
Implementation of a robotic-assisted thoracic surgery program for anatomical lung resections, a single surgeon’s experience
Highlight box
Key findings
• Robotic-assisted thoracic surgery (RATS) for anatomical pulmonary resections can be safely implemented with a learning curve of 44 cases.
• The duration of chest tube retention was shorter after robotic-assisted technique compared to uniportal video-assisted technique, but other short-term results were similar for both techniques.
What is known and what is new?
• It is known that RATS is safe and feasible with short-term results comparable to those of video-assisted thoracic surgery.
• A robotic-assisted surgical program for pulmonary resections can be safely implemented despite limited access to the robotic-assisted platform with a learning curve of approximately 44 cases.
What is the implication, and what should change now?
• Thoracic surgeons together with the multi-disciplinary thoracic team should be encouraged to take on robotic-assisted surgery for pulmonary resections also at low volume centres.
Introduction
Background
The surgical standard technique for anatomical lung resections has evolved from open surgery to minimally-invasive approaches, preponderantly the use of video-assisted thoracic surgery (VATS) (1). The results of minimally-invasive procedures with emphasis on radical resections has been shown to be non-inferior to the open techniques, but with the advantages of less postoperative pain and faster recovery as reflected in shorter length of hospital stay (2,3). Better recovery of physical function after lobectomy was found for VATS compared to open surgery in a randomized study (4). Robotic-assisted thoracic surgery (RATS) is increasingly used for anatomical pulmonary resections and several centres are using the technique as a stand-alone methodology or as a complementary technique to pulmonary resections by VATS. Since the first report of RATS procedures for pulmonary resections (5), technical advantages over VATS such as enhanced visualization and improved lymph node dissection have repeatedly been described for RATS procedures of early-stage lung cancer (6,7). Large-scale retrospective analysis has shown similar overall survival for RATS procedures and open surgical procedures for lobectomy in non-small cell lung cancer patients (8).
Rationale and knowledge gap
One impediment to the adoption of RATS surgeons experienced in VATS is that limited access to the robotic platform, which often is shared among various specialities, is still an issue in many institutions, particularly in a smaller centre with lower volumes and few robotic surgical systems.
Objective
We here describe the initial experience and learning curve for a single surgeon’s first RATS cases by comparing the results to a series of uniportal video-assisted thoracic surgery (uVATS) procedures concomitantly performed during the same period at a tertiary university hospital performing less than 180 surgeries for lung cancer per year and with access to the robotic platform limited to two cases per week. The manuscript is presented in accordance with the STROBE reporting checklist (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1182/rc).
Methods
Inclusion and exclusion
A RATS program was initiated at the Department of Cardiothoracic Surgery at Sahlgrenska University Hospital in Gothenburg, Sweden, in 2021. uVATS procedures was commenced in 2019 and fully standardized by 2021. This retrospective study of the implementation of RATS for anatomical pulmonary resections was performed using a prospectively collected database of patients who underwent a unilateral lobectomy or bilobectomy procedure by RATS or uVATS by a single surgeon (A.W.) between November 2021 to September 2023. Patients whose surgery started with intention of RATS or uVATS but who were converted to open surgery were excluded from the analysis.
Patients were preoperatively assessed in accordance with clinical routine at our department including computed tomography of the chest and positron emission tomography scan for lymph node assessment and invasive lymph node staging with endobronchial ultrasound biopsy if indicated. Assessment of preoperative physical function included spirometry for all patients, and ergospirometry or cardiopulmonary exercise test if deemed necessary for risk assessment. All patients were evaluated at a multidisciplinary board and approved for surgical intervention by consensus of a thoracic surgeon, a thoracic radiologist, and a pulmonologist and/or an oncologist. At the multidisciplinary board, decision was made about whether surgery should be primarily planned as open surgery by thoracotomy or by minimally-invasive technique. Criteria for choice of a minimally-invasive approach were not prescribed but according to consensus of the surgeons attending the multidisciplinary board an open approach would be chosen for very large tumours and advanced tumour stages, suspected growth of tumour into chest wall or mediastinal structures, or prior ipsilateral lung resection more complex than one or several wedge resections. For the first RATS cases, selection was based on smaller tumour size without specification of a conditional tumour size, but this selection was gradually abandoned as the experience of RATS procedures increased.
Duration of operation and time in the operation room were collected from the digital operative log program. Console time is time for surgeon active in console and was collected from the incorporated software of the robotic platform. Operation time (OP time) was defined as skin-to-skin time and included placement of ports and docking of robot. Operation room time (OR time) was defined as time from the start of draping the patient to the patient leaving the theatre, including extubation. Preparation time included placement of epidural catheter for analgesia, anaesthesia, and positioning of patient. Parts of preparation were in some cases done outside the operation room.
Education and preparation of implementation
After simulator training, a two-day course with hands-on practice for console-surgeons and scrub nurse was undertaken. A dedicated team of a thoracic anaesthesiologist, anaesthesiology and assistant nurses was assembled for approximately the first 20 cases. All team members had solid experience in the different aspects of VATS procedures. The team hereafter gradually educated their colleagues and by the end of the study period most staff members could act as team members during the RATS procedures. First two RATS lobectomies were performed under guardianship of a proctor. Prior to these procedures, the team performed one wedge resection and two mediastinal resections before starting with lobectomies. After the initial first cases other thoracic surgeons or residents took on the role as bedside assisting surgeons. For uniportal RATS procedures bedside assistants were limited to surgeons experienced in VATS.
