
Characteristics of symptoms among outpatients following the discontinuation of the dynamic zero-COVID-19 policy in China: insights from an online nationwide cross-sectional survey in 2022
Highlight box
Key findings
• Fever, fatigue, and expectoration were the most frequently reported symptoms, which differed from the data from other countries.
• The most common severe symptoms were myalgia, fatigue, and sore throat based on the numerical rating scale, and these symptoms tended to be milder as age increased.
What is known and what is new?
• The clinical symptoms of patients with coronavirus disease 2019 (COVID-19) vary with geographical location and underlying diseases.
• The characteristics of symptom incidence and severity of non-hospitalized COVID-19 patients were different between China and other countries.
What is the implication, and what should change now?
• Regional symptom data possess inherent distinctiveness and play a pivotal role in formulating individualized strategies for managing COVID-19.
Introduction
It has been more than three years since the outbreak of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, which was later named coronavirus disease 2019 (COVID-19) by the World Health Organization (WHO). There have been 770,437,327 cases confirmed worldwide, including 6,956,900 deaths as of September 2023 (1). Although WHO announced the end of the emergency phase of COVID-19 in May 2023 and the current incidence rate is declining, more than 15,000 people are still becoming infected every week (1). During this period, characteristics including the symptoms of infected individuals were highly variable due to a variety of factors, including differences in population level immunity, evolving variant strains, antiviral therapeutics, vaccines, and geographic location (2-4). Based on research data from infected patients reported by health workers around the world, WHO continues to update guidelines on COVID-19 surveillance strategies and therapeutics, which is a major contribution to suppressing transmission, providing optimal care for all patients, and saving lives (1).
However, to our knowledge, previous data on symptoms have mostly focused on hospitalized COVID-19 patients, despite the fact that most infected individuals do not require hospitalization (5,6). Fully elucidating the symptoms of COVID-19 can provide crucial information for early recognition, timely diagnosis, treatment evaluation, and prediction of outcomes (7,8). On the contrary, failure to reflect this symptom variation in public health messaging may contribute to COVID-19 spread and put the community at a greater risk of infection (4). Moreover, patients with COVID-19 can be concurrently infected with a broad range of community-acquired pathogens, encompassing respiratory viruses, bacteria, and other less prevalent microorganisms; investigating the symptoms induced by diverse pathogens is advantageous for prompt differential diagnosis and empirical treatment (9-12).
Until December 2022, China had adopted a dynamic zero-COVID-19 policy, which deployed mass testing and strict quarantine measures to stamp out any outbreak of COVID-19 before it could spread (13). This policy led to significantly fewer infections and deaths in comparison to other countries that had embraced a strategy of coexistence with SARS-CoV-2 (13). Following the end of the dynamic zero-COVID-19 policy, the number of infected patients in China rose dramatically (14). In the case that more than 90% of the population had been vaccinated, it was estimated that nearly 80% of the population was infected by the Omicron variant and the vast majority of them were infected for the first time (14). We speculate that the symptoms in this population have some unique characteristics and are worthy of exploration. Therefore, we conducted a nationwide anonymous online survey questionnaire on the symptom characteristics of outpatients infected with SARS-CoV-2 in China immediately after the termination of the dynamic zero-COVID-19 policy. We aimed to provide the symptomatic data of COVID-19 cases in this region for the formulation of local and global epidemic surveillance policies. We present this article in accordance with the SURGE reporting checklist (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1244/rc).
Methods
Questionnaire design
A total of 24 questions regarding demographic information, duration and severity of main symptoms, and vaccination status were included in the questionnaire. The symptoms included were based on previous epidemiological research and consultations with experts (6,15). The severity of symptoms was assessed by a numerical rating scale (NRS), which had been validated in evaluation of symptoms in a previous study (16). The NRS was an 11-point scale scored from 0 to 10, with 0 indicating no such symptom, 1 indicating very mild, and 10 indicating intolerable severity. Scores of 1–4, 5–7, and 8–10 referred to mild, moderate, and severe, respectively. Private participant information such as name and home address were avoided in the questionnaire.
