
This article has an erratum available at: http://dx.doi.org/10.21037/jtd-2021-001 and the article has been update on 2021-01-08 at here.
Impact of the novel coronavirus (COVID-19) pandemic on sleep
Introduction
In December 2019, an outbreak of the novel strain severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) emerged in China, and rapidly spread worldwide. The new condition was named coronavirus disease 2019 (COVID-19) by the World Health Organization (WHO) and was declared a pandemic on the 11th of March 2020 (1). Millions of cases were confirmed with hundreds of thousands of deaths. This unprecedented situation and the finding that the virus was highly contagious (person-to-person transmission) (2), required the adoption of non-vaccination public health measures aiming to reduce further spreading of SARS-CoV-2 (2). These measures included track-and-tracing, self-isolation, quarantine, social distancing and community containment, as well as nationwide lockdowns (3). Lockdown measures were implemented in many countries, including the UK, leading to significant social and lifestyle changes. In addition to any direct effects of COVID-19 on people’s health the lockdown had an impact on the perception of confinement, caused worries about livelihood, family or friends, and had indirect effects on the health and wellbeing of non-infected people. The psychological consequences of isolation in epidemics or during quarantine have previously been addressed (4,5), however, with scarce focus on their effects on sleep (6).
In the current pandemic, the effects of isolation have been described in cohorts from China and Italy (7,8), with poor quality sleep and comorbid psychological disturbances being identified as significant issues (9-11). We therefore hypothesised that the COVID-19 pandemic and the public lockdown significantly impacted on sleep in the general population. We compared suspected COVID-19 with non-COVID-19, and tested whether there was an association with mental health impact. In order to survey current sleep problems, the British Sleep Society (BSS) initiated the National Early Detection Screening for the COVID-19 pandemic.
We present this article in accordance with the “Strengthening the Reporting of Observational Studies in Epidemiology” (STROBE) reporting checklist (available at http://dx.doi.org/10.21037/jtd-cus-2020-015).
Methods
Study design
The BSS Research and Executive Committee approved this prospective, cross-sectional, survey-based study. The UKRI/MRC and the NHS Health Research Authority (HRA) confirmed that ethical approval was not required owing to the screening survey design (https://cdn.amegroups.cn/static/application/04712039f3b0c07e77f3c779c14d1a17/JTD-CUS-2020-015-1.pdf). The study conformed to the provisions of the Declaration of Helsinki (as revised in 2003). An online survey was available to the general public from May 12th to June 2nd, 2020 (https://cdn.amegroups.cn/static/application/1778ae9dd49ffd9dfb1da63b65421e93/JTD-CUS-2020-015-2.pdf). Eligible participants were adults aged 18 years or older, living through the 2020 COVID-19 pandemic and under national lockdown regulations. Subjects required literacy and access to the Internet. Participants were excluded if they were not living under national lockdown regulations or were underage.
Questionnaire items
Respondents were asked to answer multiple choice questions or provide a free-text response regarding baseline characteristics, and the impact of the pandemic/lockdown including COVID-19 infection history, self-isolation, key-working, mental health, alcohol and sleep medication use, and a sleep profile (current quality/quantity, change in sleep pattern and specific sleep symptoms) (https://cdn.amegroups.cn/static/application/1778ae9dd49ffd9dfb1da63b65421e93/JTD-CUS-2020-015-2.pdf; Supplementary material).
A total of 24 different sleep symptoms were grouped into six sleep symptom categories for analysis (Table S1): (I) insomnia/disrupted sleep, (II) daytime symptoms, (III) abnormal behaviours in sleep, (IV) sleep-disordered breathing, (V) restless legs, (VI) sleep phase disturbance.

Full table
Primary and secondary outcomes
The primary outcomes were to identify sleep disturbances related to the COVID-19 pandemic lockdown in the general population. Secondary outcomes included subgroup analyses for pre-defined cohorts within the population: reported suspected COVID-19, self-isolation, keyworker and mental health impact.
Statistical analysis
Continuous variables were presented as median [interquartile range, IQR], due to non-normally distributed data. Mann-Whitney U and Chi-squared analyses were performed for comparison of continuous and categorical variables, respectively, in a priori subgroup comparison. A main-effects multiple logistic regression was used to identify the strength of association between the six symptom categories or a reported sleep pattern change and factors relating to the pandemic. Further multiple logistic regression modelling was reported as odds ratios where significant associations were found between sleep symptoms and pandemic-related factors (Supplementary material).
‘Yes’ and ‘maybe’ responses regarding COVID-19 infection were grouped into ‘suspected COVID-19’ for analysis due to global limited availability of polymerase chain reaction (PCR) swab testing. “Don’t know” answers were not included in the modelling as they were non-binary.
Data were collected on Google Forms (accessed online May 2020, Google Ireland Limited, Dublin, Ireland) and analysed with SPSS version 24 (IBM Corporation, New York, USA) and Prism version 8 (GraphPad Software, San Diego, California, USA). A P value of ≤0.05 was considered statistically significant. Missing data were not imputed.