Surgical technique
Patients in the RATS group were if eligible given preoperatively a thoracic epidural catheter for analgesia which was discontinued after chest tube (CT) removal. If epidural catheterization was not possible an intercostal nerve blockade was administered in the same manner as for uVATS cases. RATS group patients were anesthetised in a standard manner with propofol and remifentanil with muscle relaxation achieved and maintained during the procedure with rocuronium infusion. uVATS patients received standard intraoperative intercostal blockade with ropivacaine and additional opioids; a thoracic epidural catheter was installed only if special postoperative pain problems were anticipated.
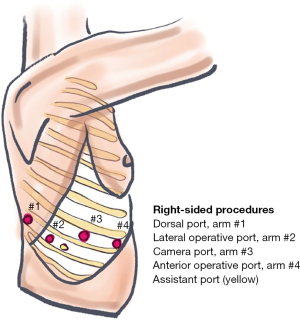
All patients were intubated with a double lumen tracheal tube for single lung ventilation and positioned in lateral decubitus position. RATS procedures were performed using the Da Vinci Xi (Intuitive Surgical, Inc., Sunnyvale, CA, USA) robotic system. For multiportal RATS (mRATS) procedures a four-arm approach with four ports and one assistant port (AirSeal, CONMED, FL, USA) for carbon dioxide insufflation were used. Ports were placed as depicted in Figure 1. With increased experience we started to place the assistant port in proximity to the lateral non-camera port to allow these two ports to be joined by skin incision at end of procedure upon lobe retrieval. For uniportal RATS (uRATS) cases, a three-arm approach through a 6–7 cm single incision placed anterolaterally in the 6th or 7th intercostal space (IC) with a soft tissue retractor (Alexis O Wound Protector-Retractor Small, Applied Medical Inc., Rancho Santa Margarita, CA, USA) and no carbon dioxide was used. uVATS were performed through a single 3–4 cm anterolateral incision in IC 4 or 5 with a soft tissue retractor (Alexis O Wound Protector-Retractor Extra Small, Applied Medical Inc.) using a 5 mm thoracoscope, (ENDOEYE, Olympus, Hamburg, Germany) without carbon dioxide insufflation. Hilar, interlobar and mediastinal lymph node stations were systematically dissected and sampled in both techniques. Lymph nodes were categorized by station and not counted by number of separate lymph nodes.
Standard postoperative routine for both groups entailed extubating the patient in the operation room as soon as possible after completed surgery. Patients spent the first 3–4 postoperative hours at an intermediate-intensity postoperative recovery unit and were then referred to an ordinary cardiothoracic ward. A bedside chest X-ray was performed on the morning of postoperative day one and the chest drain was removed if absence of air leak or excessive fluid output was confirmed. Patients underwent additional chest X-ray approximately 4–6 hours after CT removal and were discharged if mobilised and generally well on postoperative day two. Patients were advised to contact the cardiothoracic department if unforeseen problems such as fever, respiratory distress or signs of subcutaneous emphysema arose but were otherwise followed-up by their referring pulmonologist at about four to six weeks postoperatively.
Ethics statement
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the national ethical committee (Swedish Ethical Review Authority, registration number 2024-00005-02) and individual consent for this retrospective analysis was waived.
Statistical analysis
Values are presented as mean and standard deviation (SD) for normally distributed continuous variables and median and [interquartile range] for non-normally distributed continuous variables. Categorical variables are presented as frequencies and (percentage). Student’s t-test was used for comparisons of normally distributed continuous variables. Wilcoxon Rank Sum test was used for comparison of non-normally distributed continuous variables. Chi-squared test was used for categorical data with more than two categories. Fisher’s exact test was used for comparison of frequencies with respect to dichotomous variables. For duration of peri- and operative procedures, a piecewise linear model was used to evaluate OP time, OR time and console time over the study period. A piecewise linear model divides data into segments using specific cutoff points (knots) and fits linear relationships within each segment. This method is useful for analyzing operation time when the relationship between predictors and operation time shifts across different phases (9). Reduction in OP time over the course of consecutive cases within groups was presented as mean (95% CI). Cumulative sum (CUSUM) charts for OP time, OR time and console time were created as an additional analysis to the piecewise calculations. Analyses were performed using R Statistical Software (v4.1.2; R Core Team 2021) and SAS Software v9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Patient disposition
A total of 83 consecutive RATS and 61 consecutive uVATS cases were initially eligible for analysis. One patient (1.2%) with sarcoidosis in the RATS group was excluded due to conversion to open surgery because of pulmonary artery bleeding and one patient in the uVATS group (1.6%) was excluded due to early conversion to open surgery because of unexpected intraoperative finding of advanced tumour ingrowth to the parietal pleura. Hence 82 RATS and 60 uVATS cases were included in the analyses. Preoperative demographics were similar in both groups (Table 1) and preoperative lung function and occurrence of comorbidities did not differ significantly between groups. Preoperative tumour size as determined as the largest diameter in any dimension measured from computed tomography scan were larger among patients in the uVATS group compared to the RATS group but there were no significant differences in clinical tumour-node-metastasis (TNM) stage (Table 1). Procedures performed are listed in Table 2. There was no difference in distribution of procedures in the RATS and uVATS groups (P=0.11). Of the RATS cases, 12 (14.6%) were performed by a single incision as uRATS. Procedures of uRATS were right upper lobectomy (n=2), right middle lobectomy (n=3) and right lower lobectomy (n=7). All other RATS cases were performed using four ports and an AirSeal assistant port. In one RATS procedure, damage to the main right bronchus during dissection was diagnosed intraoperatively and repaired during surgery without the need for conversion to VATS or open surgery. In one mRATS case, one of the robotic arms was dysfunctional and the procedure continued with three arms.