Conduct of the survey
In early December 2022, mainland China completely ceased the zero-Covid-19 policy. Based on previous literature, the incubation period of COVID-19 was about 3–7 days, and symptoms were likely to appear 2–14 days after exposure to the virus (15), so we speculated that the first large-scale infections would occur between late December 2022 and early January 2023. The data from the Chinese Center for Disease Control and Prevention revealed that the COVID-19 testing positivity rate reached its peak at 29.2% on 25 December 2022, followed by fluctuations and subsequent decline, thereby validating our hypothesis (17). Therefore, the questionnaire was disseminated through WeChat in this period. The target participants were adult patients infected with SARS-CoV-2 in an outpatient setting. Owing to its user-friendliness, adaptability, and swift data collection capabilities, we had opted for the convenience sampling method. This methodology enabled us to quickly gather data on the initial cohort of patients infected post the cessation of the dynamic zero-COVID policy, thus mitigating the risk of outcome bias that could result from the admixture with other pre-existing infected populations due to extended delays.
Statistical analysis
Data analysis was conducted from late February to April 2023. Median [interquartile range (IQR)] was reported for continuous variables that did not follow normal distribution. Categorical data were presented as number and percentage. For the comparison of two groups, we used the Mann-Whitney U test for continuous variables and the chi-squared test for categorical variables. All P values were calculated as two-sided, with statistical significance set at P<0.05. All data were collected with Microsoft Excel (Microsoft, Redmond, WA, USA) and analyzed by SPSS 26.0 (IBM Corp., Armonk, NY, USA).
Ethical statement
This study was conducted in accordance with the Declaration of Helsinki (as revised in 2013) and approved by the Medical Ethics Committee of the First Affiliated Hospital of Air Force Medical University (also known as Xijing Hospital) (approval No. KY20222118-F-1). Written informed consent was not obtained for anonymized patient information to be published in this article. Instead, at the outset of the questionnaire, patients were asked whether they agreed to have their anonymous information used for analysis and publication; only patients who agreed could proceed with the survey.
Results
Demographic characteristics of the participants
A total of 4,836 eligible questionnaires were collected and analyzed. Among the participants, 3,133 tested positive for COVID-19 nucleic acid or antigen, whereas 1,703 showed symptoms but remained untested. The median age of all participants was 40.0 (33.0, 48.0) years, and 65% (n=3,162) of them were female. Some (12.5%) of participants were smokers, with a higher prevalence among individuals who tested positive. A total of 4,542 (93.9%) participants had received two or three doses of the vaccine and only 209 (4.3%) patients were completely unvaccinated. The most common underlying diseases were allergic rhinitis (9.7%), hypertension (9.4%), and diabetes (3.3%) (Table 1).
Table 1
| Characteristics | All responders | Tested positive | Untested symptomatic | P value |
|---|---|---|---|---|
| Number | 4,836 | 3,133 (64.8) | 1,703 (35.2) | – |
| Age, years | 40.0 (33.0, 48.0) | 39.0 (33.0, 47.0) | 40.0 (33.0, 49.0) | <0.001 |
| Gender | 0.04 | |||
| Male | 1,674 (34.6) | 1,117 (35.7) | 557 (32.7) | |
| Female | 3,162 (65.4) | 2,016 (64.3) | 1,146 (67.3) | |
| BMI, kg/m2 | 22.9 (20.8, 25.0) | 22.9 (20.8, 25.2) | 22.7 (20.8, 24.8) | 0.34 |
| Smokers | 603 (12.5) | 421 (13.4) | 182 (10.7) | 0.006 |
| Underlying diseases | ||||
| Hypertension | 454 (9.4) | 295 (6.1) | 159 (9.3) | 0.93 |
| Diabetes | 161 (3.3) | 109 (3.5) | 52 (3.1) | 0.43 |
| Allergic rhinitis | 469 (9.7) | 318 (10.1) | 151 (8.9) | 0.15 |
| Vaccination | 0.85 | |||
| Unvaccinated | 209 (4.3) | 129 (4.1) | 80 (4.7) | |
| 1 dose | 85 (1.8) | 55 (1.8) | 30 (1.8) | |
| 2 doses | 751 (15.5) | 484 (15.4) | 267 (15.6) | |
| 3 doses | 3,791 (78.4) | 2,465 (78.7) | 1,326 (77.9) |
Data are presented as count (percentage) for binary characteristics and as median (interquartile range) for continuous characteristics. The statistical comparison was conducted between respondents who tested positive for SARS-CoV-2 and presented with symptoms but were not tested. Tested positive indicates that the respondents have tested positive for either the COVID-19 nucleic acid or antigen. Untested symptomatic refers to respondents who exhibit symptoms but have not undergone COVID-19 testing. BMI, body mass index; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
Symptom occurrence
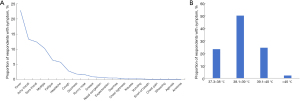
For respondents who tested positive, the most frequently reported symptoms were fever (n=2,941, 93.9%), fatigue (n=2,871, 91.6%), expectoration (n=2,847, 90.8%), and myalgia (n=2,712, 86.5%). The observed pattern was consistent among untested symptomatic individuals (Figure 1). Fever (22.8%) was the most common initial symptom, followed by itchy throat (13.3%), sore throat (12.4%), and myalgia (10.3%) (Figure 2A). The maximum temperature of 38.1–39 ℃ was observed in approximately half of all fever individuals, whereas only a mere 2.1% experienced temperatures above 40 ℃ (Figure 2B).