Results
Demographics and descriptive characteristics
844 participants completed the online survey, one underage participant was excluded, and 843 datasets were included in the final analysis. Respondents were mostly female (67.4%), middle aged [52 years (40–63 years)], predominantly white (92.2%), and overweight to obese [BMI 29.4 kg/m2 (24.1–35.5 kg/m2)] (Table 1).
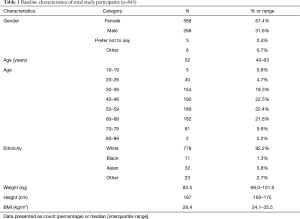
Full table
A proportion of 21.1% of respondents had suspected COVID-19 and 286 participants (33.9%) had to self-isolate. 550 participants (65.2%) reported an impact of the pandemic on their mental health. 296 participants (35.1%) were keyworkers; 219 participants (25.9%) reported drinking more alcohol during the pandemic (Table S2).
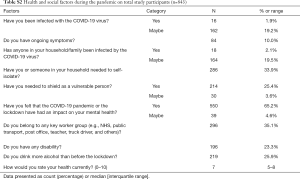
Full table
Impact on sleep
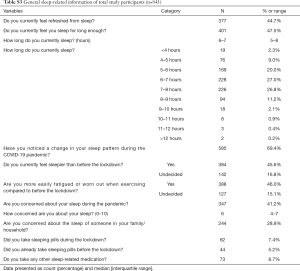
Less than half of the participants (47.5%) felt satisfied with their current sleep quantity or felt refreshed from sleep (44.7%), with 27.0% reporting to sleep 6–7 [5; 8] hours; 585 participants (69.4%) noticed a change in their sleep pattern during the pandemic, 45.6% felt sleepier than before the lockdown and 7.4% reported the use of sleep medication during the lockdown compared to 5.2% before the lockdown (Table S3); 264 (31.3%) participants reported sleep restriction, with <6 hours of sleep. These were older [55 years (42–64 years) vs. 50 (39–62 years), P=0.007), had higher BMI [31.3 (25.7–37.6) vs. 28.3 (23.6–34.4) kg/m2, P<0.001], and were more likely to report shielding due to vulnerability (31.1% vs. 22.8%, P=0.038), disability (30.3% vs. 20.0%, P=0.001) and an impact on their mental health (71.2% vs. 62.5%, P=0.049) (Table S4).
Full table
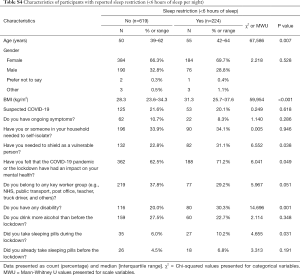
Full table
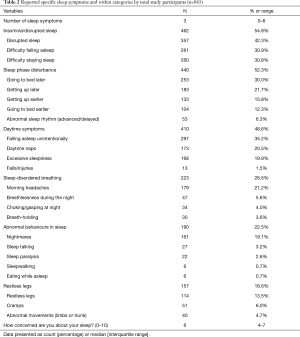
The most commonly reported specific sleep symptoms were disrupted sleep (42.3%), falling asleep unintentionally (35.2%), difficulty falling and staying asleep (30.9% and 30.8%, respectively) and going to bed later (30.0%) (Table 2). Regarding ‘other’ symptoms, a small number of respondents stated that their sleep had improved (1.9%) (Table S5).
Full table

Full table
Effect of self-isolation and keyworker status
A positive answer to household self-isolation status question was assumed to report the participants self-isolating themselves. Five cases were excluded from keyworker group comparison due to “don’t know” responses.
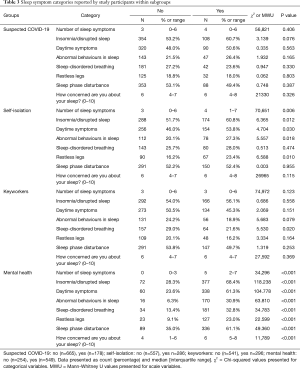
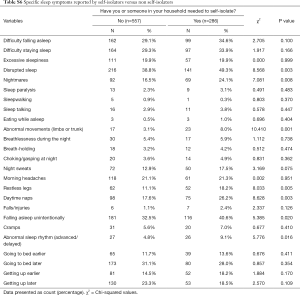
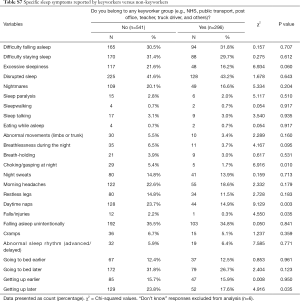
Participants who self-isolated reported significantly more insomnia/disrupted sleep (60.8% vs. 51.7%, P=0.012), daytime symptoms (53.8% vs. 46.0%, P=0.030), abnormal behaviours in sleep (27.3% vs. 20.1%, P=0.018) and restless legs (23.4% vs. 16.2%, P=0.01) (Table 3); specifically, these reported more sleep disruption (49.3% vs. 38.8%, P=0.003), nightmares (24.1% vs. 16.5%, P=0.008), abnormal movements of the limbs/trunk (8% vs. 3.1%, P=0.001), restless legs (18.2% vs. 11.1%, P=0.005), falling asleep unintentionally (40.6% vs. 32.5%, P=0.02) and abnormal sleep rhythm (9.1% vs. 4.8%, P=0.016) (Table S6). Keyworkers described significantly fewer sleep-related and daytime disturbances than others, namely choking/gasping at night (1.7% vs. 5.4%, P=0.010), daytime naps (14.9% vs. 23.7%, P=0.003), falls/injuries (0.3% vs. 2.2%, P=0.035) and getting up later (17.6% vs. 23.8%, P=0.035) (Table S7).
