
Epidemiological trends and serological profiles of respiratory pathogens in Guangzhou: an 11-year retrospective study from 2013 to 2023
Highlight box
Key findings
• This 11-year retrospective study of 96,927 cases in Guangzhou revealed an 18.01% overall positive rate for respiratory pathogens, with Mycoplasma pneumoniae (MP) emerging as the predominant pathogen (76.51%). The study identified significant age and gender variations, with the highest infection rates observed in children aged 5–14 years (36.01%) and a notable female predominance in infection susceptibility (21.30% versus 15.97% in males). Serological analysis demonstrated distinct coagulation profiles in confirmed infections.
What is known and what is new?
• Prior research has established respiratory infections as significant public health concerns globally, with documented age-dependent distribution patterns and seasonal variations, particularly in urban settings.
• This study provides unprecedented insights through its comprehensive 11-year surveillance of multiple respiratory pathogens in southern China, revealing an exceptionally high prevalence of MP compared to other regions. The research uniquely identifies a significant gender disparity in infection rates and characterizes previously undocumented coagulation profiles in confirmed respiratory infections, suggesting complex underlying pathophysiological mechanisms.
What is the implication, and what should change now?
• These findings underscore the necessity for enhanced surveillance systems and targeted intervention strategies in urban settings, particularly in subtropical regions. The observed demographic patterns indicate the need for age- and gender-specific approaches to infection prevention and management. The distinctive coagulation profiles in confirmed infections suggest the importance of integrating coagulation monitoring into clinical protocols. These results should inform the development of more effective, population-specific public health strategies for respiratory infection control in rapidly urbanizing regions.
Introduction
Respiratory infections remain a significant global health concern, with lower respiratory tract infections alone causing approximately 3 million annual deaths worldwide (1). The etiology of these infections is diverse, encompassing viruses, bacteria, and atypical organisms, each presenting unique challenges to healthcare systems (2). Among children, respiratory infections are one of the leading causes of death in children, they are responsible every year for the deaths of 4.3 million children under 5 years of age worldwide (3).
Long-term surveillance studies worldwide have revealed varying patterns of respiratory pathogen prevalence. In Europe, Cillóniz et al. (4) conducted a 10-year study in Barcelona, Spain, demonstrating significant shifts in the predominance of different respiratory pathogens, with Streptococcus pneumoniae remaining the primary bacterial pathogen. Similarly, a comprehensive study in the United States by Jain et al. (5) analyzing community-acquired pneumonia trends showed distinct seasonal and demographic patterns in pathogen distribution. In Asia, Japanese researchers documented cyclical patterns of Mycoplasma pneumoniae (MP) infections, with significant outbreaks occurring every 3–4 years (6).
In China, several studies have explored the epidemiology of respiratory pathogens. For example, a study by Li et al. (7) of 233,037 patients with acute respiratory infections across the country found that the overall positive rate of MP was 18.60%. Recent regional studies have highlighted significant variations (8), with Beijing reporting MP infection rates of 62.11% (9), while studies from Xi’an and Tianjin documented distinct temporal patterns and post-coronavirus disease 2019 (COVID-19) surge in infections (10,11).
Guangzhou, a major metropolitan area in southern China, has experienced notable fluctuations in respiratory infection incidence over the past decade (12). While established pathogens like influenza viruses and respiratory syncytial virus (RSV) continue to pose threats, emerging evidence suggests an increasing role of atypical bacteria, particularly, MP in respiratory morbidity within the region (13).
Despite the importance of understanding these epidemiological dynamics for developing effective prevention and control strategies, comprehensive long-term studies examining multiple respiratory pathogens simultaneously in this region are limited. Moreover, the relationship between pathogen prevalence and host serological profiles in urban environments like Guangzhou remains understudied.
This study aims to address the knowledge gaps by providing a retrospective analysis of respiratory pathogen detection in Guangzhou from 2013 to 2023. We investigate temporal trends, demographic variations, and potential environmental and social factors influencing pathogen prevalence. Additionally, we explore serological profiles associated with different respiratory pathogens, offering insights into host immune responses and potential diagnostic markers. We present this article in accordance with the STROBE reporting checklist (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1528/rc).
Methods
Study design and population
This retrospective observational study was conducted at The First Affiliated Hospital of Guangzhou Medical University, a tertiary care center in Guangzhou, China. We analyzed data from 96,927 patients with suspected respiratory infections (SRIs) over an 11-year period from January 1, 2013, to October 26, 2023. The overall study design and analytical workflow is illustrated in Figure 1.

Inclusion criteria were: (I) patients presenting with symptoms suggestive of respiratory infection, including but not limited to cough, fever (>37.5 ℃), dyspnea, and/or chest pain; (II) age ≥0 years; and (III) willingness to provide respiratory specimens for pathogen testing. Exclusion criteria were: (I) patients with incomplete medical records; (II) those who withdrew consent for sample collection; and (III) cases where sample quality was inadequate for testing.
The age groups are based on our reference to the relevant rules of the United Nations Population Division (United Nations, 2019) (14). This study used the age grouping method recommended by the United Nations Population Division with slight modifications for our study purposes.
Sample collection and pathogen detection
Specimens were collected from all enrolled patients by trained healthcare professionals following standard infection control procedures. Specimens were immediately placed in transport medium and transported to the hospital’s clinical microbiology laboratory within 2 hours of collection. Samples were processed within 24 hours of receipt or stored at −80 ℃ if processing was delayed. An indirect immunofluorescence assay (Vircell, S.L, Spain) was employed to detect immunoglobulin M (IgM) antibodies against nine respiratory pathogens: Legionella pneumophila (LP), MP, Coxiella burnetii (COX), Chlamydia pneumoniae (CP), adenovirus (ADV), RSV, influenza A (IFA), influenza B (IFB), and parainfluenza viruses (PIVS).