Symptom variations based on basic characteristics
The incidence of certain systemic symptoms, including headache (87.1% vs. 80.9%, P<0.001), dizziness (79% vs. 71.5%, P<0.001), nausea (49.9% vs. 39.7%, P<0.001), diarrhea (46.8% vs. 42.5%, P=0.02), and vomiting (36% vs. 25.1%, P<0.001), and nearly all respiratory symptoms were significantly higher in males compared to females, with this trend also observed when comparing nonsmokers to smokers. The incidence of almost all symptoms in vaccinated patients was higher than that in completely unvaccinated patients, and the difference in some symptoms reached statistical significance, such as expectoration (91.2% vs. 82.9%, P=0.002), nasal congestion (88.1% vs. 66.7%, P<0.001), myalgia (86.8% vs. 80.6%, P=0.043), itchy throat (85.7% vs. 76.7%, P=0.005), sore throat (85.6% vs. 71.3%, P<0.001), runny nose (82.2% vs. 69%, P<0.001), cough (81.6% vs. 69.8%, P=0.001), sneeze (77.1% vs. 53.5%, P<0.001), and anosmia (59.9% vs. 48.1%, P=0.008). The incidence of symptoms also varied among patients with different underlying diseases. For instance, patients with asthma or chronic bronchitis exhibited significantly higher incidences of chest tightness (60.5% vs. 44.9%, P=0.04 and 60.3% vs. 44.7%, P=0.008), wheezing (60.5% vs. 43%, P=0.02 and 61.6% vs. 42.8%, P=0.001), and shortness of breath (67.4% vs. 49%, P=0.01 and 63% vs. 48.9%, P=0.01) than those without either of these two diseases (Figure 3).

Symptom severity
Analysis of symptom severity according to different age groups revealed that almost all symptoms showed a trend of higher proportion of severe cases with younger age (Figure 4A). For all respondents who tested positive, myalgia (35.3%), fatigue (31.5%), and sore throat (30.9%) were the symptoms with the highest incidence of severe occurrence. This varied between age groups (Figure 4B). Among those aged over 60 years (35.9%) and 18–29 years (49.8%), severe fatigue was most prevalent, whereas among those aged 30–39 (48.2%) and 40–49 years (45%), severe myalgia was most common. Diarrhea (4.9%), wheezing (4.2%), chest tightness (4.0%), vomiting (3.6%), and chest pain (2.5%) were the symptoms with the lowest incidence of severe symptoms, although the overall incidence of each symptom was approximately 30–40% (Figure 4A). Results from the multivariate regression analysis for the occurrence of severe symptoms are presented in Table 2. Age ≥60 years was negatively correlated with the incidence of severe manifestations for all symptoms except sneezing. On the contrary, a positive association was found between smoking and the incidence of severe symptoms for almost all conditions. Asthma was associated with a greater likelihood of experiencing severe chest tightness [odds ratio (OR): 2.41, 95% confidence interval (CI): 1.01–5.80], shortness of breath (OR: 2.42, 95% CI: 1.06–5.52), diarrhea (OR: 4.19, 95% CI: 1.94–9.07), and vomiting (OR: 3.35, 95% CI: 1.37–8.21). Respondents with chronic bronchitis showed significantly higher risk of severe expectoration (OR: 1.78, 95% CI: 1.08–2.91) and wheezing (OR: 2.61, 95% CI: 1.26–5.41).