Full table
Full table
Full table
Suspected COVID-19 status
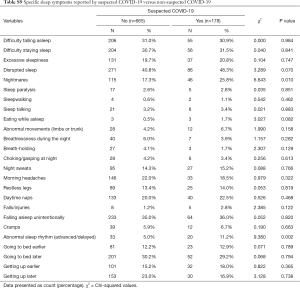
The most reported COVID-19 infection symptoms in the total study participants were dry cough and breathlessness (4.5% each), followed by headache (3.7%) and muscle pain (3.6%) (Table S8). Participants with suspected COVID-19 had significantly more nightmares (25.8% vs. 17.3%, P=0.010) and abnormal sleep rhythm (11.2% vs. 5.0%, P=0.002) (Table S9). Ongoing COVID-19 symptoms were associated with sleep disruption (OR 2.810, 95% CI, 1.632 to 4.867, P<0.001) (Figure 1).

Full table
Full table
Mental health
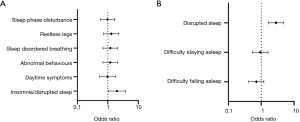
Forty “don’t know” responses were excluded from mental health impact group comparison. Participants reporting an impact of the pandemic on their mental health also had significantly more sleep symptoms {5 [2–7] vs. 0 [0–3], P<0.001} and were more concerned about their sleep {6 [5–8] vs. 4 [1–6] out of 10 points, P<0.001} (Table 3). A multiple logistic regression analysis demonstrated that reported mental health impact was a predictor of sleep pattern change during the pandemic (β=0.969, 95% CI, 0.663 to 1.281, P<0.001, Table S10). In a further regression analysis, an impact on mental health was significantly predicted by the presence of difficulty falling asleep (OR 3.600, 95% CI, 2.317 to 5.752, P<0.001), sleep disruption (OR 2.523, 95% CI, 1.696 to 3.794, P<0.001), excessive sleepiness (OR 3.488, 95% CI, 2.050 to 6.264, P<0.001), falling asleep unintentionally (OR 3.810, 95% CI, 2.588 to 5.726, P<0.001) and nightmares (OR 7.005, 95% CI, 3.951 to 13.600, P<0.001) (Figure 2). An impact on mental health was associated with both increased alcohol intake (OR 1.529, 95% CI, 1.074 to 2.203, P=0.02) and sleep medication use (OR 1.634, 95% CI, 1.036 to 2.644, P=0.039).
Full table
Predictors of a change in sleep pattern
A multiple logistic regression model including ‘change in sleep pattern’ as the categorical outcome variable with baseline demographics and pandemic-related factors as predictor variables was performed (Table S10). No co-linearity was found between predictor variables. A change in sleep pattern was overall most strongly associated with reported sleep medication use during the pandemic (β=1.276, 95% CI, 0.140 to 2.578, P=0.039), ongoing COVID-19 symptoms (β=1.124, 95% CI, 0.368 to 1.937, P=0.005) and reported mental health impact (β=0.969, 95% CI, 0.663 to 1.281, P<0.001), displaying a negative association with age (β=−0.026, 95% CI, −0.040 to −0.012, P<0.001), male gender (β=−0.350, 95% CI, −0.658 to −0.040, P=0.026) and general health rating (β=−0.244, 95% CI, −0.343 to −0.149, P<0.001) (Tjur’s R2=0.177, adjusted R2=0.165).
