
Multi-level analysis and evaluation of organizational improvements in thoracic surgery according to a Value-Based HealthCare approach
Highlight box
Key findings
• Patient Value in Thoracic Surgery (PVTS) score could allow to evaluate in a multidimensional perspective the treatment path of patients and to objectively monitoring its evolution over time, making a first step towards Value-Based HealthCare (VBHC).
What is known and what is new?
• VBHC is an evolving model of healthcare delivery that achieves better patient outcomes and greater financial sustainability. According to this innovative approach, the value is determined by a panel of indicators and the ratio between results and costs.
• We developed a panel of thoracic-fashioned key-performance indicators creating a model that could be applied in thoracic surgery for the first time, reporting our early experience.
What is the implication, and what should change now?
• Our score could successfully identify improvements needed and quantify their effectiveness in thoracic surgery, anyway further longitudinal prospective trials are demanded to establish definitive KPIs and to validate our PVTS Score.
Introduction
The SARS-CoV-2 pandemic, which has recently overwhelmed the world with effects that are still to be established, has clearly highlighted the gap between the demands and the resources available for healthcare services. In fact, the former often exceed the latter (1). In the current historical period, in which hospitals have had to reorganized themselves to face off the coronavirus disease 2019 (COVID-19) plague, the need for a new concept of healthcare system, no more lying exclusively on the principle of reducing costs but targeted to improve care for the patient, has emerged.
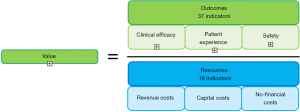
In the early 2000s, Porter and Teisberg defined the value in a healthcare stage as the resultant of patient outcomes per unit of cost, thus introducing the value-based healthcare theory (2) (Figure 1). According to their theory, achieving the highest value for the patient should become the main goal of healthcare system, since aiming at cost reduction without considering outcomes can lead to improper savings, by reducing effective care (3). The best strategy for saving money while maintaining a high quality of care is to invest more in selected services in order to reduce the need for others. This is a fundamental part of Lean, the management system based on waste reduction; “waste” is commonly defined as any action that does not add value. The concept of Value-Based HealthCare (VBHC) encompasses the concept of healthcare efficiency, but it is not limited to that. According to the VBHC theory, the value comprises the entire cycle of treatments that the patient undergoes, from a multidimensional point of view, and is obtained comparing outcomes, as the complete set of outcomes obtained, and costs, as the total costs incurred to achieve those outcomes (4,5). In the recent years several medical centers around the world have adopted components of VBHC (6-11), reporting encouraging results, even if measurement of the value is a controversial and ambiguous topic. Indeed, the complexity of fully understanding the topic and the reluctance in considering the healthcare system in terms of outcomes that matter to patients have prevented to extensively adopt VBHC (12). This is especially true in the surgical world, where the shift from volume-based to value-based care is frowned upon, indeed literature is lacking rigorous scientific reports (7). However, it is right in surgery where the maximization of the ratio between outcomes and costs could lead to a major improvement. To date, no studies have been conducted to evaluate the applicability of VBHC in thoracic surgery, even if efforts for hospital-wide solutions that allows to maximize effectiveness, efficiency and timeliness of treatments are mandatory in our field (13,14). Our project goal was to direct the improvement and integration of processes at our department towards the recognition of the value generated, by adopting an embryonic VBHC approach. Therefore, we elaborated a panel of key-performance indicators (KPIs) and we developed an operational score allowing to evaluate in a multidimensional perspective the treatment path of patients and objectively monitoring its evolution over time in the light of the improvement actions introduced. We report our early experience on the application of the score in our department with the aim of making a first step towards the VBHC in thoracic surgery, hoping to inspire other units to do the same. We present the following article in accordance with the STROBE reporting checklist (available at https://jtd.amegroups.com/article/view/10.21037/jtd-22-1294/rc).
