Malignant giant cell tumor of the rib with lung metastasis in a man
Introduction
Giant cell tumor of bone (GCTB) usually appears as a benign tumor with local aggressiveness. GCTB mainly invades the end of long bones but rarely the ribs (1). Only 2% of all cases may evolve into a malignant one, and most of these malignancies occur in GCTB patients who underwent radiotherapy. In this paper, we present the imaging and clinicopathologic findings of a 57-year-old man with primary malignancy in GCTB (PMGCTB) and superior lobe of right lung metastasis.
Patients and methods
Our study was approved by the West China Hospital Ethics Committee and the patients signed an informed consent form. The patient underwent operation and radiation treatment in our hospital. The sample was confirmed with immunohistochemistry. All the regents were kindly provided by the pathology department of our hospital.
Results
The 57-year-old male went to our hospital with the chief complaint of chest pain on the right side for about half a year. He denied tuberculosis and had no history of surgery or trauma. His medical history was unremarkable.
Physical examination showed nothing remarkable and no thoracocyllosis. Contrast-enhanced computed tomography (CT) showed a large nearly ovoid mass on the right side of the chest wall with corresponding rib destruction (Figure 1). The mass showed dense soft tissue and obvious strengthening. Some nodules were found in the right lung. A little of pleural effusion was seen in the right thoracic cavity. Both sides of pulmonary lobes show mild chronic inflammation. Single photon emission tomography computed tomography (SPETCT) scan showed a mass on the right side of the third rib. The adjacent ribs, the second and the fourth ribs were also damaged. To identify the pathologic type of this mass, a fine needle biopsy was performed and the result was giant cell tumor. Laboratory tests including serum calcium, serum phosphorus, acid phosphatase, and alkaline phosphatase tests were unremarkable. Neither contrast-enhanced CT nor SPETCT found other metastasis except the right lung. The patient had no obvious contraindication. He received operation and the tumor was resected en bloc with 13-cm-long part of the involved ribs (including the third and the fourth ribs on the right sides). Surgical margin was 4 cm far away from the mass. The superior lobe of the right lung was also resected and the chest wall was reconstructed using Dacron patch (Figure 2). A cystic zone (6 cm × 4 cm × 3 cm) with hemorrhagic necrosis was found in this lesion. Histopathologic examination confirmed the complete resection of the lesion, which was measured to be 10 cm × 6.5 cm × 4.8 cm and diagnosed to be PMGCTB with aneurysmal bone cyst. Immunohistochemical examination (IHC) showed that P63 and Ki-67 were stained positive (about 30-40%), while smooth muscle actin (SMA), S-100, CR, HBME1, Desmin, Caldesmon, PCK and CD31 were stained negative. The IHC results not only confirmed the diagnosis of MGCTB but also ruled out the possible mesothelioma of pleura or muscle-derived tumors (Figure 3). Besides, no positive cytological results were found in the pleural effusion. Subsequently, this man underwent radiation treatment to reduce the high rate of recurrence and metastasis. Follow-up was performed with CT every three months. Since the operation and radiotherapy, the man has been free of recurrence or metastasis for 1.5 years.
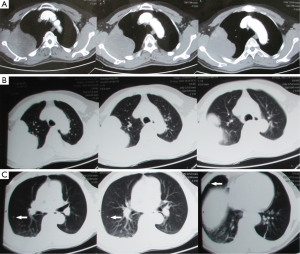
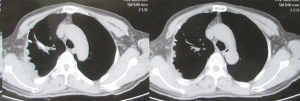

Discussion
MGCTB is a very rare subtype of GCTB and only accounts for 1-2% of all reported cases (2). This disease was previously called malignant giant cell tumor which includes giant cell—rich osteosarcomas, malignant fibrous histiocytoma, locally aggressive giant cell tumor of bone, and metastatic benign giant cell tumor. This tumor was renamed malignancy in giant cell tumor by Dahlin et al. and divided into primary and secondary malignancies (3). Of which, PMGCTB is extremely rare.
We reported a 57-year-old man suffering from PMGCTB in his rib. Unni (4), who also took part in defining and describing this disease, considered that patients suffering from MGCTB were older than those suffering from benign giant cell tumors. Our finding is consistent with Domovitov et al. (5). Like benign GCTB, the malignancies preferentially invade the ends of long bone including distal femur, proximal tibia and distal tibia. Pelvis and sacrum are also common invading sites.
The clinical manifestation of PMGCTB is always nonspecific. The most common symptoms are pain and swelling, which may last for months (average 12 months; median 6 months) (2). Radiologic findings may show osteolytic lesions with well-circumscribed margins that are similar to those of conventional GCTB (6). We could not easily distinguish malignancies from the benign ones even when the former has a metastasis or a soft tissue invasion, because the benign ones also have the capacity of local invasion.