Association between sleep symptom categories and pandemic factors
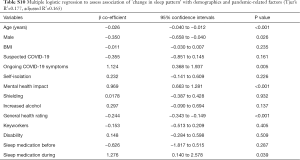
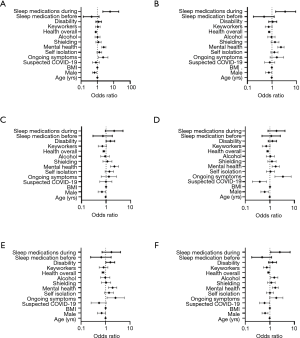
Insomnia/disrupted sleep was significantly associated with mental health impact (OR 2.435, 95% CI, 1.821 to 3.281, P<0.001), ongoing COVID-19 symptoms (OR 2.232, 95% CI, 1.159 to 4.385, P=0.018) and sleep medication use during the pandemic (OR 5.933, 95% CI, 2.142 to 20.030, P=0.002). Daytime symptoms were significantly associated with an impact on mental health (OR 2.332, 95% CI, 1.741 to 3.145, P<0.001) and sleep medication use during the pandemic (OR 3.450, 95% CI, 1.425 to 9.285, P=0.009). Abnormal behaviours in sleep were significantly associated with mental health impact (OR 2.198, 95% CI, 1.522 to 3.212, P<0.001) and reported disability (OR 1.504, 95% CI, 1.002 to 2.253, P=0.048). Sleep-disordered breathing was significantly associated with ongoing COVID-19 symptoms (OR 3.405, 95% CI, 1.610 to 7.450, P=0.002) and an impact on mental health (OR 1.725, 95% CI, 1.232 to 2.430, P=0.002). Restless legs symptoms were significantly associated with ongoing COVID-19 symptoms (OR 2.530, 95% CI, 1.143 to 5.388, P=0.024), an impact on mental health (OR 1.933, 95% CI, 1.318 to 2.862, P=0.001) and reported disability (OR 1.566, 95% CI, 1.029 to 2.364, P=0.034). Sleep phase disturbance was significantly associated with an impact on mental health (OR 1.669, 95% CI, 1.254 to 2.212, P<0.001), increased alcohol intake (OR 1.510, 95% CI, 1.080 to 2.121, P=0.017) and sleep medication use during the pandemic (OR 2.553, 95% CI, 1.084 to 6.713, P=0.041) (Figure 3).
Discussion
Summary of main findings
The pandemic lockdown had a major impact on the population. The majority of respondents described an altered sleep pattern and almost half of the studied population felt sleepier than prior to the lockdown. Reported problems included dozing off unintentionally in the day, disrupted sleep, difficulties falling/staying asleep and later bedtimes. Over a quarter of all respondents reported an increased alcohol intake during the lockdown. Furthermore, those self-isolating reported more insomnia/disrupted sleep, daytime symptoms, abnormal behaviours in sleep and symptoms of restless legs compared to those not self-isolating. Keyworkers reported fewer sleep alterations than others. In those with suspected COVID-19, nightmares and abnormal sleep rhythm were more common. A reported impact on mental health was most strongly associated with more difficulties falling asleep, sleep disruption, nightmares and daytime sleepiness. A change in sleep pattern was associated with medication use, ongoing COVID-19 symptoms and mental health impact, and it was negatively associated with age, male gender and general health.
Assessing lockdown impact on sleep and clinical significance of findings
Home confinement and isolation procedures have required modifications in lifestyle, leading to the loss of daily routines and habits. Resultant issues such as work, family and financial problems, limited exposure to natural light, and restricted opportunities to exercise may have negative effects on sleep (5). Insufficient sunlight, physical inactivity, dietary changes, weight gain, increased digital screen time, as well as the risk of alcohol use and developing addictions, are associated with prolonged lockdown (12). Additionally, sleep deprivation may lead to immunological alterations and it would therefore seem pertinent to consider the effects of altered sleep on COVID-19 susceptibility (13). Notably, almost a third of participants in our study reported sleeping less than 6 hours, and these were of older age, had higher BMI, reported more vulnerability/disability and mental health impact during the pandemic compared to respondents getting more than 6 hours of sleep.
Reported change in sleep pattern and specific sleep symptoms
Less than half of our study participants were feeling refreshed or felt that they were sleeping enough during the pandemic, and almost half of them felt sleepier than before the lockdown (Table S3). The most commonly reported specific sleep symptoms included disrupted sleep, difficulty falling and staying asleep, later bedtimes and falling asleep unintentionally in the day (Table 2). A majority of respondents reported a change in sleep pattern, and this was predicted by the presence of ongoing COVID-19 symptoms, mental health impact, sleep medication use in the pandemic, younger age and worse general health rating (Table S10). Similarly, a recent Italian survey including young adults demonstrated a significantly delayed bed and rise times during lockdown, spending overall more time in bed, but reporting a poor sleep quality (8). Also, an increased prevalence of insomnia during the outbreak was found in a Chinese survey-based study (9). Of note, a UK survey showed that half of the respondents experienced more disturbed sleep than usual; 39% of the participants reported sleeping fewer hours than before the lockdown; and 29% reported sleeping longer hours but still felt less rested (14). Consistent with our findings, only a minority of respondents reported improved sleep during the lockdown (14).