Methods
We designed a cross-sectional retrospective observational study, analyzing data relating to lung cancer patients admitted to our Thoracic Surgery Department of the San Gerardo Hospital, ASST Monza, and surgically treated by lung resection, from January 2017 to December 2019. All the patients undergoing lung resections for lung cancer in that period at our department were enrolled. Thanks to the medical and nursing staff at our unit, with the collaboration of control structure staff, an “As-is” analysis was carried out. Data were retrospectively collected from medical and nursing records. Data consisted in outlining the treatment pathway of lung cancer patients, accurately mapping their physical movements within our hospital during all hospital stay and collecting context data (e.g., personnel involved, number of visits, waiting times, etc.) for all the phases of the treatment process. Patient experience was investigated through questionnaires administered at discharge, with a level of satisfaction from 1 to 7.
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013) and informed consent was taken from all participants. Considering the purely retrospective nature of the study and that all the procedures being performed were part of the routine care, ethical approval was waived by the Ethics Committee of San Gerardo Hospital.
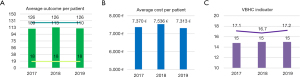
These data were collected in order to have an operational tool that allows to evaluate in a multidimensional perspective the treatment path of patients and to objectively monitoring its evolution over time in the light of the improvement actions introduced. Therefore, we developed a VBHC tool that determines the value using established indicators and named it Patient Value in Thoracic Surgery (PVTS) score. The model predicts that the value is estimated on the basis of indicators, therefore we have identified and developed a set of KPIs according to the VBHC model principles and compliant with the thoracic surgery field. To meet these criteria, we investigated the last ten years literature regarding path of care of lung cancer patients, with particular interest in (I) enhanced recovery after surgery (ERAS) protocol (15-20), (II) main indicators of clinical efficacy in thoracic surgery, (III) studies concerning the application of VBHC in other surgical fields (8-11). In particular, KPIs covered both outcomes (understood as the complete set of outcomes obtained) and resources (understood as the total costs incurred to achieve these outcomes). Thirty-seven indicators concerning outcomes, divided in 3 categories, were found: 16 related to clinical efficacy, 16 to patient experience and 5 to safety; 18 indicators regarding resources, also divided in 3 categories, were found: 10 about revenue costs, 6 on capital costs and 2 on non-financial costs (Figure 2).

Statistical analysis
The 7-level Likert scale was applied to calculate the outcomes items (where 1 represents the worst result and 7 the best), as exemplified in Table 1, trying to standardize the measurement of each variable, so that a numerical value from 1 to 7 was assigned to each outcome item. The Pareto’s principle was applied (21), meaning that only 80% of data associated to a specific indicator were significant and included in the scale, excluding the less frequent values through an ad hoc evaluation of each indicator. From this base, the minimum and the maximum values of the indicator were identified, allowing to later define ranges of each level of the scale. The calculation of the overall resources was carried out through the sum of the individual economic results on the individual defined resource indicators. The overall value was obtained by dividing the total outcomes per total resources, multiplying the ratio per 1,000 to make easier reading the results (Figure 1).
Table 1
| Outcome item | Level 1 | Level 2 | Level 3 | Level 4 | Level 5 | Level 6 | Level 7 |
|---|---|---|---|---|---|---|---|
| Average length of stay, days | ≥11 | 11–9 | 9–7 | 7–5 | 5–4 | 4–3 | <3 |
| Length of stay in intensive care, days | ≥3 | 3–2.5 | 2.5–2 | 2–1.5 | 1.5–1 | 1–0.5 | <0.5 |
| 30 days mortality | ≥3.30% | 3.30%; 2.75% | 2.75%; 2.20% | 2.20%; 1.65% | 1.65%; 1.10% | 1.10%; 0.55% | <0.55% |
| Time between first referral and surgery, days | ≥48 | 48–41 | 41–33 | 33–26 | 26–19 | 19–11 | <11 |
| Time between referral and outpatient appointment, days | ≥21 | 21–18 | 18–14 | 14–11 | 11–7 | 7–4 | <4 |
The 7-level Likert scale was applied to calculate the outcomes items (where 1 represents the worst result and 7 the best): the Pareto’s principle was applied, meaning that only 80% of data associated to a specific indicator were significant and included in the scale, excluding less frequent values through an ad hoc evaluation of each indicator.