Table 1
| Preoperative characteristics | RATS, N=82 | uVATS, N=60 | P value |
|---|---|---|---|
| Age (years) | 72 [64–76] | 73 [65–78] | 0.61ⴕ |
| Gender (male) | 29 (35.4) | 21 (35.0) | <0.99§ |
| Height (cm) | 168.29 (8.56) | 168.72 (9.55) | 0.78‡ |
| Weight (kg) | 75.00 [64.00–88.00] | 72.50 [61.00–84.00] | 0.35ⴕ |
| BMI (kg/m2) | 26.25 [23.42–30.45] | 25.20 [22.28–27.88] | 0.17ⴕ |
| FEV1 (L) | 2.20 [1.90–2.60] (n=80) | 2.10 [1.55–2.65] (n=59) | 0.22ⴕ |
| FEV1% predicted (%) | 92.10 (19.71) (n=80) | 88.54 (21.78) (n=59) | 0.32‡ |
| DLCO (%) | 80.67 (14.28) (n=79) | 77.78 (18.00) (n=50) | 0.31‡ |
| FVC (L) | 3.10 [2.70–3.80] (n=79) | 3.10 [2.60–3.80] (n=59) | 0.52ⴕ |
| VO2 max (mL/min/kg) | 17.39 (2.70) (n=18) | 17.84 (3.18) (n=19) | 0.65‡ |
| Exercise test (maximal watts) | 104.00 [90.00–127.00] (n=49) | 92.00 [81.00–132.00] (n=39) | 0.13ⴕ |
| Coronary disease | 8 (9.8) | 2 (3.3) | 0.25§ |
| Heart failure | 6 (7.3) | 2 (3.3) | 0.52§ |
| Previous heart surgery | 1 (1.2) | 0 (0.0) | >0.99§ |
| Diabetes | 13 (15.9) | 11 (18.3) | 0.87§ |
| History of CVI | 6 (7.3) | 4 (6.7) | >0.99§ |
| Renal failure | 3 (3.7) | 1 (1.7) | 0.85§ |
| Hypertension | 52 (63.4) | 32 (53.3) | 0.30§ |
| Arythmia | 8 (9.8) | 8 (13.3) | 0.69§ |
| Smoking status | 0.46§ | ||
| Non-smoker | 21 (25.6) | 21 (35.0) | |
| Former smoker >6 weeks | 44 (53.7) | 27 (45.0) | |
| Current smoker | 17 (20.7) | 12 (20.0) | |
| Preoperative tumour size (mm) | 18 [14.00–28.25] | 25 [18.00–39.00] | 0.002ⴕ |
| Clinical tumour stage | n=82 | n=59 | 0.03§ |
| cT1a | 8 (9.8) | 1 (1.7) | |
| cT1b | 38 (46.3) | 21 (35.6) | |
| cT1c | 11 (13.4) | 11 (18.6) | |
| cT2a | 7 (8.5) | 13 (22.0) | |
| cT2b | 3 (3.7) | 5 (8.5) | |
| cT3 | 15 (18.3) | 7 (11.9) | |
| cT4 | 0 | 1 (1.7) | |
| Clinical nodal stage | 0.70§ | ||
| cN0 | 75 (91.5) | 51 (86.4) | |
| cN1 | 6 (7.3) | 7 (11.9) | |
| cN2 | 1 (1.2) | 1 (1.7) | |
| Clinical TNM stage | 0.23§ | ||
| cIA1 | 8 (9.8) | 2 (3.4) | |
| cIA2 | 34 (41.5) | 17 (28.8) | |
| cIA3 | 10 (12.2) | 11 (18.6) | |
| cIB | 7 (8.5) | 10 (17.0) | |
| cIIA | 3 (3.7) | 5 (8.5) | |
| cIIB | 17 (20.7) | 11 (18.6) | |
| cIIIA | 3 (3.7) | 3 (5.1) |
Values are mean (SD) for normally distributed continuous variables and median [interquartile range] for non-normally distributed continuous variables. Categorical variables are presented as frequency (percentage). ⴕ, P value for Wilcoxon Rank Sum test; ‡, P value for Student’s t-test; §, P value for Chi-squared test or Fisher’s exact test. BMI, body mass index; CVI, cerebro-vascular insult; DLCO, diffusing capacity for carbon monoxide; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; VO2, oxygen consumption; RATS, robotic-assisted thoracic surgery; TNM, tumour-node-metastasis; uVATS, uniportal video-assisted thoracic surgery.