Effect of alcohol
Although frequently used by people who suffer from insomnia to try and help sleep (15), alcohol actually disrupts sleep architecture, possibly exacerbates sleep pathologies (such as sleep-disordered breathing and NREM parasomnias), and worsens daytime sleepiness (16). Almost 26% of our study population reported increased alcohol intake during the lockdown (Table S2). This is in line with previous evidence showing that, despite the decrease in social gathering, only a third of drinkers have reduced or stopped their alcohol intake, while 21% drink more frequently (17). Stress further contributes to alcohol seeking behaviour (18), and alcohol is also used to alleviate negative emotions (19). In our study, the presence of a sleep phase disturbance was predicted by an increased alcohol intake, reported mental health impact and sleep medication use during the pandemic (Figure 3).
Impact on mental health
The potential effect of mood alterations on sleep is relevant in the pandemic. The link between sleep and mood disturbances has previously been demonstrated in circumstances similar to the current lockdown (4,5). Confinement and quarantine have negative psychological consequences (4,5), which can have an impact on sleep. Worsened insomnia symptoms and psychological reactions attributed to the outbreak were demonstrated in the general population using online questionnaires reflecting the situation prior to and following the lockdown (9). Insomnia was associated with being female, mental illness, increased severity of anxiety and depression, COVID-19-related stress and prolonged time in bed (9). In a Chinese study the prevalence of insomnia was at 26%, and over 20% of patients with pre-existing mental health conditions reported a deterioration of these (11). Stronger changes in sleep quality in those with higher scores of depression, anxiety and stress were reported as well. Additionally, a longitudinal study investigating whether sleep quality depends on the social situation found an association between social isolation and poor sleep quality in older adults (20). Of note, over 65% of the population involved in our study reported an impact of the pandemic or lockdown on their mental health (Table S2), and this cohort reported more sleep-related symptoms and concerns about their sleep (Table 3). Importantly, an impact on mental health was one of the significant predictors for a reported change in sleep pattern (Table S10). On regression analysis, reported mental health impact was significantly associated with difficulties falling asleep, disrupted sleep, daytime sleepiness and nightmares (Figure 2).
Self-isolation status, keyworkers and sleep
Compared to those who did not self-isolate, self-isolators more frequently reported disrupted sleep, nightmares, abnormal movements, restless legs, falling asleep unintentionally and abnormal sleep rhythm (Table S6). Higher rates of difficulties to fall asleep or early awakenings in medically isolated versus self- or non-isolated individuals in the outbreak have been previously demonstrated (7).
A subgroup of the population that deserves special attention in the current circumstances are keyworkers. Healthcare workers account for a proportion of these; they have not only suffered the stress of being directly involved in the care of patients, but also faced the consequences of working long hours during the pandemic. Sleep disruption, poor quality of sleep, insomnia, and mood disturbances have all been reported in healthcare workers under pandemic circumstances (21-23). Although our study did not find an association between sleep disturbances and keyworker status, notably this group also included non-healthcare workers (postal workers, truck driver, police officer and others) (Table S7). Possibly keyworkers, considered as a wider group, experienced fewer changes in their routines during the lockdown, as they were allowed to leave the house to continue working.
Suspected COVID-19
There may be additional factors affecting sleep directly when considering individuals who suspect they have had COVID-19. Pre-existing sleep disorders, such as obstructive sleep apnoea, may also be present in COVID-19 patients (24), and might further impact on sleep quality and shortness of breath following the infection. In our study, the presence of symptoms associated with sleep-disordered breathing was significantly associated with ongoing COVID-19 symptoms (Figure 3).
Furthermore, potential neuropathogenic mechanisms of SARS-CoV-2 and a range of neurological symptoms have been reported in patients with COVID-19 (25). It could be hypothesised that involvement of structures controlling sleep-wake cycles in the central nervous system play a role in the presence of sleep-related symptoms in COVID-19 patients. We found a significant association between the presence of ongoing COVID-19 symptoms and insomnia/disrupted sleep (Figure 3). Moreover, participants with suspected COVID-19 had a higher frequency of nightmares and abnormal sleep rhythm (Table S9). Additionally, our regression analysis showed that restless legs symptoms were significantly associated with ongoing COVID-19 symptoms, mental health impact, and disability (Figure 3).
Limitations to the study
A selection bias may be expected in the study, as subjects with affected sleep may have been more interested in completing the survey. Also, as the survey was widely distributed among the population, this could have included subjects living in different countries, with different levels of lockdown measures. Additionally, the self-reported status with regards to having suspected COVID-19 was subjective and affirmatory responses did not require positive formal testing. Moreover, no formal objective assessments of specific sleep disorders were undertaken. Finally, there was limited diversity in the respondents’ ethnicity which did not permit assessing the impact of the pandemic on sleep in ethnic minorities.
Despite these limitations, and although it is clear that public health measures are required in such unprecedented circumstances, the potential effects of confinement and isolation on sleep should be screened for, and support needs to be provided for adequate management of sleep disturbances (6). Early identification of sleep disturbances and associated impact on mental health is crucial for timely intervention and support (26,27). Future studies could include formal assessments of subjective and objective measurements of sleep disorders, with more specific focus on subgroups according to infection, keyworker status and ethnicity.