Data on lung cancer patients admitted to our Thoracic Department has been uploaded to the PVTS score. The score was structured in three different sections. Section 1 dedicated to the collection and systematization of the available data relating to the outcome indicators for each patient and the cost data available for the three years under examination. The staff in charge of using the tool periodically filled in these sections with the new available data. Section 2, an automatic compilation section showing for each patient the value of the outcome indicators, the related costs, and finally the generated value. Section 3 showing the aggregate outcome and cost values and the VBHC indicator for each of the years taken into consideration. By relating the three outcome categories with the three resource categories, we were able to determine: (I) the value of care for each single patient; (II) the average value of the care for all the patients managed in a specific period of time; (III) the impact of a single indicator on the value both for the individual patient and the total number of patients managed in a specific period of time.
Results
From January 2017 to December 2019, 573 lung cancer patients have been admitted to our Thoracic Surgery Department and surgically treated through lung resections. Among them, 21 patients had missing data and were ruled out. A total of 552 eligible patients have been enrolled. 336 (61%) were male. The average age was 62±6.7 years. The treatment pathway of lung cancer patients from the first medical contact up to the surgical act has been outlined. On average 30 days have elapsed for the completion of treatment pathway. The recording of whole patient’s movements within Hospital during surgical hospitalization allowed to draft a “spaghetti” map. The overall average distance covered by patients was 1.8 km, from arrival at the hospital to discharge. The “As-is” analysis has allowed to identify strengths and weaknesses within our lung cancer patients’ treatment pathway. The strengths were (I) adherence to the ERAS protocol, (II) presence of dedicated slots for patients coming from Thoracic Surgery who have to carry out diagnostic tests, (III) short times between first referral and surgery, while the weaknesses were (I) mainly paper documentation, (II) high number of physical movements required by the patient in the diagnostic phase, (III) lack of communication to the nursing staff regarding the patient’s return to the ward after surgery, (IV) absence of a standard rehabilitation path for the patient’s respiratory physiotherapy before and after surgery, (V) lack of a nursing discharge letter containing post-operative guidance and advice. We focused on the 3 most critical weaknesses and depicted an evolutionary scenario as an accompaniment to change. To overcome the lack of communication to the nursing staff regarding the patient’s return to ward after surgery, with the aim of improving the organization of postoperative activities, a tracking system of the patient has been developed and the supplier confirmed the possibility of extending the service allowing the nursing staff to be informed about patient’s return to the ward. To remedy the absence of a standardized respiratory physiotherapy path, a new therapeutic protocol has been drafted with collaboration of specialists from Rehabilitation Unit, gaining a systematization of patient management methods through the carrying out of pre-surgery evaluation with the physiatrist to improve the patient’s physical condition, and post-surgery visit with the physiotherapist to monitor and implement recovery. To rectify the lack of a nursing discharge letter containing post-operative guidance and advice, our department, in collaboration with the nursing coordinator, has been evaluated the possibility of introducing an information support for nursing discharge letter, which was confirmed as technically feasibly by the supplier.