Table 2
| Symptoms | Variables | OR (95% CI) | P value |
|---|---|---|---|
| Fatigue | Age ≥60 years | 0.97 (0.96–0.98) | <0.001 |
| Smoking | 1.48 (1.18–1.86) | <0.001 | |
| Expectoration | Age ≥60 years | 0.96 (0.95–0.97) | <0.001 |
| Smoking | 2.67 (2.03–3.52) | <0.001 | |
| Chronic bronchitis | 1.78 (1.08–2.91) | 0.02 | |
| Nasal congestion | Age ≥60 years | 0.96 (0.95–0.97) | <0.001 |
| Smoking | 1.88 (1.44–2.45) | <0.001 | |
| Vaccination | 1.36 (1.20–1.54) | <0.001 | |
| Myalgia | Age ≥60 years | 0.97 (0.96–0.98) | <0.001 |
| Itchy throat | Age ≥60 years | 0.98 (0.96–0.99) | <0.001 |
| Male | 0.77 (0.63–0.95) | 0.01 | |
| Smoking | 2.10 (1.61–2.75) | <0.001 | |
| Coronary disease | 2.10 (1.11–3.97) | 0.02 | |
| Vaccination | 1.15 (1.02–1.29) | 0.01 | |
| Sore throat | Age ≥60 years | 0.98 (0.97–0.99) | <0.001 |
| Male | 0.80 (0.66–0.98) | 0.03 | |
| Smoking | 2.41 (1.86–3.12) | <0.001 | |
| Vaccination | 1.17 (1.05–1.30) | 0.005 | |
| Headache | Age ≥60 years | 0.96 (0.95–0.97) | <0.001 |
| Smoking | 1.40 (1.10–1.78) | 0.005 | |
| Runny nose | Age ≥60 years | 0.97 (0.96–0.98) | <0.001 |
| Cough | Age ≥60 years | 0.96 (0.95–0.97) | <0.001 |
| Allergic rhinitis | 0.71 (0.53–0.95) | 0.02 | |
| Smoking | 2.84 (2.10–3.84) | <0.001 | |
| Vaccination | 1.15 (1.02–1.29) | 0.02 | |
| Sneeze | Smoking | 1.59 (1.04–2.43) | 0.03 |
| Coronary disease | 2.33 (1.01–5.34) | 0.04 | |
| Vaccination | 1.36 (1.10–1.70) | 0.005 | |
| Dizziness | Age ≥60 years | 0.95 (0.94–0.96) | <0.001 |
| Ageusia | Age ≥60 years | 0.97 (0.96–0.99) | <0.001 |
| Parosmia | Age ≥60 years | 0.96 (0.95–0.97) | <0.001 |
| Short of breath | Age ≥60 years | 0.96 (0.94–0.98) | <0.001 |
| Smoking | 2.06 (1.24–3.41) | 0.005 | |
| Coronary disease | 3.13 (1.27–7.69) | 0.01 | |
| Asthma | 2.42 (1.06–5.52) | 0.03 | |
| Chest tightness | Age ≥60 years | 0.96 (0.94–0.98) | <0.001 |
| Asthma | 2.41 (1.01–5.80) | 0.04 | |
| Diarrhea | Age ≥60 years | 0.98 (0.96–0.99) | 0.04 |
| Asthma | 4.19 (1.94–9.07) | <0.001 | |
| Nausea | Age ≥60 years | 0.97 (0.95–0.99) | 0.005 |
| Smoking | 1.93 (1.13–3.30) | 0.01 | |
| Wheezing | Age ≥60 years | 0.97 (0.95–0.99) | 0.01 |
| Chronic bronchitis | 2.61 (1.26–5.41) | 0.01 | |
| Chest pain | Age ≥60 years | 0.95 (0.93–0.97) | <0.001 |
| Coronary disease | 2.06 (1.24–3.41) | 0.02 | |
| Vomiting | Age ≥60 years | 0.97 (0.94–0.99) | 0.01 |
| Smoking | 2.67 (1.31–5.41) | 0.006 | |
| Asthma | 3.35 (1.37–8.21) | 0.008 |
CI, confidence interval; COVID-19, coronavirus disease 2019; OR, odds ratio.