Briefly, patient sera were diluted 1:40 and incubated with antigen-coated slides for 30 minutes at 37 ℃. After washing, fluorescein-labeled anti-human IgM was added and incubated for 30 minutes. Slides were examined under a fluorescence microscope by two independent, experienced technicians. Samples were considered serologically positive if they exhibited specific fluorescence at a titer ≥1:40. While immunofluorescence assays can be subject to interpretation variability, we implemented rigorous quality control measures, including independent examination by two experienced technicians. In this study, we used the following criteria to distinguish suspected from confirmed cases: Suspected cases: Patients exhibiting clinical symptoms consistent with a respiratory infection, such as cough, fever (>37.5 ℃), dyspnea, and/or chest pain. The clinician suspects a respiratory infection based on symptoms and physical examination. Confirmed cases: meet all the criteria for suspected cases. Respiratory pathogen specific IgM antibody test positive (titer ≥1:40).
Serological analysis
Blood samples were collected from patients at the time of respiratory specimen collection. Serum was separated by centrifugation at 3,000 g for 10 minutes and analyzed for the following parameters:
Coagulation indicators: international normalized ratio (INR), prothrombin time activity (PTA), fibrinogen (FIB), prothrombin time (PT), activated partial thromboplastin time (APTT), and thrombin time (TT).
Inflammatory markers: C-reactive protein (CRP), β2-microglobulin (β2-MG), lymphocyte count (LC) and rheumatoid factor (RF), 50% hemolytic unit of complement (CH50), complement 3 (C3), and complement 4 (C4).
All serological tests were performed using standardized automated analyzers in the hospital’s clinical laboratory. Quality control procedures were performed daily using commercial control materials. For parameters with values below or above the detection limits, the respective lower or upper detection thresholds were recorded.
Data collection and management
Demographic data, clinical information, and laboratory results were extracted from the hospital’s electronic medical record system. A standardized data collection form was developed to systematically record patient demographics (age, sex), clinical presentation, temporal information (admission date, testing date), and laboratory findings. Data extraction was performed by trained research assistants using a standardized protocol. For temporal trend analysis, we divided the study period into two distinct intervals: the pre-COVID-19 period (January 1, 2013, to November 30, 2019) and the COVID-19 period (December 1, 2019, to October 26, 2023), using the date of the first reported COVID-19 case as the cutoff point. Double data entry and regular data audits were performed to ensure accuracy.
Statistical analysis
Statistical analyses were performed using R software (version 4.4.3). Continuous variables were presented as median [interquartile range (IQR)], and categorical variables as frequencies and percentages. The Shapiro-Wilk test was used to assess data normality.
Age-specific infection rates were analyzed using Pearson’s chi-square test. To account for potential confounding factors, we employed multiple logistic regression models:
where Y represents infection status (1= positive, 0= negative), and X variables represent age groups (0–4, 5–14, 15–24, 25–59, ≥60 years), gender, testing period (pre- vs. post-COVID-19), seasonal variations, and weekend admission status, respectively. Results were presented as adjusted odds ratios (ORs) with 95% confidence intervals (CIs).
For serological indicators, between-group differences were initially assessed using the Mann-Whitney U test. To adjust for confounders, we developed multiple linear regression models:
where Yi represents each serological indicator. The Benjamini-Hochberg procedure was applied to control the false discovery rate in multiple comparisons. To address potential multicollinearity, variance inflation factors (VIFs) were calculated, with VIF >5 indicating problematic collinearity. Missing data were handled using multiple imputation with chained equations (MICEs) when the missing rate was <20%; cases with higher missing rates were excluded from the analysis. The Benjamini-Hochberg procedure was applied to control the false discovery rate in multiple comparisons. Statistical significance was denoted as: *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001, and ns, no statistical significance.
Ethical considerations
This study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the Institutional Review Board of The First Affiliated Hospital of Guangzhou Medical University (approval number: Medical Ethics Review 2016 No. 73). Written informed consent was obtained from all patients or their legal guardians. Patient confidentiality was maintained throughout the study, and all data were de-identified before analysis.
Results
Overall patient characteristics
Throughout the study period, 96,927 admitted patients who met the inclusion criteria were enrolled. The demographic indicators for each year of the study period are shown in Table 1.
Table 1
| Year | Total | Gender | Age (years) | Season | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Male | Female | 0–4 | 5–14 | 15–24 | 25–59 | ≥60 | Mean age | Spring | Summer | Autumn | Winter | ||||
| 2013 | 3,607 | 2,158 | 1,449 | 1,366 | 464 | 80 | 855 | 842 | 29.5 | 935 | 900 | 934 | 838 | ||
| 2014 | 6,352 | 4,005 | 2,347 | 1,506 | 385 | 156 | 1,814 | 2,491 | 46.95 | 1,785 | 1,710 | 1,606 | 1,251 | ||
| 2015 | 7,201 | 4,501 | 2,700 | 1,304 | 367 | 189 | 2,168 | 3,173 | 47.22 | 1,825 | 2,040 | 1,790 | 1,546 | ||
| 2016 | 8,193 | 5,124 | 3,069 | 1,413 | 463 | 230 | 2,434 | 3,653 | 47.42 | 2,377 | 2,191 | 1,824 | 1,801 | ||
| 2017 | 9,640 | 5,978 | 3,662 | 1,837 | 633 | 196 | 2,685 | 4,289 | 46.19 | 2,401 | 2,553 | 2,415 | 2,271 | ||
| 2018 | 10,911 | 6,711 | 4,200 | 2,267 | 748 | 260 | 2,996 | 4,640 | 44.73 | 2,990 | 2,805 | 2,393 | 2,723 | ||
| 2019 | 10,688 | 6,424 | 4,264 | 2,315 | 1,141 | 310 | 2,709 | 4,213 | 42.03 | 3,003 | 2,725 | 2,229 | 2,731 | ||
| 2020 | 6,758 | 4,092 | 2,666 | 811 | 320 | 220 | 2,426 | 2,981 | 49.25 | 1,287 | 1,781 | 1,908 | 1,782 | ||
| 2021 | 8,652 | 5,344 | 3,308 | 1,201 | 600 | 245 | 3,068 | 3,538 | 45.12 | 2,229 | 1,856 | 2,181 | 2,386 | ||
| 2022 | 10,584 | 6,318 | 4,266 | 1,244 | 757 | 324 | 3,769 | 4,490 | 48.28 | 2,452 | 3,433 | 2,695 | 2,004 | ||
| 2023 | 14,341 | 8,525 | 5,816 | 1,688 | 1,433 | 303 | 4,755 | 6,162 | 45.66 | 4,634 | 4,877 | 2,629 | 2,201 | ||
Total detection results of respiratory pathogens
From 2013 to 2023, a total of 96,927 SRI cases were reported in our hospital, of which 18.01% (17,454/96,927) were serologically positive for at least one respiratory pathogen (Table 2). The serologically positive rate of each monitoring year ranged from 9.50% (year 2016) to 30.53% (year 2023), and the serologically positive rate showed a downward trend from 2013 to 2016, and increased year by year from 2016 until a decline after 2019, and an increase after 2021 (Figure 2).