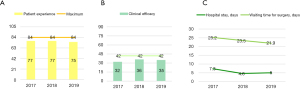
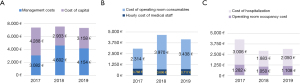
We then applied our operational tool made up of a panel of the KPIs previously developed according to a VBCH approach. Using the score, final results were obtained. Figure 3A shows the average outcome per patient in each year, calculated through the sum of all the outcome features collected: the upper line represents the maximum achievable (n=126) whereas the lower line represents the minimum (n=18). Based on the available variables, the outcome performance of our clinical center, in a 3-year analysis, was constantly high: 109 in 2017, 113 in 2018, 110 in 2019. Average cost per patient, calculated through the sum of all the cost features collected, were: 7.370 euros in 2017, 7.536 in 2018, 7.313 in 2019 (Figure 3B). The annual trend of outcomes and costs indicators is reflected on the PVTS score (VBHC indicators) as showed in Figure 3C. According to our data, the values are aligned in the last three years on n=15. The indicator differs slightly from the maximum obtainable with the economic resources used in that period, represented by the upper line (n=17.1 in 2017, n=16.7 in 2018, n=17.2 in 2019). Regarding outcomes in detail, Figure 4 represents patient experience for each year analyzed and clinical efficacy gained per year. Based on variables evaluated at the ASST, the patient experience is practically constant over the years, approaching the maximum achievable value (n=84), in particular it approximated to 77 in 2017 and 2018, while is about 75 in 2019 (Figure 4A). There is an improvement in clinical efficacy value, from n=32 in 2017 to n=35 in 2019, getting closer to the maximum value (n=42) (Figure 4B). The average hospital stays and waiting time for surgery went from n=25.2 in 2017 to n=21.9 in 2019 and from 7.3 in 2017 to 5 in 2019 respectively, with a clinically significant reduction (Figure 4C). Regarding resources in detail, Figure 5 shows the amount of money spent per year divided in management costs and cost of capital. Based on cost items collected (a minority compared to those identified in the panel, which would allow a complete assessment), a decrease in the cost of capital compared to 2017 emerges, from 4.288 to 3.158 euros, and, on the contrary, management costs increased from 3.082 to 4.154 euros (Figure 5A). Operating costs were divided in consumables and medical staff for each year. The increase in operating costs in 2018 and 2019 was due to an increase in cost of materials for the operatory room (materials are allocated to the year of purchase even if used for several years), which reached the peak in 2018 with 3.970 euros spent for operatory room consumables, whilst the hourly cost of medical staff has evolved steadily (Figure 5B). Lastly, costs of hospitalization per year considerably decreased from 3.006 euros in 2017 to 2.050 euros in 2019 (Figure 5C).
Discussion
Since 2006, when Porter and Teisberg first introduced the concept of VBHC (2), involvement in this field has exponentially increased. The focus at the center of this model is to increase the value for patients, by aligning everyone around the common aim of doing what’s right for patients. The shift from volume-based to value-based healthcare is an incredible breakthrough in the medical world, which mirrors the shift from a paternalistic doctor-patient relationship to a peer to peer one, based on mutual collaboration. The patient in fact, instead of being still an empty jar prone to be fulfilled by physicians will, has become an active player in the healthcare system, aware of himself and empowered in implementing his own well-being. In this context, the evolution of the healthcare, aimed at what matters to the patient, is unavoidable and can no longer being postponed. Fantini et al. (6) has well underlined the need for a new culture of health, re-thinking the delivery of prevention and healthcare. The ideas have proven their worth in renowned centers worldwide, including the Cleveland Clinic, UCLA Medical Center, Joslin Diabetes Center, MD Anderson Cancer Center, Children’s Hospital of Philadelphia, the Schön Klinik, Oak Street Health, Santeon, and Erasmus MC. Results presented in literature are encouraging. Goretti and colleagues (8), in the bariatric surgery setting, demonstrated that developing a VBHC framework as a patient-centered approach is sustainable and replicable, and it can increase health outcomes, by improving patients’ adherence to therapeutic pathway. Gabriel and collaborators (9), in the orthopedic field, highlighted that a redesigned pathway of care based on VBHC can increase value, pointing at development of patient-oriented outcomes and evaluation of economic resources as key points to achieve better value care. Mofidi et al. (22) have tried to describe the creation of VBHC delivery systems in vascular surgery services in the United Kingdom. They underlined the importance of adopting a value chain, made up with mutually reinforcing steps for change: organizing care into integrated practice units, defined as extensions of multidisciplinary teams organized around a specific medical