Discussion
This study investigated symptoms of outpatients infected with SARS-CoV-2 nationwide after the termination of China’s dynamic zero-COVID-19 policy and assessed the severity of these symptoms for the first time. The findings indicated that, first, during the COVID-19 pandemic, there was a resemblance in clinical symptoms between patients who tested positive and symptomatic patients who were not tested. Second, the incidence of symptoms was related to the patient’s gender, vaccination status, and underlying diseases. Third, myalgia, fatigue, and sore throat were the symptoms with the highest incidence of severe occurrence. Fourth, age 60 years and above was negatively associated, whereas smoking was positively associated with the occurrence of severe symptoms across nearly all conditions. These discoveries provide crucial insights for the timely detection and targeted management of SARS-CoV-2 infection.
Our research findings indicated that fever, fatigue, expectoration, nasal congestion, and myalgia were the top five symptoms reported by COVID-19-positive patients, which slightly deviated from the results reported in another survey conducted in China (18). The latter study suggested that fatigue (73.31%), cough (70.02%), fever (65.25%), overall soreness (58.64%), and headache/dizziness (57.61%) were the most prevalent symptoms, with a lower incidence compared to our findings. This difference might arise from the fact that most of our respondents were located in Shaanxi Province, whereas their study population was predominantly situated in Hubei Province. Another provincial survey showed variations in symptoms among different city populations (19). Geographic variations in symptoms were also seen between countries (4,20). For instance, Mexico and Brazil had higher rates of itchy eyes and headaches, whereas India and Pakistan had higher rates of nasal congestion and muscle pain (4). Compared to patients outside of China, Chinese patients exhibited a higher prevalence of fever, cough, and dyspnea. Conversely, they demonstrated a significantly lower incidence of diarrhea (21). The potential factors contributing to this outcome could be attributed to variances in cultural contexts and perceptions of the disease across diverse nations. As with geographic location, the reported symptoms also varied with specific chronic diseases. For example, individuals without underlying chronic disease were less likely to report shortness of breath in comparison to those who had asthma (4). In our study, respondents with asthma or chronic bronchitis exhibited significantly higher incidences of chest tightness, wheezing, and shortness of breath than those without either of these diseases. The multivariate regression analysis revealed a greater propensity for severe expectoration and wheezing among the respondents with chronic bronchitis. These results might be partially attributed to the combined effects of asthma or chronic bronchitis, allergic sensitization, and associated airway inflammation, which could collectively contribute to the development of respiratory symptoms and ultimately result in a poorer prognosis (22,23). Additionally, our study indicated that vaccinated individuals experienced more upper respiratory tract symptoms than those who were unvaccinated. This finding aligned with prior survey in China indicating that vaccination might lead to a rise in upper respiratory tract symptoms such as sore throat, nasal congestion, runny nose, and expectoration (18). Meanwhile, the same prior survey indicated that vaccination alleviated symptoms such as taste and smell loss, aligning with another retrospective cohort study showing the protective effects of SARS-CoV-2 vaccination against these sequelae (24); however, this was contrary to our research findings. In a retrospective cohort study conducted in Mexico, individuals who received three vaccine doses had a higher likelihood of severe symptoms compared to the unvaccinated. However, no such association was observed between those receiving one or two vaccine doses and the unvaccinated (25). Interestingly, the two studies had conflicting conclusions on the booster shot, with the former suggesting it could reduce disease severity and shorten symptoms duration, whereas the latter suggested the opposite. The mechanisms behind these conflicting results remained unclear, and we speculated that they might be related to a combination of factors, including the type of vaccine received, the effect of vaccination on immune system reactivity, and the baseline demographic characteristics of the vaccinated population.