Conclusions
Altered sleep patterns and specific sleep-related symptoms are common in the general population during the pandemic lockdown. These are mainly associated with mental health impact, self-isolation, suspected COVID-19 infection and ongoing symptoms. The potential consequences of the lockdown on sleep should not be overlooked, as they can have an impact on the future wellbeing of society. Sleep issues may be addressed early with appropriate guidance and/or counselling to avoid the longer-term impact of these on a public health scale.
Supplementary
Methods
Study design
Following internal discussions in the research committee, the BSS undertook a prospective, cross-sectional, survey-based study that was conducted from May 12th, 2020 to June 2nd, 2020, available to the general public. The survey was advertised via the BSS webpage, social media, NHS hospital trusts and specialist society and patient charities networks. The survey was submitted to the UKRI/MRC and the NHS Health Research Authority (HRA) ‘is it research?’ tool, confirming that ethical approval was not required (http://www.hra-decisiontools.org.uk/research/result7.html; outcome of assessment in https://cdn.amegroups.cn/static/application/04712039f3b0c07e77f3c779c14d1a17/JTD-CUS-2020-015-1.pdf; 09/05/2020). It was approved internally by the BSS Research and Executive Committees (11/05/2020; BSS-NEDS-SURVEY_V1-0; Appendix 2) and distributed via Google Forms on an open domain on May 12th, 2020. Data collection stopped on June 2nd, 2020. Eligible participants were adults aged 18 years, or older, living through the 2020 COVID-19 pandemic and under national lockdown regulations. Due to the nature of the survey, subjects required literacy and access to the Internet. Participants were excluded if they were not living under national lockdown regulations or were underage.
Questionnaire items
Participants were asked to read an introduction preceding the questionnaire items outlining the nature and purpose of the survey and the interested bodies (BSS). Following an item reduction phases involving specialists from all participating affiliations, respondents were asked to answer multiple choice questions, including “yes”, “no”, “don’t know” and context- specific answers, or provide a free-text response to “other, please specify”.
The following demographics were collected:
❖ Age (years);
❖ Gender (male/female/X/prefer not to say/other);
❖ Ethnicity (White/Black/Asian/other);
❖ Height (m);
❖ Weight (kg);
❖ Body-mass-index (BMI), calculated by weight (kg)/height (m2);
❖ Keyworker status (yes/no);
❖ Disability (yes/no/don’t know);
❖ Need to shield due to vulnerability status (yes/no/don’t know).
Specific questions to assess the impact of the pandemic/lockdown related to the following:
❖ Suspected COVID-19 history [yes/no/maybe (possibly, but they had no test)];
❖ Ongoing COVID-19 symptoms (tick all that apply: no/dry cough/sore throat/headache/fever/breathlessness/shortness of breath/chest pain/muscle pain/leg pain/loss of smell/skin rash/other);
❖ Self-isolation history in household (yes/no);
❖ Impact of the pandemic on mental health (yes/no/don’t know);
❖ Currently feeling refreshed from sleep (yes/no/undecided);
❖ Current feeling of sleeping enough (yes/no);
❖ Current sleep quantity (tick all that apply: less than 4 hours/4–5 hours/5–6 hours/6–7 hours/7–8 hours/8–9 hours/9–10 hours/10–11 hours/11–12 hours/more than 12 hours);
❖ Change in sleep pattern during the pandemic (yes/no);
❖ Specific sleep symptoms (tick all that apply: difficulties falling asleep/difficulties staying asleep/excessive sleepiness/disrupted sleep/nightmares/sleep paralysis (unable to move when waking)/sleepwalking/talking in your sleep/eating while asleep/abnormal movements (e.g., limbs or trunk)/breathlessness/shortness of breath during the night/breath holding/choking/gasping at night/night sweats/morning headache/restless legs/periodic limb movements/naps required by daytime/falls/injuries/falling asleep unintentionally/cramps/abnormal sleep rhythm (advanced/delayed)/going to bed earlier/going to bed later/getting up earlier/getting up later/other);
❖ Daytime symptoms of fatigue and/or sleepiness compared to before the lockdown (yes/no/undecided);
❖ Concern regarding sleep disturbance [rating on a scale from ‘0’ (worst) to ‘10’ (best)];
❖ Increased alcohol intake (yes/no/prefer not to say);
❖ Increased sleep medication use before and during the lockdown (yes/no/prefer not to say);
❖ Current rate of general health [scale from ‘0’ (worst) to ‘10’ (best)].
❖ Overweight was defined as BMI of 25–29.9 kg/m2 and obesity was defined as BMI ≥30 kg/m2.
A total of 24 different sleep symptoms were grouped into six sleep symptom categories for analysis (Table S1):
- Insomnia/disrupted sleep;
- Daytime symptoms;
- Abnormal behaviours in sleep;
- Sleep-disordered breathing;
- Restless legs;
- Sleep phase disturbance.