condition; measuring outcome and cost for every patients, to identify how reorganizing activities in order to increase the former and to reduce the latter; integrated care across separate facilities, utilizing hub and spoke arrangements; the importance of enabling technology platform, to enhance the delivery of services and to support value based improvements. Pollock et al. (10) reported significant improvement in cancer-related outcomes at MD Anderson cancer center in Houston, Texas, after the introduction of a wide reorganization of the institution, based on two key elements: the establishment of dedicated medical practice units and the permanent assessment of the quality that is produced. Novikov et al. (23) outlined the development and establishment of VBHC in orthopedic units in the United States of America, whose breakthrough was introduced by the Acute Care Episode Demonstration Project, a pilot study assessing the feasibility of per-episode payment systems to reduce costs and to improve quality in healthcare services. Moreover, Reitblat et al. (24) depicted the adoption of VBHC in the urology field, suggesting promising early results, once again based on the value agenda: organize care around medical conditions, measure outcomes and costs for every patient, move to bundled payments for care cycles, integrate care delivery systems, expand geographic reach, build an enabling information technology platform. They interestingly conclude that healthcare delivery has become fragmented with divergent goals but improving value for patients represents a unifying goal. In a recent systematic review by Zanotto et al. (12) focused on the identification of measures adopted in the application of the value agenda, it clearly appears that in a real-world setting there is still a gap in measuring outcomes which are important to patients, since a considerable proportion of studies were still based on cost savings rather than on patient-centered items. So, while most experts agree on the importance of putting the value of the patient at the center of healthcare system, it is still not clear how to do it. First, we suggest carrying out an “As-is” analysis, through which to evaluate the strengths and identify the weaknesses of the ongoing process, to implement quality. In particular, the application of the ERAS protocol, the presence of dedicated slots for thoracic surgery patients and the short-term period of diagnosis have emerged as strong points of our department; otherwise, the detection of weak points has allowed us to promptly design solutions to correct them. Through our experience, we have drawn and proposed a panel of 55 key performance indicators, describing how the value can be defined in lung cancer patient, trying to consider the multidimensionality of the patient. Then we have developed a PVTS score, based on the KPI proposed. The application of our score has given us the opportunity to calculate the value generated for each patient and the average value in a reference period, allowing us to monitor its evolution over time and to measure the impact of the improvement actions introduced. We believe that the multidimensional nature of the panel pushes clinical centers to measure and improve themselves also on the management aspects, which affect both the outcomes perceived by the patient and the employed resources. The score was developed so that it can be used independently by the clinical center, thus allowing the staff at Monza ASST to measure their performance periodically. Based on the variables evaluated at our hospital in the last three years, an overall positive performance was recorded, aligned in the last three years around a value of n=15, which differs slightly from the maximum obtainable with the economic resources used, in particular thanks to the results obtained. In fact, the average outcome per patient has slightly increased compared to 2017, reflecting the high satisfaction reported by the patients. Moreover, there was an improvement in clinical efficacy value, from n=32 in 2017 to n=35 in 2019, getting closer to the maximum value (n=42), mainly thanks to the reduction in days of hospitalization and number of days that elapse between the patient’s inclusion on the waiting list and surgery. However, we think it is mandatory to pay attention to the costs, especially the management ones, subject to annual fluctuations. In fact, the average cost per patient slightly increased in 2018, mainly because of an unexpected increase of the management costs. In addition, in the same year, a rise in the cost of operating room (OR) consumables have been registered; however, despite the increase in the number of patients, due to the decrease in hospital stay in 2018 and 2019, the average cost of hospitalization also decreased, positively impacting the overall cost of capital and mitigating the higher cost of consumables in the OR. These results are most likely due to the fact that between 2017 and 2018 a uniportal surgery program was started, entailing on the one hand an initial cost for the instrumentation and on the other a learning curve distributed over the months.