Table 2
| Year | Age (years) | SRI cases (n) | Detected cases (n) | Positive rate (%) | ||||
|---|---|---|---|---|---|---|---|---|
| 0–4 | 5–14 | 15–24 | 25–59 | ≥60 | ||||
| 2013 | 282 | 116 | 18 | 95 | 64 | 3,607 | 575 | 15.94 |
| 2014 | 218 | 93 | 24 | 158 | 122 | 6,352 | 615 | 9.68 |
| 2015 | 207 | 90 | 28 | 227 | 187 | 7,201 | 739 | 10.26 |
| 2016 | 203 | 84 | 35 | 228 | 228 | 8,193 | 778 | 9.5 |
| 2017 | 447 | 181 | 48 | 379 | 494 | 9,640 | 1,549 | 16.07 |
| 2018 | 581 | 188 | 74 | 548 | 637 | 10,911 | 2,028 | 18.59 |
| 2019 | 874 | 439 | 66 | 438 | 454 | 10,688 | 2,271 | 21.25 |
| 2020 | 247 | 91 | 52 | 392 | 343 | 6,758 | 1,125 | 16.65 |
| 2021 | 567 | 225 | 43 | 288 | 304 | 8,652 | 1,427 | 16.49 |
| 2022 | 451 | 298 | 75 | 617 | 528 | 10,584 | 1,969 | 18.6 |
| 2023 | 954 | 828 | 110 | 1,294 | 1,192 | 14,341 | 4,378 | 30.53 |
| Total | 5,025 | 2,633 | 573 | 4,664 | 4,553 | 96,927 | 17,454 | 18.01 |
SRI, suspected respiratory infection.

Pathogen-specific serologically positive rates
Among the 17,454 serologically positive cases, MP was the predominant pathogen, accounting for 76.51% (13,354/17,454) of all serologically positive detections. This was followed by PIVS at 9.75% (1,701/17,454) and IFB at 9.54% (1,665/17,454). The least frequently detected pathogen was CP at 1.13% (197/17,454) (Table 3).
Table 3
| Year | Detected cases (n) | LP (n) | MP (n) | COX (n) | CP (n) | ADV (n) | RSV (n) | IFA (n) | IFB (n) | PIVS (n) |
|---|---|---|---|---|---|---|---|---|---|---|
| 2013 | 575 | 1 | 492 | 1 | 0 | 26 | 7 | 14 | 73 | 35 |
| 2014 | 615 | 9 | 482 | 2 | 1 | 11 | 13 | 9 | 106 | 23 |
| 2015 | 739 | 14 | 620 | 9 | 7 | 13 | 7 | 6 | 92 | 13 |
| 2016 | 778 | 43 | 610 | 29 | 13 | 19 | 13 | 15 | 99 | 40 |
| 2017 | 1,549 | 88 | 1,002 | 46 | 29 | 53 | 115 | 26 | 152 | 288 |
| 2018 | 2,028 | 470 | 977 | 115 | 34 | 81 | 184 | 22 | 330 | 103 |
| 2019 | 2,271 | 61 | 2,038 | 16 | 7 | 50 | 59 | 4 | 135 | 28 |
| 2020 | 1,125 | 127 | 750 | 42 | 14 | 21 | 29 | 20 | 118 | 158 |
| 2021 | 1,427 | 156 | 998 | 65 | 16 | 262 | 23 | 6 | 24 | 93 |
| 2022 | 1,969 | 37 | 1,607 | 50 | 10 | 40 | 52 | 69 | 187 | 173 |
| 2023 | 4,378 | 94 | 3,778 | 43 | 66 | 82 | 185 | 239 | 349 | 747 |
| Total | 17,454 | 1,100 | 13,354 | 418 | 197 | 658 | 687 | 430 | 1,665 | 1,701 |
| Positive rate (%) | 6.30 | 76.51 | 2.39 | 1.13 | 3.77 | 3.94 | 2.46 | 9.54 | 9.75 |
ADV, adenovirus; COX, Coxiella burnetii; CP, Chlamydia pneumoniae; IFA, influenza A; IFB, influenza B; LP, Legionella pneumophila; MP, Mycoplasma pneumoniae; PIVS, parainfluenza viruses; RSV, respiratory syncytial virus.
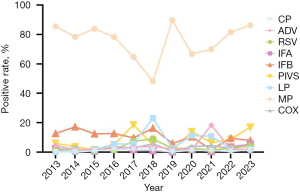
Temporal analysis revealed significant variations in pathogen prevalence over the study period. MP showed the highest serologically positive rate in 2019 (89.74% of serologically positive cases) and the lowest in 2018 (48.18%) (P<0.001). Notably, CP was not detected in 2013 (Figure 3).
Age distribution of infections
Analysis of age-specific infection rates revealed that patients aged 0–4 years and those over 25 years had the highest number of infections (Table 4). However, the highest serologically positive rate was observed in the 5–14 age group (36.01%, 2,633/7,311), followed by the 0–4 age group (30.32%, 5,025/16,592) (Figure 4A). The difference in serologically positive rates across age groups was statistically significant (P<0.001) (Table 5).