Our study revealed no statistically significant difference in symptom incidence between COVID-19-positive and the untested symptomatic respondents, except for itchy throat, shortness of breath, and chest tightness. This might indicate that the presence of these symptoms warranted a high suspicion of SARS-CoV-2 infection in the context of the pandemic. However, a previous review of 42 prospective studies involving over 50,000 individuals found that a single symptom alone cannot constitute an accurate diagnosis of the disease (24). Individuals experiencing anosmia or ageusia were at a fivefold increased risk of being infected with SARS-CoV-2 compared to those without such symptoms. These two symptoms were reported by 59.3% and 63.1% of participants in our study, respectively. These symptoms, plus fever and cough, could serve as key indicators to screen patients for further COVID-19 testing (26). Meanwhile, the incidence of symptoms in COVID-19 patients could be influenced by various factors, including geographical location (4), comorbidities, age (27), and vaccination status (18), which undoubtedly increased the difficulty of diagnosing COVID-19 through symptoms. In this study, the incidence of most symptoms was found to be higher in males compared to females, in vaccinated individuals compared to unvaccinated individuals, and in nonsmokers compared to smokers, as well as being associated with the presence of underlying diseases. Consequently, we believe that integrating symptoms with additional factors, such as contact history, vaccination status, age, gender, and local epidemiological characteristics may enhance the accuracy of COVID-19 diagnosis.
The severity of symptoms directly impacted the patient’s quality of life and occupational status, thereby determining the treatment focus and strategy. The WHO recommended symptomatic treatment or urgent care depending on the patient’s symptoms and severity (1). In addition, among non-hospitalized COVID-19 patients, emerging evidence suggested that the severity and specific manifestations of initial symptoms could significantly impact daily functioning, such as activities of daily living (ADL), and exerted a profound influence on psychological well-being (28-30). The decline in quality of life might occur even if symptoms were not severe during the initial few days when symptoms were prevalent. Multiple-site pain played a crucial role in this decline (28). Similarly, the most frequently severe positive symptoms in our study were also pain-related symptoms, including myalgia and sore throat. However, almost all symptoms showed a trend of higher proportion of severe cases with younger age, and this was consistent with previous research (25). In addition, multivariate regression analysis suggested that respondents over 60 years old had a lower risk of experiencing severe symptoms. This could potentially be associated with alterations in the innate immune response observed in the elderly, coupled with an inability to elicit a proficient acquired immune response (31). Moreover, the clinical symptoms of COVID-19 in older adults was atypical, which might lead to delays in diagnosis and treatment, thereby aggravating the prognosis of patients (27,31). The reported rate of mild symptoms in elderly patients was around 80%, whereas approximately 14% experienced severe symptoms with shortness of breath and lung involvement (32), which was higher than the severe incidence of 4.4% in our study. Taken together, these findings suggested that, in addition to the high prevalence of severe symptoms among young patients, it is crucial to remain vigilant regarding the potential risks associated with atypical symptoms in elderly patients. Besides, smoking had detrimental effects on the immune system, particularly by disrupting the macrophage and cytokine response, thereby compromising the body’s capacity to manage and control infections (33). This partially explained our research findings, which indicated that smokers had a higher risk of experiencing severe symptoms for almost all conditions. Therefore, every smoker should be encouraged to quit, and provided with guidance, support, and pharmacotherapy, if accessible. Often, times of crisis can serve as a catalyst for motivating individuals to cease smoking (33).
Limitations
There are some limitations in this study. First, according to the epidemiological background, our respondents were predominantly infected with the Omicron variant. Given that new variants may exhibit distinct symptom variations, it is important to note that the findings of this study may not be directly applicable for assessing novel SARS-CoV-2 variants. Second, although the NRS is widely utilized in questionnaires to assess respondents’ feelings, opinions, and interests, its evaluation remains highly subjective and constrained by variations in patients’ perceptions of symptoms, which may ultimately compromise the reliability of the results. Third, the population distribution is uneven, with 72% of respondents originating from Shaanxi Province, potentially impacting the overall assessment of SARS-CoV-2 infections nationwide. Fourth, the observation time is too short to determine the long-term effects of symptoms on patients, such as long COVID-19 symptoms.