Online links were provided for official guidance from the BSS regarding sleep and from the UK government regarding COVID-19. Participants were advised to contact the helplines provided (111/999) if they required urgent or emergent medical care
Primary and secondary outcomes
The primary outcomes were to identify sleep disturbances and specific sleep symptoms related to the COVID-19 pandemic lockdown in the general population. Secondary outcomes included subgroup analyses for pre-defined cohorts within the population: suspected COVID-19, self-isolation, keyworkers and those reporting an impact on mental health.
Statistical analysis
Continuous variables including demographics and scale ratings were presented as median [interquartile range, IQR], due to non-normally distributed data and Mann-Whitney U analysis was performed for comparison. Chi-squared analysis was performed to compare categorical variables. Group comparisons were made based on suspected COVID-19 vs non- infected, self-isolation, keyworker status and impact on mental health due to the COVID-19 pandemic.
A multiple logistic regression model including ‘change in sleep pattern’ as the categorical outcome variable with ‘age’, ‘male gender’, ‘BMI’, ‘suspected COVID-19’, ‘ongoing COVID-19 symptoms’, ‘self-isolation’, ‘impact on mental health’, ‘shielding due to vulnerability’, ‘alcohol use’, ‘global general health rating’, ‘keyworking’, ‘disability’, ‘sleep medication before’ and ‘sleep medication during’, as predictor variables, was performed.
These variables were chosen due to their nature as either baseline demographics or pandemic- related factors.
A main-effects multiple logistic regression model was used to identify the strength of association and odds ratios with 95% CI between the six different symptom categories (insomnia/disrupted sleep, daytime symptoms, abnormal behaviours in sleep, sleep-disordered breathing, restless legs and sleep phase disturbance) and factors relating to the COVID-19 pandemic. Where significant associations were found, further multiple logistic regression modelling was performed and reported as odds ratios between sleep symptoms and factors relating to the COVID-19 pandemic.
‘Yes’ and ‘maybe’ responses regarding COVID-19 infection were grouped into ‘suspected COVID-19’ for analysis due to the limited availability of polymerase chain reaction (PCR) swab testing to the general public and the small sample size of confirmed infected participants. “Don’t know” answers were not included in the modelling as they were non- binary.
Data were collected on Google Forms (accessed online May 2020, Google Ireland Limited, Dublin, Ireland) and exported using MS Excel (Microsoft, Version 2004, Seattle, WA, USA). Data were then imported into IBM SPSS version 24 (IBM Corporation, New York, USA) and Prism version 8 (GraphPad Software, San Diego, California, USA) for further analysis. A P value of ≤0.05 was considered statistically significant. Missing data were not included in the analysis based on case-by-case decision.
Acknowledgments
The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health.
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editor (Joerg Steier) for the series “5th Clinical Update Sleep” published in Journal of Thoracic Disease. The article has been reviewed by the British Sleep Society (BSS) and nation-wide sleep centres (UK), and then was sent for in-house review.
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at http://dx.doi.org/10.21037/jtd-cus-2020-015
Data Sharing Statement: Available at http://dx.doi.org/10.21037/jtd-cus-2020-015
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jtd-cus-2020-015). The series “5th Clinical Update Sleep” was commissioned by the editorial office without any funding or sponsorship. JS served as the unpaid Guest Editor of the series and serves as an unpaid editorial board member of Journal of Thoracic Disease. JS’s contribution was partially supported by the National Institute for Health Research (NIHR) Biomedical Research Centre based at Guy’s and St Thomas’ NHS Foundation Trust and King’s College London. JE reports other from Sleep Research and Consulting Limited, grants from NHS Health Education England, grants from NHS Wales, grants from NHS Education Scotland, grants from Irish Rugby Football Union, grants from Royal College of Surgeons in Ireland, grants from Sleep Council, grants from Mayborn, grants from Public Health England, and grants from Third City, outside the submitted work. The other authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The BSS Research and Executive Committee approved this prospective, cross-sectional, survey-based study. The UKRI/MRC and the NHS Health Research Authority (HRA) confirmed that ethical approval was not required owing to the screening survey design (https://cdn.amegroups.cn/static/application/04712039f3b0c07e77f3c779c14d1a17/JTD-CUS-2020-015-1.pdf). The study conformed to the provisions of the Declaration of Helsinki (as revised in 2003).
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Available online: (accessed June 7, 2020).https://www.who.int/news-room/detail/27-04-2020-who-timeline---covid-19
- Chan JF, Yuan S, Kok KH, et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. Lancet 2020;395:514-23. [Crossref] [PubMed]
- Wilder-Smith A, Freedman DO. Isolation, quarantine, social distancing and community containment: pivotal role for old-style public health measures in the novel coronavirus (2019-nCoV) outbreak. J Travel Med 2020;27:taaa020.