Table 4
| Year | 0–4 years old | 5–14 years old | 15–24 years old | 25–59 years old | ≥60 years old | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Detection number | Positive rate (%) | Detection number | Positive rate (%) | Detection number | Positive rate (%) | Detection number | Positive rate (%) | Detection number | Positive rate (%) | |||||
| 2013 | 1,366 | 20.65 | 464 | 25.00 | 80 | 22.50 | 855 | 11.11 | 842 | 15.94 | ||||
| 2014 | 1,506 | 14.48 | 385 | 24.46 | 156 | 15.38 | 1,814 | 8.71 | 2,491 | 4.90 | ||||
| 2015 | 1,304 | 15.87 | 367 | 24.52 | 189 | 14.81 | 2,168 | 10.47 | 3,173 | 5.89 | ||||
| 2016 | 1,413 | 14.37 | 463 | 18.14 | 230 | 15.22 | 2,434 | 9.37 | 3,653 | 6.24 | ||||
| 2017 | 1,837 | 24.33 | 633 | 28.59 | 196 | 24.49 | 2,685 | 14.12 | 4,289 | 11.52 | ||||
| 2018 | 2,267 | 25.63 | 748 | 25.13 | 260 | 28.46 | 2,996 | 18.29 | 4,640 | 13.73 | ||||
| 2019 | 2,315 | 37.75 | 1,141 | 38.48 | 310 | 21.29 | 2,709 | 16.17 | 4,213 | 10.78 | ||||
| 2020 | 811 | 30.46 | 320 | 28.44 | 220 | 23.64 | 2,426 | 16.16 | 2,981 | 11.51 | ||||
| 2021 | 1,201 | 47.21 | 600 | 37.50 | 245 | 17.55 | 3,068 | 9.39 | 3,538 | 8.59 | ||||
| 2022 | 1,244 | 36.25 | 757 | 39.37 | 324 | 23.15 | 3,769 | 16.37 | 4,490 | 11.76 | ||||
| 2023 | 1,688 | 56.52 | 1,433 | 57.78 | 303 | 36.30 | 4,755 | 27.21 | 6,162 | 19.34 | ||||
| Total | 16,592 | 30.32 | 7,311 | 36.01 | 2,513 | 22.80 | 29,679 | 15.71 | 40,472 | 11.25 | ||||

Table 5
| Age (years) | OR (95% CI) | χ2 | P value |
|---|---|---|---|
| 5–14 (ref.) | – | 3,534.14 | <0.001 |
| 0–4 | 0.702 (0.655–0.751) | <0.001 | |
| 15–24 | 0.487 (0.431–0.550) | <0.001 | |
| 25–25 | 0.305 (0.285–0.326) | <0.001 | |
| ≥60 | 0.219 (0.205–0.234) | <0.001 |
CI, confidence interval; OR, odds ratio.
Pathogen-specific age distributions showed distinct patterns (Figure 4B). LP, COX, and IFA infections were predominantly found in individuals over 15 years old. In contrast, ADV and RSV showed the highest serologically positive rate in children under 4 years. CP exhibited peak prevalence in the 15–24 age group. MP and PIVS showed relatively uniform distribution across all age groups. Figure 4C shows the positive rate of each age group in different years.
Gender differences in infection rates
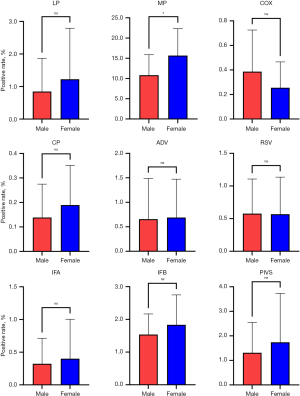
A significant gender disparity was observed in infection rates (Figure 5). Among 58,880 male SRI patients, 9,415 (15.97%) tested serologically positive for at least one pathogen. In contrast, of 37,747 female SRI patients, 8,039 (21.30%) were serologically positive (Table 6, P<0.001). After adjusting for age group, testing period, season, and weekend admission status, we observed a significantly higher risk of respiratory pathogen infection in females compared to males (OR =1.46, 95% CI: 1.41–1.52). This finding suggests that females had 46% higher odds of being infected compared to males, independent of other demographic and temporal factors.

Table 6
| Year | Male | Female | |||||
|---|---|---|---|---|---|---|---|
| Detection number | Positive number | Positive rate (%) | Detection number | Positive number | Positive rate (%) | ||
| 2013 | 2,158 | 282 | 13.07 | 1,449 | 293 | 20.22 | |
| 2014 | 4,005 | 314 | 7.85 | 2,347 | 301 | 12.82 | |
| 2015 | 4,501 | 409 | 9.09 | 2,700 | 330 | 12.22 | |
| 2016 | 5,124 | 417 | 8.14 | 3,069 | 361 | 11.76 | |
| 2017 | 5,978 | 863 | 14.44 | 3,662 | 686 | 18.73 | |
| 2018 | 6,711 | 1,133 | 16.88 | 4,200 | 895 | 21.31 | |
| 2019 | 6,124 | 1,176 | 18.31 | 4,264 | 1,095 | 25.68 | |
| 2020 | 4,092 | 626 | 15.30 | 2,666 | 499 | 18.72 | |
| 2021 | 5,344 | 842 | 15.76 | 3,308 | 585 | 17.68 | |
| 2022 | 6,318 | 1,030 | 16.30 | 4,266 | 939 | 22.01 | |
| 2023 | 8,525 | 2,323 | 27.25 | 5,816 | 2,055 | 35.33 | |
| Total | 58,880 | 9,415 | 15.97 | 37,747 | 8,039 | 21.30 | |
The higher infection rate in females was consistent across all years of the study period. Notably, MP infections showed a significantly higher rate in females compared to males. Other pathogens, except COX, also showed higher rates in females, although these differences were not statistically significant (Figure 6).