Conclusions
This study revealed that fever, fatigue, expectoration, nasal congestion, and myalgia were the most frequently reported symptoms by individuals infected with SARS-CoV-2 immediately after the discontinuation of the zero-COVID-19 policy in China. The incidence of these symptoms differs from that in other countries outside China, confirming our hypothesis that data on symptoms in China are unique and highlighting the importance of regional symptom studies. Myalgia, fatigue, and sore throat had the highest incidence of severe occurrence. Furthermore, the incidence of severe symptoms exhibited an age disparity, with younger individuals demonstrating a higher prevalence compared to older individuals. The aforementioned statement furnishes crucial insights for the formulation of COVID-19 management and treatment strategies.
Acknowledgments
We are thankful for the support from faculties and staff in the study sites.
Footnote
Reporting Checklist: The authors have completed the SURGE reporting checklist. Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1244/rc
Data Sharing Statement: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1244/dss
Peer Review File: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1244/prf
Funding: This work was supported by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1244/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. This study was conducted in accordance with the Declaration of Helsinki (as revised in 2013) and was approved by the Medical Ethics Committee of the First Affiliated Hospital of Air Force Medical University (also known as Xijing Hospital) (approval No. KY20222118-F-1). Written informed consent was not obtained for anonymized patient information to be published in this article. Instead, at the outset of the questionnaire, patients were asked whether they agreed to have their anonymous information used for analysis and publication. Only patients who agreed could proceed with the survey.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- World Health Organization. WHO Coronavirus (COVID-19) Dashboard. Available online: https://covid19.who.int/
- Piersiala K, Kakabas L, Bruckova A, et al. Acute odynophagia: A new symptom of COVID-19 during the SARS-CoV-2 Omicron variant wave in Sweden. J Intern Med 2022;292:154-61. [Crossref] [PubMed]
- El-Shabasy RM, Nayel MA, Taher MM, et al. Three waves changes, new variant strains, and vaccination effect against COVID-19 pandemic. Int J Biol Macromol 2022;204:161-8. [Crossref] [PubMed]
- Kadirvelu B, Burcea G, Quint JK, et al. Variation in global COVID-19 symptoms by geography and by chronic disease: A global survey using the COVID-19 Symptom Mapper. EClinicalMedicine 2022;45:101317. [Crossref] [PubMed]
- Lane A, Hunter K, Lee EL, et al. Clinical characteristics and symptom duration among outpatients with COVID-19. Am J Infect Control 2022;50:383-9. [Crossref] [PubMed]
- Yang W, Yang S, Wang L, et al. Clinical characteristics of 310 SARS-CoV-2 Omicron variant patients and comparison with Delta and Beta variant patients in China. Virol Sin 2022;37:704-15. [Crossref] [PubMed]
- Talukder A, Razu SR, Alif SM, et al. Association Between Symptoms and Severity of Disease in Hospitalised Novel Coronavirus (COVID-19) Patients: A Systematic Review and Meta-Analysis. J Multidiscip Healthc 2022;15:1101-10. [Crossref] [PubMed]
- Tavakolifard N, Moeini M, Haddadpoor A, et al. Clinical Symptoms of COVID-19 and Their Association with Disease Outcome. Adv Biomed Res 2022;11:2. [Crossref] [PubMed]
- Bai Y, Tao X. Comparison of COVID-19 and influenza characteristics. J Zhejiang Univ Sci B 2021;22:87-98. [Crossref] [PubMed]
- Chen L, Shen L, Wu W, et al. Co-infecting pathogens can contribute to inflammatory responses and severe symptoms in COVID-19. J Thorac Dis 2022;14:355-70. [Crossref] [PubMed]
- Lansbury L, Lim B, Baskaran V, et al. Co-infections in people with COVID-19: a systematic review and meta-analysis. J Infect 2020;81:266-75. [Crossref] [PubMed]
- Sreenath K, Batra P, Vinayaraj EV, et al. Coinfections with Other Respiratory Pathogens among Patients with COVID-19. Microbiol Spectr 2021;9:e0016321. [Crossref] [PubMed]
- Burki T. Dynamic zero COVID policy in the fight against COVID. Lancet Respir Med 2022;10:e58-9. [Crossref] [PubMed]
- He Y, Zhang F, Liu Y, et al. Clinical Characteristics of Mild Patients with Breakthrough Infection of Omicron Variant in China after Relaxing the Dynamic Zero COVID-19 Policy. Vaccines (Basel) 2023;11:968. [Crossref] [PubMed]
- CDC. COVID-19 and Your Health. Centers for Disease Control and Prevention. 2020. Available online: https://www.cdc.gov/coronavirus/2019-ncov/symptoms-testing/symptoms.html. Accessed 8 Apr 2024.