Table 2
| Intraoperative and postoperative variables | RATS, N=82 | uVATS, N=60 | P value |
|---|---|---|---|
| Procedures | 0.11§ | ||
| Right upper lobectomy | 26 (31.7) | 17 (28.3) | |
| Middle lobectomy | 8 (9.8) | 2 (3.3) | |
| Right lower lobectomy | 21 (25.6) | 10 (16.7) | |
| Left upper lobectomy | 16 (19.5) | 11 (18.3) | |
| Left lower lobectomy | 9 (11.0) | 16 (26.7) | |
| Right upper bilobectomy | 0 | 1 (1.7) | |
| Right lower bilobectomy | 2 (2.4) | 3 (5.0) | |
| Number of patients with lymph nodes sampled | n=82 | n=59 | |
| Number of lymph node stations sampled | 5 [4–5] | 4 [4–5] | 0.77 ⴕ |
| Number of patients with subcarinal lymph nodes sampled | 80 (97.6) | 57 (96.6) | 0.11§ |
| Perioperative results | |||
| Operation time (min) | 136.00 [114.50–152.25] | 100.00 [84.25–120.00] | <0.001ⴕ |
| Operation room time (min) | 270.00 [240.25–297.00] | 245.00 [224.00–278.50] | 0.009ⴕ |
| Preparation time (min) | 59.50 [52.25–69.75] | 57.50 [47.00–66.25] | 0.16ⴕ |
| Reoperation | 1 (1.3) | 5 (8.3) | 0.12§ |
| Console time (min) | 101.50 [83.00–116.75] | – | – |
| Chest tube (days) | 1.00 [1.00–1.00] | 1.00 [1.00–3.75] | 0.01ⴕ |
| Transfusion of blood | 2 (2.4) | 1 (1.7) | 0.75§ |
| Pneumothorax postop | 0 (0.0) | 2 (3.3) | 0.36§ |
| Arythmia postop | 3 (3.8) | 3 (5.0) | >0.99§ |
| Pneumonia postop | 0 (0.0) | 2 (3.3) | 0.36§ |
| Pulmonary embolism postop | 1 (1.3) | 0 (0.0) | >0.99§ |
| Phrenic paralysis postop | 1 (1.3) | 0 (0.0) | >0.99§ |
| Length of stay postop (days) | 2.00 [2.00–3.00] | 2.00 [2.00–5.00] | 0.07ⴕ |
| Discharge to other unit/hospital | 6 (7.6) | 2 (3.3) | 0.48§ |
| Histology, pathology report | – | ||
| AIS/MIA | 2 (2.4) | 0 | |
| Adenocarcinoma | 51 (62.2) | 41 (68.3) | |
| Squamous cell carcinoma | 7 (8.5) | 4 (6.7) | |
| Other pulmonary malignancy | 18 (22.0) | 7 (11.7) | |
| Benign | 0 | 2 (3.3) | |
| Metastasis | 4 (4.9) | 6 (10) | |
| Pathological tumour stage | n=78 | n=52 | 0.18§ |
| pT(is) | 2 (2.6) | 0 | |
| pT1a | 7 (9.0) | 1 (1.9) | |
| pT1b | 38 (48.7) | 20 (38.5) | |
| pT1c | 10 (12.8) | 11 (21.2) | |
| pT2a | 10 (12.8) | 12 (23.1) | |
| pT2b | 3 (3.9) | 3 (5.8) | |
| pT3 | 8 (10.3) | 4 (7.7) | |
| pT4 | 0 | 1 (1.9) | |
| Pathological nodal stage | n=78 | n=52 | 0.01§ |
| pN0 | 73 (93.6) | 43 (82.7) | |
| pN1 | 4 (5.1) | 2 (3.9) | |
| pN2 | 1 (1.3) | 7 (13.5) | |
| Pathological TNM stage | n=78 | n=52 | 0.03§ |
| pT(is)N0M0 | 2 (2.6) | 0 | |
| pIA1 | 6 (7.7) | 1 (1.9) | |
| pIA2 | 38 (48.7) | 17 (32.7) | |
| pIA3 | 8 (10.3) | 7 (13.5) | |
| pIB | 9 (11.5) | 12 (23.1) | |
| pIIA | 3 (3.9) | 2 (2.9) | |
| pIIB | 10 (12.8) | 5 (9.6) | |
| pIIIA | 2 (2.6) | 8 (15.4) |
Values are mean (SD) for normally distributed continuous variables and median [interquartile range] for non-normally distributed continuous variables. Categorical variables are presented as frequency (percentage). ⴕ, P value for Wilcoxon Rank Sum test; §, P value for Chi-squared test or Fisher’s exact test. AIS, adenocarcinoma in situ; MIA, minimally invasive adenocarcinoma; RATS, robotic-assisted thoracic surgery; TNM, tumour-node-metastasis; uVATS, uniportal video-assisted thoracic surgery.