Serological indicators of SRI patients
As shown in Figure 7, coagulation-related parameters including PT, APTT, and FIB showed significant differences between groups. Figure 8 demonstrates the changes in inflammatory markers, with notable elevations in complement components and immune cell counts in the confirmed infection group (CIG). After adjusting for age, sex, testing period, season, and weekend admission status, multivariate analysis revealed significant differences in multiple serological parameters between CIG and suspected infection group (SIG) (Table 7). The coagulation profile showed noteworthy changes: APTT demonstrated significant differences between groups (unadjusted difference: −3.21 seconds, adjusted difference: 1.57±0.18 seconds, P<0.001), followed by PTA (unadjusted: −6.44%, adjusted: 3.29%±0.36%, P<0.001). These findings remained robust after adjustment for potential confounding factors.


Table 7
| Serological indicators | SIG | CIG | Unadjusted | Adjusted | P value |
|---|---|---|---|---|---|
| CH50 (U/mL) | 25.77±25.25 | 29.59±25.80 | 3.82 (−4.29 to −3.34) | 1.30±0.24 | <0.001/<0.001 |
| CRP (mg/L) | 1.76±4.39 | 1.74±3.78 | −0.02 (−0.05 to 0.09) | 0.00±0.04 | 0.57/0.95 |
| INR | 0.99±0.63 | 0.89±0.62 | −0.10 (0.09 to 0.11) | 0.05±0.01 | <0.001/<0.001 |
| β2-MG (mg/L) | 1.68±2.85 | 1.65±2.32 | −0.03 (−0.02 to 0.07) | −0.10±0.03 | 0.24/<0.001 |
| PT (seconds) | 12.31±7.36 | 11.05±7.43 | −1.25 (1.12 to 1.39) | 0.45±0.06 | <0.001/<0.001 |
| PTA (%) | 81.87±40.52 | 75.43±44.68 | −6.44 (5.62 to 7.25) | 3.29±0.36 | <0.001/<0.001 |
| TT (seconds) | 16.69±19.83 | 14.64±16.83 | −2.06 (1.73 to 2.38) | 0.43±0.18 | <0.001/0.02 |
| APTT (seconds) | 35.63±20.18 | 32.41±20.68 | −3.21 (2.83 to 3.59) | 1.57±0.18 | <0.001/<0.001 |
| LC (×109/L) | 2.50±2.32 | 2.75±2.29 | 0.26 (−0.30 to −0.21) | −0.34±0.02 | <0.001/<0.001 |
| RF (IU/mL) | 9.46±48.77 | 11.98±58.10 | 2.52 (−3.56 to −1.47) | 3.27±0.50 | <0.001/<0.001 |
| FIB (g/L) | 4.06±2.46 | 3.51±2.41 | −0.55 (0.50 to 0.59) | 0.15±0.02 | <0.001/<0.001 |
| C3 (g/L) | 0.56±0.52 | 0.64±0.50 | 0.07 (−0.08 to −0.06) | −0.02±0.00 | <0.001/<0.001 |
| C4 (g/L) | 0.16±0.18 | 0.18±0.20 | 0.03 (−0.03 to −0.02) | 0.01±0.00 | <0.001/<0.001 |
SIG and CIG columns show mean ± standard deviation; unadjusted column shows difference (95% confidence interval); adjusted column shows adjusted difference ± standard error. P value column is presented as ‘unadjusted P value/adjusted P value’. APTT, activated partial thromboplastin time; β2-MG, β2-microglobulin; C3, complement 3; C4, complement 4; CH50, 50% hemolytic unit of complement; CRP, C-reactive protein; CIG, confirmed infection group; FIB, fibrinogen; INR, international normalized ratio; LC, lymphocyte count; PT, prothrombin time; PTA, prothrombin time activity; RF, rheumatoid factor; SIG, suspected infection group; TT, thrombin time.
Among inflammatory markers, CH50 levels were significantly higher in the CIG (unadjusted difference: 3.82 U/mL, adjusted: 1.30±0.24 U/mL, P<0.001). Notably, the magnitude of this difference was attenuated after adjustment, suggesting partial influence of demographic and temporal factors. CRP levels showed no significant difference between groups after adjustment (0.00±0.04 mg/L, P=0.95).
The immune cell profile, represented by lymphocyte count (LC), showed a consistent pattern both before and after adjustment, with significantly higher values in the confirmed group [unadjusted: 0.26×109/L, adjusted: (−0.34±0.02)×109/L, P<0.001]. Complement components exhibited subtle but significant changes, with C3 showing minimal variation after adjustment (−0.02±0.00 g/L, P<0.001).
Importantly, while most markers showed significant differences in unadjusted analyses, the magnitudes of these differences were generally attenuated after controlling for confounding factors, indicating that demographic and temporal factors partially explained the observed variations (Figures S1,S2). However, the persistence of significant differences after adjustment suggests genuine biological associations between infection status and specific serological parameters.
Discussion
This comprehensive study, analyzing 96,927 cases of suspected or confirmed respiratory infections from 2013 to 2023 at The First Affiliated Hospital of Guangzhou Medical University, revealed several significant epidemiological trends. Respiratory pathogen infections were predominantly observed in children aged 0–14 years, with a consistent and statistically significant higher serologically positive rate in women compared to men (21.30% vs. 15.97%, P<0.001). Notably, MP emerged as the predominant pathogen, accounting for 76.51% of all serologically positive cases. The temporal analysis showed fluctuating trends, with a decline from 2013 to 2016, followed by an increase until 2019, and a resurgence post-2021. Intriguingly, our serological analysis revealed lower coagulation-related indicators in the CIG compared to the SIG. These findings raise important questions about the dynamics of respiratory infections in our region and warrant a deeper exploration of the underlying factors influencing these patterns.