- Jeong H, Yim HW, Song YJ, et al. Mental health status of people isolated due to Middle East Respiratory Syndrome. Epidemiol Health 2016;38:e2016048. [Crossref] [PubMed]
- Brooks SK, Webster RK, Smith LE, et al. The psychological impact of quarantine and how to reduce it: rapid review of the evidence. Lancet 2020;395:912-20. [Crossref] [PubMed]
- Altena E, Baglioni C, Espie CA, et al. Dealing with sleep problems during home confinement due to the COVID-19 outbreak: Practical recommendations from a task force of the European CBT-I Academy. J Sleep Res 2020. [Crossref] [PubMed]
- Xue Z, Lin L, Zhang S, et al. Sleep problems and medical isolation during the SARS-CoV-2 outbreak. Sleep Med 2020;70:112-5. [Crossref] [PubMed]
- Cellini N, Canale N, Mioni G, et al. Changes in sleep pattern, sense of time and digital media use during COVID-19 lockdown in Italy. J Sleep Res 2020. [Crossref] [PubMed]
- Li Y, Qin Q, Sun Q, et al. Insomnia and psychological reactions during the COVID-19 outbreak in China. J Clin Sleep Med 2020;16:1417-8. [Crossref] [PubMed]
- Huang Y, Zhao N. Generalized anxiety disorder, depressive symptoms and sleep quality during COVID-19 outbreak in China: a web-based cross-sectional survey. Psychiatry Res 2020;288:112954. [Crossref] [PubMed]
- Zhou J, Liu L, Xue P, et al. Mental Health Response to the COVID-19 Outbreak in China. Am J Psychiatry 2020;177:574-5. [Crossref] [PubMed]
- Lippi G, Henry BM, Bovo C, et al. Health risks and potential remedies during prolonged lockdowns for coronavirus disease 2019 (COVID-19). Diagnosis (Berl) 2020;7:85-90. [Crossref] [PubMed]
- Belingheri M, Paladino ME, Riva MA. Working schedule, sleep quality and susceptibility to COVID-19 in healthcare workers. Clin Infect Dis 2020. [Crossref] [PubMed]
- Available online: (accessed June 4, 2020).https://www.kcl.ac.uk/news/how-the-uk-is-sleeping-under-lockdown
- Ancoli-Israel S, Roth T. Characteristics of insomnia in the United States: results of the 1991 National Sleep Foundation Survey. I. Sleep 1999;22 Suppl 2:S347-53. [PubMed]
- Roehrs T, Roth T. Sleep, sleepiness, sleep disorders and alcohol use and abuse. Sleep Med Rev 2001;5:287-97. [Crossref] [PubMed]
- Alcohol Change UK. Available online: (accessed May 24, 2020).https://alcoholchange.org.uk/blog/2020/covid19-drinking-during-lockdown-headline-findings
- Ramchandani VA, Stangl BL, Blaine SK, et al. Stress vulnerability and alcohol use and consequences: From human laboratory studies to clinical outcomes. Alcohol 2018;72:75-88. [Crossref] [PubMed]
- Cooper ML, Frone MR, Russell M, et al. Drinking to regulate positive and negative emotions: a motivational model of alcohol use. J Pers Soc Psychol 1995;69:990-1005. [Crossref] [PubMed]
- Yu B, Steptoe A, Niu K, et al. Prospective associations of social isolation and loneliness with poor sleep quality in older adults. Qual Life Res 2018;27:683-91. [Crossref] [PubMed]
- Lai J, Ma S, Wang Y, et al. Factors Associated With Mental Health Outcomes Among Health Care Workers Exposed to Coronavirus Disease 2019. JAMA Netw Open 2020;3:e203976. [Crossref] [PubMed]
- Li X, Yu H, Bian G, et al. Prevalence, risk factors, and clinical correlates of insomnia in volunteer and at home medical staff during the COVID-19. Brain Behav Immun 2020;87:140-1. [Crossref] [PubMed]
- Zhang C, Yang L, Liu S, et al. Survey of Insomnia and Related Social Psychological Factors Among Medical Staff Involved in the 2019 Novel Coronavirus Disease Outbreak. Front Psychiatry 2020;11:306. [Crossref] [PubMed]
- McSharry D, Malhotra A. Potential influences of obstructive sleep apnea and obesity on COVID-19 severity. J Clin Sleep Med 2020;16:1645. [Crossref] [PubMed]
- Zubair AS, McAlpine LS, Gardin T, et al. Neuropathogenesis and Neurologic Manifestations of the Coronaviruses in the Age of Coronavirus Disease 2019: A Review. JAMA Neurol 2020;77:1018-27. [Crossref] [PubMed]
- Goodman-Casanova JM, Dura-Perez E, Guzman-Parra J, et al. Telehealth Home Support During COVID-19 Confinement for Community-Dwelling Older Adults With Mild Cognitive Impairment or Mild Dementia: Survey Study. J Med Internet Res 2020;22:e19434. [Crossref] [PubMed]
- Sher L. COVID-19, anxiety, sleep disturbances and suicide. Sleep Med 2020;70:124. [Crossref] [PubMed]