Perioperative results
The number of days with a CT in situ was significantly shorter in the RATS group [1.00 (1.00–1.00)] than in the uVATS group [1.00 (1.00–3.75); P=0.01]. Length of stay (LOS) in the RATS group was two days [2.00–3.00] compared with two days [2.00–5.00] in the uVATS group (P=0.07). In the RATS group, six patients (7.6%) were discharged to other institution compared to two patients (3.3%) in the uVATS group (P=0.48), but none of patients in either group were discharged with a CT in situ.
There were no significant differences between groups in the consumption of blood transfusion (Table 2). One patient in the RATS group underwent re-operation by uVATS due to prolonged air leak. Five patients in the uVATS group underwent re-operation due to prolonged air leak, but the inter-group difference did not reach statistical significance. Re-operations in the uVATS group were performed by uVATS and included suturing or stapling of parenchyma. No central bronchial tears or fistulas were encountered. One patient in the RATS group suffered from postoperative phrenic paralysis. Postoperative histology from pathological reports are presented in Table 2. Nodal upstaging was present in 2 (2.6%) of the patients in the RATS group compared to 7 (13.5%) in the uVATS group (P=0.03).
Time in operating room and learning curve
OP time and OR time were longer in the RATS group than in the uVATS group (Table 2); preparation time was similar in both groups.
For OP time, using a piecewise linear model with a break point in October 2022, there was a significant decrease in OP time for the RATS group, with mean reduction of 5.4 min (95% CI: −7.7 to −3.1) per month from start of the surgeries to October 2022 (P<0.001). After October 2022 (case No. 44) no significant further change in OP time was recorded [mean alteration 0.16 minutes (95% CI: −2.0 to 2.3); P=0.88].
For OR time using a piecewise linear model with a break point in October 2022, there was a significant decrease in OR time for the RATS group, with mean reduction of 5.4 min (95% CI: −8.5 to −2.3) per month from start of the surgeries to October 2022 (P<0.001). After October 2022 (case no. 44) no significant further change in OR time was recorded [mean alteration −0.5 minutes (95% CI: −3.4 to 2.3), P=0.70].
In contrast to experience in the RATS group no reduction of OP or OR times was apparent in the uVATS group during the study period.
Time in console for RATS cases was available for 80 cases and was 101.50 [83.00–116.75] minutes. There was a significant decrease in console time for the RATS group, with mean reduction of 1.4 minutes per month (95% CI: −2.3 to −0.5) during the surgery calendar period (P=0.002). CUSUM charts for RATS and uVATS of OP time (Figure 2), OR time (Figure 3) and RATS consol time (Figure 4) showed similar learning curves as the piecewise linear model calculations. In the CUSUM charts, the process initially displays an upward trend in cumulative deviations, indicating that the surgery times were consistently longer than the target value during these cases. After crossing the upper decision boundary (UDB), the process gradually stabilized as shown by the downward trend and returned to near-zero levels, indicating improved efficiency over time with duration of procedures starting to stabilize around the centre line after the first 44 cases. For time in the console, the learning curve seemed to stabilise after approximately 25 cases.



Discussion
Key findings
In our experience of implementing RATS with limited access to the robot surgery system, we found a stabilization of operating time after approximately 44 cases. Compared with uVATS we found that CTs were retained for a shorter time in the RATS group but there was no difference in LOS or in prevalence of postoperative complications.
Strengths and limitations
The principal strength of this study is that it describes a real-world situation for a single surgeon taking on a new technology and implementing it on a regular basis. However, several limitations of the present study must be acknowledged. The sample size of this retrospective analysis of consecutive RATS and uVATS series is small and it is likely that the study is underpowered to detect clinically relevant differences. We did not perform a power analysis, but ended inclusion at a time where we perceived the RATS program to be well implemented. Nevertheless, the number of included RATS patients exceeds the number of patients reported by other authors as necessary to reach the peak of learning curve (10-13).
Our chosen metric for describing the learning curve was based on duration of procedures and not on complications or rate of conversion. We believe that the procedural time, together with separate account for intraoperative problems, accurately reflect the process of implementation and skill acquisition. We wished to include only patients in whom we could analyse operation times and postoperative outcomes for a full minimally-invasive procedure and therefore chose to exclude from analysis the two patients who were converted to open surgery, one in each of the study groups. OP time for the patient who was converted to open surgery in the RATS group was 308 minutes. OP time for the patient who was converted to open surgery in the uVATS group was 147 minutes. It is unlikely that an intention-to-treat analysis including these two cases of conversion would have had a substantially different outcome from our analysis with these two patients excluded but a risk-adjusted CUSUM analysis including these two cases would have been an alternative to the present analysis.
Another possible limitation of our research is that the single surgeon perspective could make it difficult to extrapolate our results to a team of several surgeons taking on the robotic technique. On the other hand, we believe that the study offers a realistic description of the implementation of a robotic surgical program. Senior surgeons are often the first ones to adopt a new technique before proctoring junior colleagues. There were no training of residents or fellows in the console during the period of implementation. We chose to report the learning curve of RATS for an experienced uVATS surgeon instead of comparing our first RATS series to the first uVATS cases of the same surgeon to reduce biases arising from comparing operations performed in different years.