Our study has some limitations. Firstly, the application of the VBHC Tool at Monza ASST was partial, as it was possible to calculate only a part, equal to about one third (Figure 2) of the indicators identified in the panel: therefore, results obtained in 2017, 2018 and 2019 are incomplete and represent only a starting point, which is worth developing shortly. Some variables considered are not easily available in real-life setting, but we believe that they should be anyway included in the PVTS score to make it completer and more holistic. Today, multidisciplinary team working is mandatory, because most thoracic malignancies are not just surgical problems, and the process of care includes many more resources than surgery-related ones. Unfortunately, data not directly related to surgical activity were not available and therefore they were not applied in the score. In addition, follow-up of the patients enrolled is still ongoing and related parameters were not included in the score, however they will be analyzed and integrated as soon as the follow-up will be complete. Evolution of the PVTS score should be taken with caution in accordance with the numerous lacking data and, before considering our tool to be universally applied, it requires longitudinal prospective validation. Moreover, data from our study were specifically derived from lung cancer patients, narrowing the field of interest to a Thoracic Oncology Surgery Department and preventing us from generalizing our conclusions to non-neoplastic thoracic surgery activity. Further studies focused on the whole surgical activity of a Thoracic Unit must be conducted. Nevertheless, our experience testifies that a first step towards VBHC delivery implementation is feasible in the setting of lung cancer patients’ surgical management, confirming the encouraging results presented in literature on the feasibility of introducing VBHC in lung cancer patients’ medical management (25). Solidity and cleanliness of the underlying data are fundamental elements for obtaining results that adhere to reality: the PVTS score can therefore represent an opportunity to review and strengthen the logic with which some information is traced (e.g., consumables used in the OR). We strongly believe that the application of a validated score could allow to promote value-based medicine, looking with equal attention to results and costs, introducing and monitoring improvement actions. The use of the score, therefore, could allow to better understand the impacts of an improvement action on the different dimensions, helping to make decisions aimed at maximizing the value for the patient. For example, considering the improvement action “Tracking the patient’s return to the ward after surgery”, despite the additional cost required by the improvement, there was a positive impact on 5 outcome indicators. PVTS score allows to analyze an improvement action based on the effects it will have not only on implementation and management costs, but also on clinical effectiveness, patient’s experience and safety, so as to incentivize a procurement model based on the benefits, the evaluation of agreements of “risk-sharing” with suppliers, overcoming the “silos” management logic.
Conclusions
The adoption of a VBHC system is demanding but required by the times we live. Introducing a new concept of value, VBHC model applied to thoracic surgery may revolutionize traditional organizational management in lung cancer patients showing how value delivered can increase in accordance with outcomes, despite the growth of part of the costs. Our operational PVTS score allowed us to analyze the value trend over the recent years and our descriptive report showed encouraging results on its partial application in our department. Moreover, our panel of indicators has been created to provide an innovative score to successfully identify improvements needed, quantify their effectiveness, and ideally allow comparison between different thoracic surgery centers. Further longitudinal prospective trials are demanded to establish definitive KPIs and to validate our PVTS score.
Acknowledgments
We thank Dr. Gerardo Cioffi, a native English speaker, for reviewing the English language.
The manuscript was presented at the 29th European Conference on General Thoracic Surgery (virtual meeting), and it was read on Sunday 20 June 2021.
Funding: None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://jtd.amegroups.com/article/view/10.21037/jtd-22-1294/rc
Data Sharing Statement: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-22-1294/dss
Peer Review File: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-22-1294/prf
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jtd.amegroups.com/article/view/10.21037/jtd-22-1294/coif). PF serves as an unpaid editorial board member of Journal of Thoracic Disease from October 2022 to September 2024. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013) and informed consent was taken from all participants. Considering the purely retrospective nature of the study and that all the procedures being performed were part of the routine care, ethical approval was waived by the Ethics Committee of San Gerardo Hospital.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Gray M. Value based healthcare. BMJ 2017;356:j437. [Crossref] [PubMed]
- Porter ME, Teisberg EO. Redefining Health Care: Creating Value-Based Competition on Results. 2006 Boston, MA: Harvard Business School Press.
- Porter ME. What is value in health care? N Engl J Med 2010;363:2477-81. [Crossref] [PubMed]
- Porter M, Lee TH. The strategy that will fix health care. Harv Bus Rev 2013;91:50-705.
