secretions. The presence and extent of the TEF can then be evaluated and diagnosed on esophagography and CT imaging ( 3, 4). Supplemental information may also be acquired with esophagoscopy
and bronchoscopy ( 5). In addition to direct signs of TEF, imaging
can indentify associated problems such as pneumonia, gaseous distention of the esophagus, pneumomediastinum, and subcutaneous
air.
Adequste enteral or parenteral nutrition should be initiated in
perioperation period. Fluid and electrolyte imbalances should be
corrected if necessary, and patients should receive broad-spectrum
antibiotic prophylaxis ( 6). In both our cases, patients suffered serious consequences due to reflux and aspiration through the TEF.
Often a staged operative recovery is necessary. The aim of the
first-stage is to exclude oesophageal contents from the respiratory
tree. Gastrostomy and negative pressure drainage can reduce dangerous gastric content reflowing and aspiration.
After diagnosis of TEF is confirmed, surgical repair should be
carried out as soon as possible. Currenlty, non-operative mortality
is 80% while operative mortality is 9.3% ( 6). Closure of the mem
branous trachea in one layer and closure of the oesophagus in two
layers should be undertaken ( 7). Structural interposition between
the trachea and oesophagus is advised to decrease the recurrence of
TEF. This is achieved either with a muscle (intercostals, sternothyroid,
sternocleidomastoid, strap) or with a pericardial/pleural flap
to separate the lumen of esophagus and trachea ( 6, 8- 12, 38- 43), or
with fibrin glue being applied together ( 13). The size of TEF is 0.5
to 3.0 cm in lengthfor most patients. However, for patients with
so-called huge TEF (> 5 cm in length) such procedures are more
difficult due to necrosis of local tissues associated with infectionafter blunt chest trauma. In these cases, the tension of the muscle or
pericardial/pleural flap will not be enough as the posterior membranous
wall of the trachea to sustain the trachea to preserve the
normal respiration in patients with huge TEF. Esophageal excision
is useful in patients with simple esophagus fistula but the reconstruction of trachea is more difficult, the risk of failure in patients
with TEF is very high, and because the vascular supply of the trachea is often damaged. Esophageal excision with anastomosis between tubular stomach and the esophagus above the fistula, reported by Steiger et al. is described as classical palliative surgery in patients with malignant TEF ( 14). A review of the literature, demonstrates only one case of surgical repair involving esophageal resection with colonic interposition in patients with TEF after the blunt
chest trauma ( 44).
In this study, we define huge TEF as TEF greater than 5 cm in
length In another review by Freitag et al., the fistulas from 1 to 4
cm in length were defined as
“very large fistulas”
( 45). We believe
that operative decisions regarding management of TEF should consider
both the size of fistula as well as the timing of the diagnosis.
Early diagnosis is critical to obtaining ideal results and prompt
treatment as well as structural interposition between the trachea
and oesophagus to decrease the recurrence of TEF. Delayed diagnosis of TEF,
especially when the trachea and esophagus merge into a huge cavity as in our two cases presented, make surgical repair
even more difficult.
We successfully treated three patients with huge TEF after blunt
chest trauma by esophageal exclusion (cervical gastroesophagostomy) and incorporated esophagus segment in situ as replacement of
the posterior membranous wall of the trachea. To our knowledge
this is the first report of this method to treat patients with huge TEF
after blunt chest trauma. The risk of operation was decreased greatly since this direct and simple procedure not only blocked the reflux to trachea, also avoided the further surgical intervention in the
inflammation, edema and necrosis area. Furthermore, the trachea
and the tube stomach were separated after reconstruction since the
tube stomach was located in anterior mediastinum, but trachea in
postmediastinum, this was also avoid the change of TEF recurrence. By using this surgical method to treat the huge TEF, two
questions have to be considered. Firstly, whether the remained
esophageal cavity in situ affect the patient’s pulmonary function
short term and long term. Secondary, whether the secretion from
remained esophageal cavity in situ continually cause the pulmonary infection. In our three cases, self-breathing was achieved at
1, 2 and 24 hours after removing tracheal intubation, respectively.
In case 1, the chest tube was removed 72 hours after the operations. Patients began oral nutrition 9 days, 12 days and 20 days after the operation and were discharged home on day 18, 22 and 24
after operation, respectively. These cases have been followed now
for 2, 18 and 23 months, respectively, and all patients have resumed normal activities of daily living. Pathology indicated mucous membrane of esophagus was with good activity, there was only a little inflammatory cell infiltration. The lung function and airway resistance recovered well during the follow-up on 15 and 9
months later in case 1 and 2, respectively. In case 1, no sign of gastric reflux in esophagus was seen using 24-hours PH monitor
around gastro-esophageal anastomostic stoma. However, these results need to be further studied and confirmed by long term follow-up. An obvious limitation of our report is that there are only
three cases to describe from a single institution and this should be
evaluated in more cases studies from multiple centers.