While this study primarily focused on Guangzhou, comparing our findings with data from other regions in China provides valuable insights into the prevalence of respiratory pathogens, particularly MP. A comprehensive study by Li et al. (7) encompassing 233,037 patients with acute respiratory infections across mainland China reported an MP positive rate of 18.60%. Interestingly, our study revealed a substantially higher MP positive rate of 83.18% in a comparable age group, contrasting with the 62.11% reported by Guo et al. (9) in Beijing, northern China. Although methodological differences may partially account for this disparity, the marked variation warrants further investigation. The exceptionally high prevalence of MP in Guangzhou may be attributed to several factors inherent to its urban landscape and climate. As an economically developed and densely populated metropolis in southern China, Guangzhou’s urban planning, characterized by high-density residential areas and extensive public transportation networks, creates an environment conducive to pathogen transmission. Moreover, Guangzhou’s subtropical climate, featuring high temperatures and humidity, provides an optimal environment for the survival and proliferation of MP. This climatic factor likely contributes to the elevated positive rates observed in our study, a phenomenon also noted in previous research (15). Additionally, the often mild and nonspecific nature of initial MP infection symptoms frequently results in delayed diagnosis and treatment. This delay not only extends the period of potential transmission but also complicates early detection efforts (16). The combination of these factors—Guangzhou’s urban density, its climate, and the distinctive properties of MP—collectively contribute to the high positive rates observed across different age groups in our study. This underscores the need for targeted surveillance and control strategies specific to urban environments in subtropical regions.
Our study found that the 5–14-year age group had the highest serologically positive rate (36.01%), followed by the 0–4-year age group (30.32%). This may be related to greater exposure of children and adolescents in these age groups to pathogens, still-developing immune systems, and frequent exposure in closed environments such as schools (17). Given the differences in infection rates among different age groups, it is necessary to develop targeted prevention strategies. For example, strengthening health measures in schools and kindergartens, raising awareness among parents and teachers, and considering vaccination programs targeting high-risk age groups (if available).
Our study reveals a notable gender disparity, with women exhibiting higher serologically positive rates of respiratory pathogen infections compared to men. This difference was statistically significant for MP infections. This observed gender difference likely stems from a complex interplay of physiological, social, and behavioral factors. From a physiological perspective, sex-related differences in immune responses appear to play a crucial role. As highlighted in a comprehensive review by Klein et al. (18), women generally possess more active immune systems than men, a phenomenon largely attributed to the regulatory effects of sex hormones, particularly estrogen. Estrogen has been shown to enhance B and T cell activity and increase antibody production, potentially enabling women to recognize and clear pathogens more effectively in the early stages of infection (19-21). Paradoxically, this enhanced immune response, while beneficial for pathogen clearance, may contribute to higher rates of positive test results. The heightened immune activity can trigger positive test outcomes even when pathogen levels are relatively low (22,23), potentially inflating the observed infection rates in women.
Social and behavioral factors further compound this gender disparity. Women often occupy social roles that increase their exposure to respiratory pathogens. For instance, as mothers, caregivers, or educators, women may have more frequent contact with children and older adults—demographics that our study and others have identified as high-risk groups for respiratory infections (24). Moreover, gender-specific lifestyle factors may contribute to differential exposure rates. Women’s daily hygiene habits, which may involve more frequent engagement in household activities and patient care, could inadvertently increase their exposure to pathogens (25). Additionally, as noted by Thompson et al. (26), women’s greater propensity to seek medical attention for health concerns and undergo regular health check-ups may lead to higher rates positive of pathogen detection. This complex interplay of biological and sociocultural factors not only explains our observed gender differences but also highlights the importance of considering sex and gender as critical variables in respiratory pathogen epidemiology. These findings emphasize the need for gender-sensitive approaches in both research design and the development of targeted prevention and treatment strategies for respiratory infections.
The relationship between infection and coagulation presents a complex interplay that challenges traditional understanding. While classical theory suggests that pathogen-induced inflammation depletes clotting factors, leading to prolonged PT and APTT (27,28), our findings reveal an unexpected pattern. Patients with confirmed infections demonstrated shortened PT and APTT compared to those with suspected infections, indicating an enhanced hypercoagulable state that represents a more sophisticated physiological response than previously recognized.
This hypercoagulability likely serves as an adaptive defense mechanism against severe infections (29), with the coagulation system undergoing hyperactivation to contain pathogen spread. Rather than experiencing clotting factor depletion, the body appears to maintain hemostatic equilibrium through compensatory mechanisms, including accelerated factor production (27,30,31). Furthermore, infection-induced local microvascular injury may trigger a cascade leading to systemic hyperactivation of the coagulation system (32).
These observations highlight the dynamic nature of coagulation during infection, where profiles vary with disease stage and severity. For instance, fibrinogen typically shows early elevation but may decline as infection progresses (33,34). These findings not only challenge our understanding of infection-related coagulation changes but also have important clinical implications. The observed hypercoagulable state in confirmed infections suggests that patients may be at increased risk of thrombotic complications, necessitating careful monitoring and potentially prophylactic anticoagulation. Moreover, these results highlight the need for a more nuanced interpretation of coagulation parameters in the context of acute infections.
Our analysis revealed significant elevations in key immunological markers—rheumatoid factor, complement components, and lymphocyte counts—in the CIG, reflecting a robust immune response during respiratory infections. Notably, the elevation in rheumatoid factor, typically associated with autoimmune conditions, suggests broader immune system activation during acute infections (35). The enhanced complement system activity and elevated lymphocyte counts demonstrate the body’s coordinated response to pathogens, with the complement pathway playing crucial roles in both bacterial and viral infections (36,37), while increased lymphocytes indicate robust cellular immunity (38,39). These dynamic immunological changes reveal the sophisticated interplay between immune components and respiratory pathogens. The distinct immune profiles observed may aid in differentiating between bacterial and viral etiologies, potentially guiding more targeted therapeutic approaches. These findings not only demonstrate the immune system’s adaptability but also provide valuable insights for improving diagnostic and therapeutic strategies in respiratory infections.