Possible selection biases in the assignment of patients to RATS or uVATS were not adjusted for, which may affect the external validity of the study. However, the baseline characteristics in terms of cardiorespiratory function and comorbidities in both groups were well balanced and there was only a small amount of missing data. Numbers of standard spirometry measurements were missing for three patients in the RATS group and 10 patients in the uVATS group patients in the preoperative evaluation. These cases were accepted for surgery after assurance of sufficient respiratory function by the referring pulmonologist. Ergospirometry and exercise testing were only performed for patients at risk of severe perioperative respiratory or cardiac dysfunction and hence a greater number of missing data were found for these variables. For console time data was missing for two patients due to technical failure of log program and these times could not be recovered.
Robotic-assisted platforms are being increasingly widely adopted in several surgical fields as the technology advances and availability improves. Randomized studies comparing RATS to conventional VATS have repeatedly showed similar short-term results for both techniques, but an increased number of lymph nodes dissected with RATS compared to VATS (14-16). However, these studies failed to show a significant upstaging of pathological TNM stage, possibly due to thorough preoperative clinical TNM stage evaluation. This is consistent with our experience that one of the greatest advantages of RATS is enhanced visualization, enhancing not only lymph node dissection but dissection overall. In the present study we found no inter-group difference regarding number of lymph node stations sampled. Pathological lymph node stage (pN) were higher in the uVATS group (P=0.01) and nodal upstaging was present in more patients in the uVATS group than in the RATS group (P=0.03). Although the clinical TNM stages were not significantly different in groups, these findings could reflect a selection bias of less complicated cases selected for RATS in the early period of implementation.
In parallel with the evolution of RATS for lung surgery, there has been a development of VATS techniques towards using a single port approach—uVATS (17). The benefits of potentially reduced postoperative pain by fewer thoracoscopic ports associated with uVATS, might also be delivered by mRATS, by virtue of less traumatic movements as the robotic port pivots in the middle of the intercostal spaces, leading to reduced mechanical stress on costae and adjacent nerves. A randomized study investigating quality of life parameters showed less perceived postoperative pain in a mRATS group compared to a uVATS group of patients at four weeks after surgery (18), but no significant differences were noted in a battery of quality of life indices at any time during postoperative follow-up. As we here compare two minimally-invasive procedures, we did not expect any large differences in postoperative pain between groups. In our department we routinely use epidural analgesia for minimally-invasive procedures but switched to intercostal block for most VATS cases around about the start of the study period. Epidural analgesia was retained for RATS cases, as more ports were used in RATS than VATS and the technique was ipso facto new to us. Although our dataset did not support having a specific endpoint of patient-perceived postoperative pain, our experience with RATS leads us to believe that it is possible to stop using epidural analgesia when performing RATS procedures.
Explanation of findings
The time in console seemed to be more evenly distributed over the study period while time for docking varied during the learning curve. This underlines our experience of the importance of port placement and docking. This part of the operation is important and should not be neglected as proper placement of ports are crucial for effective time in console.
We gradually extended inclusion of patients for RATS procedures. Without having a clear break point, our belief is that we after the approximately first 25 cases, deemed all cases which were eligible to uVATS also to be eligible to RATS. Even though no predefined selection criteria of first RATS cases were set, there were no RATS surgery performed on patients who had prior ipsilateral lung surgery. The presence of pleural adhesions can be difficult to predict and there were several cases of severe pleural adhesions in the RATS group. Both cases with prior cardiac surgery and history of radiation to breast or chest for other reason than lung malignancy were included in the RATS group. Severe pleural adhesions remain a challenge but can be taken down with RATS. The study subjects were not matched to controls, but there were no significant differences in preoperative characteristics including lung function and comorbidities. In the RATS group we did have one case with intraoperative damage to the bronchus. High pressures of carbon dioxide insufflation cause the membranous part of the bronchus to collapse, making the anatomical outline of the bronchus more difficult to discern at dissection.
The loss of haptic sensation in RATS is well-known, but a larger challenge at start of the learning curve is handling low-grade bleeding without the option of manual suctioning. (We did not have access to a wristed suction irrigator controlled at the surgeon console. Suction of the operative field was managed by the assistant through the assistant port). Therefore, there is a need for meticulous haemostasis during the whole procedure. We did not record bleeding volumes in this study. However, no difference in blood transfusions given during the perioperative period was found. Our results support the findings of other authors of low incidence of catastrophic bleeding events during RATS procedures (19,20).
uVATS for anatomical pulmonary resections has become an additional minimally-invasive technique (21). Likewise, single incision robotic-assisted technique has emerged as a feasible option (22-24). The uRATS technique has been reported to be potentially more demanding (25), but to be as feasible and safe as conventional mRATS (26,27). uRATS has been part of our RATS program and used by us on a regular basis as a complement to the mRATS technique. Indications for choosing a uRATS approach rather than a mRATS approach–and when not to–are questions still needing to be further addressed.