In summary, huge TEF is rare and potentially fatal c
In summary, huge TEF is rare and potentially fatal complication
of blunt chest trauma. In our three cases, the surgical management
of patients with huge TEF by esophageal exclusion (cervical gas
troesophagostomy) and use of esophagus segment in situ as replacement of the posterior membranous wall of the trachea is feasible. Improving the nutrition status and controlling the lung infection were critical in perioperation period. We emphasize that the
risk of operation and TEF recurrent were decreased greatly since
this direct and simple procedure avoided the further surgical intervention in the inflammation, edema and necrosis area.
|
|
References
- Vinson PP. External trauma as a cause of lesions of the esophagus. Am J Dig Dis 1936;3:456-9.
- Beal SL, Pottmeyer EW, Spisso JM. Esophageal perforation following external blunt trauma. J Trauma 1988;28:1425-32.
[LinkOut]
- Berkmen YM, Auh YH. CT diagnosis of acquired tracheoesophageal fistula in adults. J Comput Assist Tomogr 1985;9:302-4.
[LinkOut]
- Sakamoto Y, Seki Y, Tanaka N, Nakazawa T, Nobori M. Tracheoesophageal fistula after blunt chest trauma: successful diagnosis by computed tomography. Thorac Cardiovasc Surg 2000;48:102-3.
[LinkOut]
- Vasquez RE, Landay M, Kilman WJ, Estrera A, Schreiber T. Benign esophagorespiratory fistulas in adults. Radiology 1988;167:93-6.[LinkOut]
- Reed WJ, Doyle SE, Aprahamian C. Tracheoesophageal fistula after blunt chest trauma. Ann Thorac Surg 1995;59:1251-6.
[LinkOut]
- Banerjee A, Subbarao KSVK, Venkatarman S. Tracheo-oesophageal fistula following blunt trauma. J Laryngol Otol 1984;98:743-4.
[LinkOut]
- McNicholas K, Stark J. Unusual case of benign tracheo-oseophageal fistula. Thorax 1984;39:311-2.
[LinkOut]
- Weber SM, Schurr MJ, Pellett JR. Delayed presentation of a tracheoesophageal fistula after blunt chest trauma. Ann Thorac Surg 1996;62:1850-2.
[LinkOut]
- Layton TR, DiMarco RF, Pellegrini RV. Tracheoesophageal fistula from nonpenetrating trauma. J Trauma 1980;20:802-5.
[LinkOut]
- Stanbridge RD. Tracheo-oesophageal fistula and bilateral recurrent laryngeal nerve palsies after blunt chest trauma. Thorax 1982;37:548-9.
[LinkOut]
- Feliciano DV, Bitondo CG, Mattox KL, Romo T, Burch JM, Beall AC Jr, et al. Combined tracheoesophageal injuries. Am J Surg 1985;150:710-5.
[LinkOut]
- Elliott M, Brady P, Smith R. Tracheo-esophageal fistula following a fall. ANZ J Surg 2001;71:772-3.
[LinkOut]
- Steiger Z, Wilson RF, Leichman L, Franklin R, Dindogru A, Kinzie J. Management of malignant bronchoesophageal fistulas. Surg Gynecol Obstet 1983;157:201-4.[LinkOut]
- He J, Chen M, Shao W, Wang D. Surgical management of huge tracheo-oesophageal fistula with oesophagus segment in situ as replacement of the posterior membranous wall of the trachea. Eur J Cardiothorac Surg 2009;36:600-2.
[LinkOut]
- Burt M, Diehl W, Martini N, Bains MS, Ginsberg RJ, McCormack PM, et al. Malignant esophagorespiratory fistula: management options and survival. Ann Thorac Surg 1991;52:1222.
[LinkOut]
- Gudovsky LM, Koroleva NS, Biryukov YB, Chernousov AF, Perelman MI. Tracheoesophageal fistulas. Ann Thorac Surg 1993;55:868.
[LinkOut]
- Gerzic Z, Rakic S, Randjelovic T. Acquired benign esophagorespiratory fistula:report of 16 consecutive cases. Ann Thorac Surg 1990;50:724-7.
[LinkOut]
- Hoshino K, Satoh N, Kajitani S. A case of tracheoesophageal fistula cured by surgical therapy after blunt trauma received 38 years ago. Kyobu Geka 1999; 52:1052-7.[LinkOut]
- Chapman ND, Braun RA. The management of traumatic tracheoesophageal fistula caused by blunt chest trauma. Arch Surg 1970;100:681-4.
- Stothert JC Jr, Buttorff J, Kaminski DL. Thoracic esophageal and tracheal injury following blunt trauma. J Trauma 1980;20:992-5.
[LinkOut]
- Shorr RM, Crittenden M, Indeck M, Hartunian SL, Rodriguez A. Blunt thoracic trauma. Analysis of 515 patients. Ann Surg 1987;206:200-5.
[LinkOut]
- Karmy-Jones R, Jurkovich GJ. Blunt chest trauma. Curr Probl Surg 2004;41:211-380.
[LinkOut]
- Sebastian MW, Wolfe WG. Traumatic thoracic fistulas. Chest Surg Clin N Am 1997;7:385-400.[LinkOut]
- Rampaul RS, Naraynsingh V, Dean VS. Tracheoesophageal fistula following blunt chest trauma: diagnosis in the ICU-the "breathing bag sign". Chest 1999;116:267.