There are some similarities and differences between our findings and other domestic studies. For example, Hu et al. (13) and Chen et al. (40) also found that MP predominate in respiratory infections. At the same time, Lv et al. (41) and Qu et al. (42) also support the idea of a higher rate of detection of respiratory pathogens (especially MP) in children in this study. However, the gender differences we observed were not evident in certain other studies, which may reflect regional characteristics or differences in research methods. Compared to international studies, our study shows some unique features. For example, the high positive rate of MP that we observed was not common in studies in certain European and American countries (5,43). This may reflect factors such as geography, climate and population density. However, the age distribution pattern we observed is like the findings of many international studies (6,44), highlighting that children and adolescents are a high-risk group for respiratory infections.
This study offers several innovative contributions to the field of respiratory pathogen epidemiology. Firstly, our 11-year consecutive analysis provides an unprecedented long-term perspective on respiratory pathogen trends in a rapidly urbanizing Chinese metropolis, offering valuable insights into the shifting landscape of infections in the context of urban development. Secondly, the comprehensive examination of nine major respiratory pathogens simultaneously allows for a nuanced understanding of their relative prevalence and interactions. The observed predominance of MP, particularly its high serologically positive rate compared to other regions, highlights the unique epidemiological profile of Guangzhou. Furthermore, our finding of significant gender disparities in infection rates, especially for MP, opens new avenues for research into sex-based differences in respiratory infections. The detailed age-specific analysis not only confirms the vulnerability of pediatric populations but also reveals unexpected patterns in adult age groups, challenging existing assumptions about age-related risks. Lastly, our novel findings regarding coagulation abnormalities in confirmed infections provide a new perspective on the systemic effects of respiratory pathogens, potentially informing future diagnostic and therapeutic approaches. Collectively, these innovations contribute to a more comprehensive understanding of respiratory pathogen dynamics in urban settings and may guide targeted public health interventions.
Several limitations of this study warrant discussion. First, despite our comprehensive dataset, the single-center design in Guangzhou may not fully represent broader geographical patterns of respiratory infections. Second, while immunofluorescence assays offer practical advantages for large-scale screening, their inherent technical limitations could have resulted in false negatives, particularly for novel pathogen variants. Third, the COVID-19 pandemic introduced potential confounding effects through altered healthcare utilization patterns and enhanced infection control protocols, especially during 2020–2023. Fourth, the retrospective nature of our analysis precluded the collection of certain potentially relevant clinical and behavioral data, limiting our ability to explore causal mechanisms. Nevertheless, our study’s strengths, including its large sample size (n=96,927) and extended temporal scope (11 years), provide robust evidence for understanding respiratory pathogen dynamics in urban Chinese populations.
Conclusions
This comprehensive study provides crucial insights into the changing landscape of respiratory pathogen infections in Guangzhou over an 11-year period. The predominance of MP, particularly among school-age children and females, highlights the need for targeted interventions. The coagulation profiles in confirmed infections suggest complex pathophysiological mechanisms that warrant further investigation. These findings have important implications for clinical management, diagnostic approaches, and public health strategies in urban settings.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1528/rc
Data Sharing Statement: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1528/dss
Peer Review File: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1528/prf
Funding: This study was supported by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1528/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study protocol was approved by the Institutional Review Board of The First Affiliated Hospital of Guangzhou Medical University (approval number: Medical Ethics Review 2016 No. 73). Written informed consent was obtained from all patients or their legal guardians.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Global burden of 369 diseases and injuries in 204 countries and territories, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020;396:1204-22. [Crossref] [PubMed]
- Shi T, McLean K, Campbell H, et al. Aetiological role of common respiratory viruses in acute lower respiratory infections in children under five years: A systematic review and meta-analysis. J Glob Health 2015;5:010408. [Crossref] [PubMed]
- Vinod A, Kaimal RS. Study on acute respiratory infection in children aged 1 year to 5 years-A hospital-based cross-sectional study. J Family Med Prim Care 2023;12:666-71. [Crossref] [PubMed]
- Cillóniz C, Ewig S, Polverino E, et al. Community-acquired pneumonia in outpatients: aetiology and outcomes. Eur Respir J 2012;40:931-8. [Crossref] [PubMed]
- Jain S, Self WH, Wunderink RG, et al. Community-Acquired Pneumonia Requiring Hospitalization among U.S. Adults. N Engl J Med 2015;373:415-27. [Crossref] [PubMed]
- Morozumi M, Takahashi T, Ubukata K. Macrolide-resistant Mycoplasma pneumoniae: characteristics of isolates and clinical aspects of community-acquired pneumonia. J Infect Chemother 2010;16:78-86. [Crossref] [PubMed]
- Li ZJ, Zhang HY, Ren LL, et al. Etiological and epidemiological features of acute respiratory infections in China. Nat Commun 2021;12:5026. [Crossref] [PubMed]
- Ruan Z, Qi J, Qian ZM, et al. Disease burden and attributable risk factors of respiratory infections in China from 1990 to 2019. Lancet Reg Health West Pac 2021;11:100153. [Crossref] [PubMed]
- Guo DX, Hu WJ, Wei R, et al. Epidemiology and mechanism of drug resistance of Mycoplasma pneumoniae in Beijing, China: A multicenter study. Bosn J Basic Med Sci 2019;19:288-96. [Crossref] [PubMed]
- Shi D, Yu X, Sun J, et al. An exceptionally large wave of M. pneumoniae infections among children in Tianjin post COVID-19 pandemic. Transl Pediatr 2024;13:1703-10. [Crossref] [PubMed]
- Meng GL, Kang R, Cheng XY, et al. Laboratory analysis of positive rate of Mycoplasma pneumoniae antibody among 53,273 children with respiratory tract infections in Xi'an from 2017 to 2020. Transl Pediatr 2022;11:625-30. [Crossref] [PubMed]
- Tong MX, Hansen A, Hanson-Easey S, et al. Infectious Diseases, Urbanization and Climate Change: Challenges in Future China. Int J Environ Res Public Health 2015;12:11025-36. [Crossref] [PubMed]
- Hu H, Hou X, Wu J, et al. Impact of COVID-19 Pandemic Control Measures on Infection of Other Respiratory Pathogens: A Real-world Data Research in Guangzhou, China. J Transl Int Med 2022;10:272-6. [Crossref] [PubMed]
- United Nations DoEaSA, Population Division. World Population Prospects 2019, Online Edition. Rev. 1.; 2019.