- Hollen PJ, Gralla RJ, Kris MG, et al. A comparison of visual analogue and numerical rating scale formats for the Lung Cancer Symptom Scale (LCSS): does format affect patient ratings of symptoms and quality of life? Qual Life Res 2005;14:837-47. [Crossref] [PubMed]
- Chinese Center for Disease Control and Prevention. Available online: https://en.chinacdc.cn/. Accessed 16 Apr 2024.
- Qin S, Li Y, Wang L, et al. Assessment of vaccinations and breakthrough infections after adjustment of the dynamic zero-COVID-19 strategy in China: an online survey. Emerg Microbes Infect 2023;12:2258232. [Crossref] [PubMed]
- Zhong BL, Zhou DY, He MF, et al. Mental health problems, needs, and service use among people living within and outside Wuhan during the COVID-19 epidemic in China. Ann Transl Med 2020;8:1392. [Crossref] [PubMed]
- Grant MC, Geoghegan L, Arbyn M, et al. The prevalence of symptoms in 24,410 adults infected by the novel coronavirus (SARS-CoV-2; COVID-19): A systematic review and meta-analysis of 148 studies from 9 countries. PLoS One 2020;15:e0234765. [Crossref] [PubMed]
- Ahmed A, Ali A, Hasan S. Comparison of Epidemiological Variations in COVID-19 Patients Inside and Outside of China-A Meta-Analysis. Front Public Health 2020;8:193. [Crossref] [PubMed]
- Zhu Z, Hasegawa K, Ma B, et al. Association of asthma and its genetic predisposition with the risk of severe COVID-19. J Allergy Clin Immunol 2020;146:327-329.e4. [Crossref] [PubMed]
- Awatade NT, Wark PAB, Chan ASL, et al. The Complex Association between COPD and COVID-19. J Clin Med 2023;12:3791. [Crossref] [PubMed]
- Omori T, Hanafusa M, Kondo N, et al. Specific sequelae symptoms of COVID-19 of Omicron variant in comparison with non-COVID-19 patients: a retrospective cohort study in Japan. J Thorac Dis 2024;16:3170-80. [Crossref] [PubMed]
- Mendoza-Cano O, Trujillo X, Ríos-Silva M, et al. Association between Vaccination Status for COVID-19 and the Risk of Severe Symptoms during the Endemic Phase of the Disease. Vaccines (Basel) 2023;11:1512. [Crossref] [PubMed]
- Struyf T, Deeks JJ, Dinnes J, et al. Signs and symptoms to determine if a patient presenting in primary care or hospital outpatient settings has COVID-19. Cochrane Database Syst Rev 2022;5:CD013665. [PubMed]
- Unim B, Palmieri L, Lo Noce C, et al. Prevalence of COVID-19-related symptoms by age group. Aging Clin Exp Res 2021;33:1145-7. [Crossref] [PubMed]
- Tütün Yümin E, Sürmeli M, Topcuoğlu C, et al. The Symptom Experience and Functioning of Non-Hospitalized COVID-19 Patients Within the First 20 days. Clin Nurs Res 2023;32:608-17. [Crossref] [PubMed]
- Mitropoulos A, Goodwill S, Copeland R, et al. The relationship between physical activity and severity of COVID-19 symptoms in non-hospitalized individuals. Eur J Public Health 2022;32:794-8. [Crossref] [PubMed]
- Seckman C. The impact of COVID-19 on the psychosocial well-being of older adults: A literature review. J Nurs Scholarsh 2023;55:97-111. [Crossref] [PubMed]
- Gadó K, Kovács AK, Domján G, et al. COVID-19 and the elderly. Physiol Int 2022; Epub ahead of print. [Crossref] [PubMed]
- Nanda A, Vura NVRK, Gravenstein S. COVID-19 in older adults. Aging Clin Exp Res 2020;32:1199-202. [Crossref] [PubMed]
- van Zyl-Smit RN, Richards G, Leone FT. Tobacco smoking and COVID-19 infection. Lancet Respir Med 2020;8:664-5. [Crossref] [PubMed]