[LinkOut]
- Jolliet P, Pichard C, Biolo G, Chiolé ro R, Grimble G, Leverve X, et al. Enteral nutrition in intensive care patients: a practical approach. Working Group on Nutrition and Metabolism, ESICM. European Society of Intensive Care Medicine. Intensive Care Med 1998;24:848-59.
[LinkOut]
- Moore FA, Feliciano DV, Andrassy RJ, McArdle AH, Booth FV, Morgenstein-Wagner TB, et al. Early enteral feeding compared with parenteral reduces postoperative septic complications. The results of metaanalysis. Ann Surg 1992;216:172-83.
[LinkOut]
- Kalfarentzos F, Kehagias J, Mead N, Kokkinis K, Gogos CA. Enteral nutrition is superior to parenteral nutrition in severe acute pancreatitis: results of a randomised prospective trial. Br J Surg 1997;84:1665-9.
[LinkOut]
- Demeyer I, Bataillie K, Baute I. Long-term sedation in the ICU: enteral versus parenteral feeding. Clin Intens Care 1994;5(Suppl):13.[LinkOut]
- Illig KA, Ryan CK, Hardy DJ, Rhodes J, Locke W, Sax HC. Total parenteral nutrition-induced changes in gut mucosa function: atrophy alone is not the issue.Surgery 1992;112:631-7.[LinkOut]
- Kotler DP, Levine GM, Shiau YF. Effects of luminal nutrition and metabolic status on in vivo glucose absorption. Am J Physiol 1981;240:432-6.[LinkOut]
- Weser E, Bell D, Tawil T. Effects of octapeptide-cholecystokinin, secretin and glucagon on intestinal mucosal growth in parenteral nourished rats. Digestive Dis Sci 1981;26:409-16.
[LinkOut]
- Pironi L, Paganelli GM, Miglioli M, Biasco G, Santucci R, Ruggeri E, Di Febo G,Barbara L. Morphologic and cytoproliferative patterns of duodenal mucosa in two patients after long-term total parenteral nutrition: changes with oral refeeding and relation to intestinal resection. JPEN J Parenter Enteral Nutr 1994;18:351-4.
[LinkOut]
- Groos S, Hunefeld G, Luciano L. Parenteral versus enteral nutrition: morphological changes in human adult intestinal mucosa. J Submicrosc Cytol Pathol 1996;28:61-74.[LinkOut]
- Shou J, Lappin J, Minnard EA, Daly JM. Total parenteral nutrition, bacterial translocation, and host immune function. Am J Sur 1994;167:145-50.
[LinkOut]
- Ott L, Annis K, Hatton J, McClain M, Young B. Postpyloric enteral feeding costs for patients with severe head injury: blind placement, endoscopy, and PEG/J versus TPN. J Neurotrauma 1999;16:233-42.
[LinkOut]
- Frost P, Bihari D. The route of nutritional support in the critically ill: physiological and economical considerations. Nutrition 1997;13:58S-63S.
[LinkOut]
- Ott L, Annis K, Hatton J, McClain M, Young B. Successful early repair of a traumatic tracheoesophageal fistula after blunt chest trauma. J Thorac Cardiovasc Surg 2006;132:1495-6.
[LinkOut]
- Kanne JP, Stern EJ, Pohlman TH. Trauma cases from Harborview Medical Center. Tracheoesophageal fistula from a gunshot wound to the neck. AJR Am J Roentgenol 2003;180:212.[LinkOut]
- John V, Mathai J, Chacko J, Sen S, Jacob R, Karl S. Tracheoesophageal fistula in a child after blunt chest trauma. J Pediatr Surg 2006;41:E27-29.
[LinkOut]
- Martel G, Al-Sabti H, Mulder DS, Sirois C, Evans DC. Acute tracheoesophageal burst injury after blunt chest trauma: case report and review of the literature. J Trauma 2007;62:236-42.
[LinkOut]
- Devendra K, Mahajan JK, Samujh R, Rao KL. Recurrent tracheoesophageal fistula after foreign body impaction. Pediatr Surg Int 2003;19:214-6.[LinkOut]
- Jacob R, Kunder S, Mathai J, Chacko J, John V. Traumatic tracheoesophageal fistula in a 5-year old. Paediatr Anaesth 2006;16:1068-72.
[LinkOut]
- Conn JH, Hardy JD, Fain WR, Neterville RE. Thoracic trauma: analysis of 1022 cases. J Trauma 1963;3:22-40.
[LinkOut]
- Freitag L, Tekolf E, Steveling H, Donovan TJ, Stamatis G. Management of malignant esophagotracheal fistulas with airway stenting and double stenting. Chest 1996;110:1155-60.
[LinkOut]
Cite this article as: He JX, Chen MY, Shao WL, Li SB, Yin WQ, Gu YY, Wang DY, Tucker S. Surgical Management Of 3 Cases With Huge Tracheoesophageal Fistula With Esophagus Segment in situ As Replacement Of The Posterior Membranous Wall Of The Trachea. J Thorac Dis 2009;1:39-45. doi: 10.3978/j.issn.2072-1439.2009.12.01.014
|