Other studies on the implementation of RATS programs align with our experience, reporting steep learning curves with a peak at approximately 32–60 cases (10-13): similar findings are reported for the learning curves of uVATS (28). We believe that a great part of the learning curve of RATS technique is accounted for by learning proper port placement, exposure of the surgical field and plan for angulation while stapling. The need for this is accentuated in uRATS procedures in which experience in uVATS for surgeon and assistant is likely to be facilitative.
A RATS lobectomy costs more than a uVATS lobectomy (29,30) and it is not clear that savings in other areas can recoup that extra cost. Programs for enhanced recovery after surgery are routine for all types of minimally-invasive procedures. Days with CT in situ and length of hospital stay are already short for these kinds of procedures and it may not be feasible to reduce them further sufficiently to show savings in cost-effectiveness analysis. Some material costs can probably be decreased. For example, at the start of our program we routinely used disposable energy devices but found with experience that a reusable bipolar forceps was often sufficient for dissection. Our in-series development of port placement in which the assistant port is placed near and in the same IC as one of the other ports is now our standard technique in mRATS procedures. This approach is, in our opinion, less traumatic than enlarging only the incision for the assistant port for lobe retrieval as the incision needs to be large enough not to damage costae and leaves the patient with four incisions instead of five.
Conclusions
This study shows that the implementation of a robotic-assisted program for anatomical lung resections is feasible and can safely be introduced even with limited access to the robotic platform. It further shows that the results are similar with the otherwise standard uVATS approach for lobectomies and bilobectomies. The initial learning curve for a single surgeon experienced in uVATS was reached at 44 cases after which no further time reduction was seen in duration of procedures. The CUSUM charts showed a similar trend. Further studies of cost-effectiveness and long-term results of RATS for anatomical pulmonary resections in comparison to uVATS can be of value for surgeons to encourage expansion of RATS programmes in routine clinical settings.
Acknowledgments
Statistical analysis and counselling were provided by Aldina Pivodic and Hussnain Khalid, APNC Sweden AB. Editorial support was provided by Peter Hughes, Hughes associates, Oxford, UK.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1182/rc
Data Sharing Statement: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1182/dss
Peer Review File: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1182/prf
Funding: This work was supported by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1182/coif). A.W. reports receiving honoraria from CONMED Corporation for given lecture. G.D. reports receipt of research grants from Astellas and Abbot, XVIVO AB and being a board member of XVIVO AB. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the national ethical committee (Swedish Ethical Review Authority, registration number 2024-00005-02) and individual consent for this retrospective analysis was waived.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Detterbeck F, Molins L. Video-assisted thoracic surgery and open chest surgery in lung cancer treatment: present and future. J Vis Surg 2016;2:173. [Crossref] [PubMed]
- Higuchi M, Yaginuma H, Yonechi A, et al. Long-term outcomes after video-assisted thoracic surgery (VATS) lobectomy versus lobectomy via open thoracotomy for clinical stage IA non-small cell lung cancer. J Cardiothorac Surg 2014;9:88. [Crossref] [PubMed]
- Hireche K, Lounes Y, Bacri C, et al. VATS versus Open Lobectomy following Induction Therapy for Stage III NSCLC: A Propensity Score-Matched Analysis. Cancers (Basel) 2023;15:414. [Crossref] [PubMed]
- Lim E, Batchelor TJP, Dunning J, et al. Video-Assisted Thoracoscopic or Open Lobectomy in Early-Stage Lung Cancer. NEJM Evid 2022;1:EVIDoa2100016.
- Melfi FM, Menconi GF, Mariani AM, et al. Early experience with robotic technology for thoracoscopic surgery. Eur J Cardiothorac Surg 2002;21:864-8. [Crossref] [PubMed]
- Pan H, Zhang J, Tian Y, et al. Short- and long-term outcomes of robotic-assisted versus video-assisted thoracoscopic lobectomy in non-small cell lung cancer patients aged 35 years or younger: a real-world study with propensity score-matched analysis. J Cancer Res Clin Oncol 2023;149:9947-58. [Crossref] [PubMed]
- Mattioni G, Palleschi A, Mendogni P, et al. Approaches and outcomes of Robotic-Assisted Thoracic Surgery (RATS) for lung cancer: a narrative review. J Robot Surg 2023;17:797-809. [Crossref] [PubMed]