- Teisberg E, Wallace S, O'Hara S. Defining and Implementing Value-Based Health Care: A Strategic Framework. Acad Med 2020;95:682-5. [Crossref] [PubMed]
- Fantini B, Vaccaro CM. Value based healthcare for rare diseases: efficiency, efficacy, equity. Ann Ist Super Sanita 2019;55:251-7. [PubMed]
- van Deen WK, Spiro A, Burak Ozbay A, et al. The impact of value-based healthcare for inflammatory bowel diseases on healthcare utilization: a pilot study. Eur J Gastroenterol Hepatol 2017;29:331-7. [Crossref] [PubMed]
- Goretti G, Marinari GM, Vanni E, et al. Value-Based Healthcare and Enhanced Recovery After Surgery Implementation in a High-Volume Bariatric Center in Italy. Obes Surg 2020;30:2519-27. [Crossref] [PubMed]
- Gabriel L, Casey J, Gee M, et al. Value-based healthcare analysis of joint replacement surgery for patients with primary hip osteoarthritis. BMJ Open Qual 2019;8:e000549. [Crossref] [PubMed]
- Pollock RE. Value-based health care: the MD Anderson experience. Ann Surg 2008;248:510-6; discussion 517-8. [Crossref] [PubMed]
- Harrison CJ, Tyler MPH, Rodrigues JN. Value-based plastic surgery. J Plast Reconstr Aesthet Surg 2020;73:2106-10. [Crossref] [PubMed]
- Zanotto BS, Etges APBDS, Marcolino MAZ, et al. Value-Based Healthcare Initiatives in Practice: A Systematic Review. J Healthc Manag 2021;66:340-65. [Crossref] [PubMed]
- Horvath KA. Finding the Value in Value-Based Care. Ann Thorac Surg 2021;112:16-21. [Crossref] [PubMed]
- Medbery RL, Force SD. Quality and Cost in Thoracic Surgery. Thorac Surg Clin 2017;27:267-77. [Crossref] [PubMed]
- Gonzalez M, Abdelnour-Berchtold E, Perentes JY, et al. An enhanced recovery after surgery program for video-assisted thoracoscopic surgery anatomical lung resections is cost-effective. J Thorac Dis 2018;10:5879-88. [Crossref] [PubMed]
- Rogers LJ, Bleetman D, Messenger DE, et al. The impact of enhanced recovery after surgery (ERAS) protocol compliance on morbidity from resection for primary lung cancer. J Thorac Cardiovasc Surg 2018;155:1843-52. [Crossref] [PubMed]
- Li S, Zhou K, Che G, et al. Enhanced recovery programs in lung cancer surgery: systematic review and meta-analysis of randomized controlled trials. Cancer Manag Res 2017;9:657-70. [Crossref] [PubMed]
- Van Haren RM, Atay SM. Enhancing the study of enhanced recovery after thoracic surgery: methodology and population-based approaches for the future. J Thorac Dis 2019;11:S612-8. [Crossref] [PubMed]
- Semenkovich TR, Hudson JL, Subramanian M, et al. Enhanced Recovery After Surgery (ERAS) in Thoracic Surgery. Semin Thorac Cardiovasc Surg 2018;30:342-9. [Crossref] [PubMed]
- Mayor MA, Khandhar SJ, Chandy J, et al. Implementing a thoracic enhanced recovery with ambulation after surgery program: key aspects and challenges. J Thorac Dis 2018;10:S3809-14. [Crossref] [PubMed]
- Juran JM, Godfrey AB. Juran’s Quality Handbook. 5th ed. New York: McGraw-Hill, 1999.
- Mofidi R, Wong P, Gatenby T, et al. Value based healthcare and delivery of vascular surgery services in the United Kingdom. Reviews in Vascular Medicine 2016;6:1-9. [Crossref]
- Novikov D, Cizmic Z, Feng JE, et al. The Historical Development of Value-Based Care: How We Got Here. J Bone Joint Surg Am 2018;100:e144. [Crossref] [PubMed]
- Reitblat C, Bain PA, Porter ME, et al. Value-Based Healthcare in Urology: A Collaborative Review. Eur Urol 2021;79:571-85. [Crossref] [PubMed]
- Demedts I, Himpe U, Bossuyt J, et al. Clinical implementation of value based healthcare: Impact on outcomes for lung cancer patients. Lung Cancer 2021;162:90-5. [Crossref] [PubMed]