- Yan C, Xue GH, Zhao HQ, et al. Current status of Mycoplasma pneumoniae infection in China. World J Pediatr 2024;20:1-4. [Crossref] [PubMed]
- Waites KB, Xiao L, Liu Y, et al. Mycoplasma pneumoniae from the Respiratory Tract and Beyond. Clin Microbiol Rev 2017;30:747-809. [Crossref] [PubMed]
- Roh EJ, Shim JY, Chung EH. Epidemiology and surveillance implications of community-acquired pneumonia in children. Clin Exp Pediatr 2022;65:563-73. [Crossref] [PubMed]
- Klein SL, Flanagan KL. Sex differences in immune responses. Nat Rev Immunol 2016;16:626-38. [Crossref] [PubMed]
- Straub RH. The complex role of estrogens in inflammation. Endocr Rev 2007;28:521-74. [Crossref] [PubMed]
- Taneja V. Sex Hormones Determine Immune Response. Front Immunol 2018;9:1931. [Crossref] [PubMed]
- Kovats S. Estrogen receptors regulate innate immune cells and signaling pathways. Cell Immunol 2015;294:63-9. [Crossref] [PubMed]
- vom Steeg LG, Klein SL. SeXX Matters in Infectious Disease Pathogenesis. PLoS Pathog 2016;12:e1005374. [Crossref] [PubMed]
- Furman D, Hejblum BP, Simon N, et al. Systems analysis of sex differences reveals an immunosuppressive role for testosterone in the response to influenza vaccination. Proc Natl Acad Sci U S A 2014;111:869-74. [Crossref] [PubMed]
- Gabriel G, Arck PC. Sex, immunity and influenza. J Infect Dis 2014;209:S93-9. [Crossref] [PubMed]
- Giefing-Kröll C, Berger P, Lepperdinger G, et al. How sex and age affect immune responses, susceptibility to infections, and response to vaccination. Aging Cell 2015;14:309-21. [Crossref] [PubMed]
- Thompson AE, Anisimowicz Y, Miedema B, et al. The influence of gender and other patient characteristics on health care-seeking behaviour: a QUALICOPC study. BMC Fam Pract 2016;17:38. [Crossref] [PubMed]
- Levi M, van der Poll T. Coagulation and sepsis. Thromb Res 2017;149:38-44. [Crossref] [PubMed]
- Short KR, Kroeze EJBV, Fouchier RAM, et al. Pathogenesis of influenza-induced acute respiratory distress syndrome. Lancet Infect Dis 2014;14:57-69. [Crossref] [PubMed]
- Jackson SP, Darbousset R, Schoenwaelder SM. Thromboinflammation: challenges of therapeutically targeting coagulation and other host defense mechanisms. Blood 2019;133:906-18. [Crossref] [PubMed]
- Kapur R, Zufferey A, Boilard E, et al. Nouvelle cuisine: platelets served with inflammation. J Immunol 2015;194:5579-87. [Crossref] [PubMed]
- Zarà M, Guidetti GF, Camera M, et al. Biology and Role of Extracellular Vesicles (EVs) in the Pathogenesis of Thrombosis. Int J Mol Sci 2019;20:2840. [Crossref] [PubMed]
- Semeraro N, Ammollo CT, Semeraro F, et al. Sepsis, thrombosis and organ dysfunction. Thromb Res 2012;129:290-5. [Crossref] [PubMed]
- Machlus KR, Cardenas JC, Church FC, et al. Causal relationship between hyperfibrinogenemia, thrombosis, and resistance to thrombolysis in mice. Blood 2011;117:4953-63. [Crossref] [PubMed]
- Wolberg AS, Rosendaal FR, Weitz JI, et al. Venous thrombosis. Nat Rev Dis Primers 2015;1:15006. [Crossref] [PubMed]
- Listing J, Gerhold K, Zink A. The risk of infections associated with rheumatoid arthritis, with its comorbidity and treatment. Rheumatology (Oxford) 2013;52:53-61. [Crossref] [PubMed]
- Cedzyński M, Thielens NM, Mollnes TE, et al. The role of complement in health and disease. Frontiers Media SA; 2019:1869.
- Ricklin D, Reis ES, Lambris JD. Complement in disease: a defence system turning offensive. Nat Rev Nephrol 2016;12:383-401. [Crossref] [PubMed]
- Zhao Q, Meng M, Kumar R, et al. Lymphopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: A systemic review and meta-analysis. Int J Infect Dis 2020;96:131-5. [Crossref] [PubMed]
- Clarice CSH, Abeysuriya V, de Mel S, et al. Atypical lymphocyte count correlates with the severity of dengue infection. PLoS One 2019;14:e0215061. [Crossref] [PubMed]
- Chen Z, Ji W, Wang Y, et al. Epidemiology and associations with climatic conditions of Mycoplasma pneumoniae and Chlamydophila pneumoniae infections among Chinese children hospitalized with acute respiratory infections. Ital J Pediatr 2013;39:34. [Crossref] [PubMed]
- Lv YT, Sun XJ, Chen Y, et al. Epidemic characteristics of Mycoplasma pneumoniae infection: a retrospective analysis of a single center in Suzhou from 2014 to 2020. Ann Transl Med 2022;10:1123. [Crossref] [PubMed]
- Qu J, Yang C, Bao F, et al. Epidemiological characterization of respiratory tract infections caused by Mycoplasma pneumoniae during epidemic and post-epidemic periods in North China, from 2011 to 2016. BMC Infect Dis 2018;18:335. [Crossref] [PubMed]
- Cillóniz C, Ewig S, Polverino E, et al. Microbial aetiology of community-acquired pneumonia and its relation to severity. Thorax 2011;66:340-6. [Crossref] [PubMed]
- Jacobs E, Ehrhardt I, Dumke R. New insights in the outbreak pattern of Mycoplasma pneumoniae. Int J Med Microbiol 2015;305:705-8. [Crossref] [PubMed]