- Kent MS, Hartwig MG, Vallières E, et al. Pulmonary Open, Robotic, and Thoracoscopic Lobectomy (PORTaL) Study: Survival Analysis of 6646 Cases. Ann Surg 2023;277:1002-9. [Crossref] [PubMed]
- Tibshirani R. Regression Shrinkage and Selection via the Lasso. J R Statist Soc. B 1996;58:267-88. [Crossref]
- Andersson SE, Ilonen IK, Pälli OH, et al. Learning curve in robotic-assisted lobectomy for non-small cell lung cancer is not steep after experience in video-assisted lobectomy; single-surgeon experience using cumulative sum analysis. Cancer Treat Res Commun 2021;27:100362. [Crossref] [PubMed]
- Gómez-Hernández MT, Fuentes MG, Novoa NM, et al. The robotic surgery learning curve of a surgeon experienced in video-assisted thoracoscopic surgery compared with his own video-assisted thoracoscopic surgery learning curve for anatomical lung resections. Eur J Cardiothorac Surg 2022;61:289-96. [Crossref] [PubMed]
- Streit A, Le Reun C, Lampridis S, et al. Learning curve of robotic surgery for lung cancer: analysis for two surgeons during the COVID-19 pandemic. Gen Thorac Cardiovasc Surg 2024;72:240-6. [Crossref] [PubMed]
- Paglialunga PL, Molins L, Guzmán R, et al. Robotic Lobectomy Learning Curve Has Better Clinical Outcomes than Videothoracoscopic Lobectomy. J Clin Med 2024;13:1653. [Crossref] [PubMed]
- Veronesi G, Abbas AE, Muriana P, et al. Perioperative Outcome of Robotic Approach Versus Manual Videothoracoscopic Major Resection in Patients Affected by Early Lung Cancer: Results of a Randomized Multicentric Study (ROMAN Study). Front Oncol 2021;11:726408. [Crossref] [PubMed]
- Jin R, Zheng Y, Yuan Y, et al. Robotic-assisted Versus Video-assisted Thoracoscopic Lobectomy: Short-term Results of a Randomized Clinical Trial (RVlob Trial). Ann Surg 2022;275:295-302. [Crossref] [PubMed]
- Patel YS, Baste JM, Shargall Y, et al. Robotic Lobectomy Is Cost-effective and Provides Comparable Health Utility Scores to Video-assisted Lobectomy: Early Results of the RAVAL Trial. Ann Surg 2023;278:841-9. [Crossref] [PubMed]
- Li Y, Dai T. Meta-analysis comparing the perioperative efficacy of single-port versus two and multi-port video-assisted thoracoscopic surgical anatomical lung resection for lung cancer. Medicine (Baltimore) 2023;102:e32636. [Crossref] [PubMed]
- Jin R, Zhang Z, Zheng Y, et al. Health-Related Quality of Life Following Robotic-Assisted or Video-Assisted Lobectomy in Patients With Non-Small Cell Lung Cancer: Results From the RVlob Randomized Clinical Trial. Chest 2023;163:1576-88. [Crossref] [PubMed]
- Cao C, Cerfolio RJ, Louie BE, et al. Incidence, management and outcomes of intraoperative catastrophes during robotic pulmonary resection. Ann Thorac Surg 2019;108:1498-504. [Crossref] [PubMed]
- Baig MZ, Razi SS, Agyabeng-Dadzie K, et al. Robotic-assisted thoracoscopic surgery demonstrates a lower rate of conversion to thoracotomy than video-assisted thoracoscopic surgery for complex lobectomies. Eur J Cardiothorac Surg 2022;62:ezac281. [Crossref] [PubMed]
- Gonzalez D, Paradela M, Garcia J, et al. Single-port video-assisted thoracoscopic lobectomy. Interact Cardiovasc Thorac Surg 2011;12:514-5. [Crossref] [PubMed]
- Yang Y, Song L, Huang J, et al. A uniportal right upper lobectomy by three-arm robotic-assisted thoracoscopic surgery using the da Vinci (Xi) Surgical System in the treatment of early-stage lung cancer. Transl Lung Cancer Res 2021;10:1571-5. [Crossref] [PubMed]
- Gonzalez-Rivas D, Bosinceanu M, Motas N, et al. Uniportal robotic-assisted thoracic surgery for lung resections. Eur J Cardiothorac Surg 2022;62:ezac410. [Crossref] [PubMed]
- Gonzalez-Rivas D, Bosinceanu M, Manolache V, et al. Uniportal fully robotic-assisted major pulmonary resections. Ann Cardiothorac Surg 2023;12:52-61. [Crossref] [PubMed]
- Palleschi A, Mattioni G, Mendogni P, et al. A real-world experience of transition to robotic-assisted thoracic surgery (RATS) for lung resections. Front Surg 2023;10:1127627. [Crossref] [PubMed]
- Manolache V, Motas N, Bosinceanu ML, et al. Comparison of uniportal robotic-assisted thoracic surgery pulmonary anatomic resections with multiport robotic-assisted thoracic surgery: a multicenter study of the European experience. Ann Cardiothorac Surg 2023;12:102-9. [Crossref] [PubMed]
- Mercadante E, Martucci N, De Luca G, et al. Early experience with uniportal robotic thoracic surgery lobectomy. Front Surg 2022;9:1005860. [Crossref] [PubMed]
- Laven IEWG, Daemen JHT, Franssen AJPM, et al. Uniportal video-assisted thoracoscopic surgery for lobectomy: the learning curve. Interdiscip Cardiovasc Thorac Surg 2023;37:ivad135. [Crossref] [PubMed]
- Heiden BT, Mitchell JD, Rome E, et al. Cost-Effectiveness Analysis of Robotic-assisted Lobectomy for Non-Small Cell Lung Cancer. Ann Thorac Surg 2022;114:265-72. [Crossref] [PubMed]
- Sadri H, Fung-Kee-Fung M, Shayegan B, et al. A systematic review of full economic evaluations of robotic-assisted surgery in thoracic and abdominopelvic procedures. J Robot Surg 2023;17:2671-85. [Crossref] [PubMed]