The diagnostic criteria of MGCTB were first made by Jaffe et al. (6). The MGCTB contained a giant-cell lesion and sarcomatous stroma. Hutter et al. (7) and Dahlin et al. (3) further elaborated the criteria. Briefly, MGCTB had plump spindle or ovoid shaped stromal cells with multinucleated giant cells intermingling. The nuclei were elongated and vesicular. The cytoplasm was pink and has distinctly circumscribed borders. Most cells had hyperchromatic nuclei and their nucleoli were prominent. Mitotic activity could be always present with atypical mitosis. Usually, there was almost no intercellular substance. The case in our study suited all the MGCTB characteristics mentioned above. According to the definition by Dahlin et al. (3), MGCTB is a high-grade sarcoma since it contains both giant-cell lesion and sarcomatous stroma (8). Almost all lesions of PMGCTB contain many conventional giant cells. Except for benign GCTB, the differential diagnosis can be made difficult and confusing by osteosarcoma, fibrosarcoma and undifferentiated high-grade pleomorphic sarcoma which also contain giant cells. MGCTB is quite different from the benign ones in terms of treatment and prognosis, and thus, pathological diagnosis is of great importance. We suggest that more samples should be obtained when using biopsy. Other than pathological examinations, immunohistochemistry argument is also as helpful and important. P53 was a tumor suppressor gene, the expression of which would be very strong in secondary MGCTB and play a role in the malignant transformation of benign GCTB (9,10). P63, one kind of P53 family, is considered to be a special mark of GCTB (11). Besides, we doubt whether the cell-cycle-associated proteins, such as P63 and Ki67 that reflect cell proliferation, may help with differential diagnosis (9).
In addition to correct diagnose, resection and reconstruction of the chest wall is another important thing. Different views are existent referring to the extension of resection and 4 cm is selected as a common distance between surgical margin and malignant lesions (12). Chest wall reconstruction is thought to be necessary if the defect is wide and located in anterior or lateral chest wall (13). Dacron patch, medical polymethacrylate and metallic material are commonly used. Marlex and polypropylene net are popular in recent years (14).
The treatments of PMGCTB include surgery, chemotherapy, radiotherapy, and surgery combined with chemotherapy. Previous studies show that patients treated by surgery combined with chemotherapy had a significantly higher one-year survival rate than those treated by surgery alone, but the five-year survival rates were almost the same. Since radiotherapy alone was not recommended (8) and Ki67-positive cells are chemo-resistant (9), we chose surgery combined radiotherapy according to the National Comprehensive Cancer Network Guideline. The prognosis of this disease is controversial. But Stepan thought that PGMCTB had a high survival rate of 50-70% (5).
In summary, PMGCTB is a confusing disease, since its clinical manifestation and radiologic findings were nonspecific and similar to that of benign GCTB. Even pathological examination as the gold standard may make a mistake. We know the secondary MGCTB is derived from surgery or radiotherapy-treated benign ones, but the origination of PMGCTB is still unknown though P53 may play a role in this progress (10). We reported a person suffering from PMGCTB in ribs and treated by combining surgery with radiotherapy. The patient has been free of recurrence or metastasis for 18 months. From this result we could learn that an operation followed by radiotherapy was effective for the patients suffered from Ki67-positive-PMGCTB. Follow-up should continue and more samples should be collected to further study this disease.
Acknowledgements
We appreciate the staff of the Department of Thoracic Surgery and Department of Pathology, West China Hospital, Sichuan University, Chengdu, China.
Disclosure: The authors declare no conflict of interest.
References
- Moschouris H, Marinis A, Bouma E, et al. Nonepiphyseal giant cell tumor of the rib: a case report. Case Rep Oncol Med 2012;2012:745292.
- Bertoni F, Bacchini P, Staals EL. Malignancy in giant cell tumor of bone. Cancer 2003;97:2520-9. [PubMed]
- Dahlin DC, Cupps RE, Johnson EW Jr. Giant-cell tumor: a study of 195 cases. Cancer 1970;25:1061-70. [PubMed]
- Unni KK. eds. Dahlin’s bone tumors—general aspects and data on 11,087 cases. 5th ed. Philadelphia: Lippincott-Raven Publishers, 1996:263-87.
- Domovitov SV, Healey JH. Primary malignant giant-cell tumor of bone has high survival rate. Ann Surg Oncol 2010;17:694-701. [PubMed]
- Jaffe HL, Lichtenstein L, Portis RB. Giant cell tumour of bone. Its pathologic appearance, grading, supposed variants and treatment. Arch Pathol 1940;30:993-1031.
- Hutter RV, Worcester JN Jr, Francis KC, et al. Benign and malignant giant cell tumors of bone. A clinicopathological analysis of the natural history of the disease. Cancer 1962;15:653-90. [PubMed]
- Anract P, De Pinieux G, Cottias P, et al. Malignant giant-cell tumours of bone. Clinico-pathological types and prognosis: a review of 29 cases. Int Orthop 1998;22:19-26. [PubMed]
- Alberghini M, Kliskey K, Krenacs T, et al. Morphological and immunophenotypic features of primary and metastatic giant cell tumour of bone. Virchows Arch 2010;456:97-103. [PubMed]
- Gong L, Liu W, Sun X, et al. Histological and clinical characteristics of malignant giant cell tumor of bone. Virchows Arch 2012;460:327-34. [PubMed]
- Lee CH, Espinosa I, Jensen KC, et al. Gene expression profiling identifies p63 as a diagnostic marker for giant cell tumor of the bone. Mod Pathol 2008;21:531-9. [PubMed]
- Sun L, Zhang G, Wang G, et al. Chest wall tumor resection and chest wall reconstruction. Chin J Clin Oncol Rehabil 2011;18:346-9.
- Ma S, Shen L, Li S, et al. Chest wall resection and reconstruction for thoracic tumor invading the chest wall: a report of 12 cases. Zhongguo Fei Ai Za Zhi 2012;15:90-6. [PubMed]
- Mansour KA, Thourani VH, Losken A, et al. Chest wall resections and reconstruction: a 25-year experience. Ann Thorac Surg 2002;73:1720-5; discussion 1725-6.


