
Lung cancer in young individuals; risk factors and epidemiology
Introduction
Lung cancer presented the worst morbidity and mortality statistics among all diseases worldwide in 2022, with approximately 2.5 million new incidences and 1.8 million deaths. Furthermore, lung cancer ranks first among men and second among women in terms of both incidence and mortality (1). Despite the development of medical treatments that have led to a gradual decrease in mortality rates in recent years, lung cancer continues to be the primary cause of cancer-associated deaths globally (2). While the median age at diagnosis of lung cancer is 71 years, 6.0% of patients are diagnosed at less than 55 years. Lung cancer in the under 45 and 35 population are relatively uncommon, and approximately 1.1% and 0.2% of all patients with lung cancer, respectively (3). A study from the United States reported that the incidence of lung cancer in the adolescents and young adults (AYA) population decreases in the long term. Specifically, the incidence of lung cancer in the AYA population dropped down from 0.9 per 100,000 in 1975 to 0.4 per 100,000 in 2010 in female patients, and a similar pattern was observed in male patients, from 1.1 per 100,000 in 1975 to 0.4 per 100,000 in 2010 (4). While these statistics provided important information, we should treat their information carefully because several factors have changed, such as improved diagnostic techniques, changes in reporting methods, and risk factor exposure. Interestingly, despite its rarity in younger populations, the impact of lung cancer on patient prognosis is high. Despite being less common in younger individuals. The Cancer Statistics 2022 and the Surveillance, Epidemiology, and End Results Program 22 database provides demographics that indicate that approximately 14,000 individuals under 45 years of age per year are dying from lung cancer in the United States (2,3). In addition to poor prognosis, the unique characteristics of lung cancer in younger patients, including clinical and pathological features and genomic variations, make us aware of the importance of innovative research in this area (5-12). Considering this background, many studies have focused on the unique characteristics of lung cancer, especially non-small cell lung cancer (NSCLC). Although recent research has provided valuable insights into the internal predisposition to NSCLC in younger patients, few studies have focused on external predispositions to NSCLC in young patients. In this review, we summarize the characteristics of cancer in younger generations, including its clinical, pathological, and genomic status. Furthermore, we investigated the causes of early onset, based on internal and external predispositions, in young patients with lung cancer.
At the outset, we wish to state an important issue in this research area, which is cut-off for defining individuals as “young”. “AYA” refers to one of the major classifications for a generation and typically indicates individuals aged 15 to 39 years at the time of their initial cancer diagnosis (13). However, the cut-off age for the younger generation remains an issue of ongoing discussion and has varied considerably across studies (5,8-12,14-17). Notably, approximately 40% studies examined using the conventional 40-year threshold (15,18-23). However, other studies have used diverse cut-offs ranging from 30 to 50 years (11,16,17,24-30), which indicates a lack of consensus on this issue. In addition to age as a continuous variable, risk factors such as clinical and pathological characteristics and genetic profiles associated with lung cancer may change across different age groups, which makes it challenging to establish a universally accepted cutoff. Even when the age range of the AYA generation has been traditionally defined as 15–39 years, notable differences have been observed in previous studies (5,8-12,14-17). Indeed, differences have been observed in the characteristics of patients with lung cancer, including stage, pathological subtype, and prognosis, between cohorts under 30 and between 30 and 39 years of age (7). Other studies with threshold set at 50 years of age, the incidence of genetic alterations was significantly higher in young patients (8,11). Although the age threshold has varied between studies, these findings have highlighted the critical need for age-specific approaches, particularly when studying younger patient populations.
Clinicopathological characteristics
Lung cancer occurs in males more frequently; however, several studies that have focused on the younger generation have indicated a reversal of this trend in specific populations (5,7,31-33). Most recent studies reported a higher proportion of young female patients diagnosed with lung cancer than young male patients (5,6,31,34).
Several researches of lung cancer in young individuals had been focused on ground glass opacity (GGO) component. Qu et al. reported 133 of 165 patients had GGO components in surgically resectable patients who aged under 40 years (35), and Ma et al. provided compelling evidence of increasing the number of teenage patients incidentally diagnosed with lung cancer, presenting as GGO because of the coronavirus disease 2019 (COVID-19) pandemic (36). However, because these studies are limited to Asians and not comparative analyses between young and elderly patients, further investigation is needed.
When we focused on the histological subtype, there were differential patterns between the younger generation and other cohorts. Although NSCLC is the most common subtype in all age groups, adenocarcinoma has a distinct prevalence in younger patients (5-8,18,22). Notably, Rich et al. provided compelling evidence for this age-related trend in the English National Lung Cancer Audit. They divided patients with NSCLC into six age categories, ranging from <39 to >80 years, and found that the proportion of adenocarcinoma decreased steadily with age (37). While adenocarcinoma accounted for 48% of lung cancer incidences in patients under 39 years old, adenocarcinoma accounted for 31% of lung cancers in patients over 80 years of age. This age-related pattern raises questions about the novel biological mechanisms that might influence the development of lung cancer at different life stages. When we focused on the subtype and differentiation, while Zhong et al. reported that NSCLC in younger patients frequently diagnosed as well differentiated (23), most of prior investigations have reported equivalent patterns of differentiation and subtype in younger and older patients (9,20,21).
Younger patients with lung cancer often face unique diagnostic challenges. Younger patients are frequently diagnosed with lung cancer in the late phase and present with more severe symptoms and more advanced tumors than older patients (38). Several factors contribute to the delayed diagnosis in younger patients, and lung cancer is often not initially considered when diagnosing lung lesions in younger patients. Furthermore, there may be issues with both healthcare providers and patients. Although chest pain is the most frequent symptom in younger patients (39), young people might dismiss early symptoms because they are too young for serious illnesses such as cancer, and the good physical condition of younger patients may obscure early warning signs, thus making symptoms less apparent. Previous research has revealed that a higher proportion of younger patients are diagnosed with metastatic disease than older patients (4,5,14). Furthermore, the younger generation has few opportunities for radiography or computed tomography; thus, accidental detection during the examination of other diseases is extremely valuable and effective. Awareness-building activities for pediatricians and other specialists, such as general internal medicine specialists, are important.
Molecular biomarkers of lung cancer in the AYA generation
In this section, we focused on the distinction of molecular biomarkers of lung cancer in the AYA generation. Considering the differences in exposure to carcinogens, radiological characteristics, and differences in histological types and their subtypes, it is highly likely that biomarkers are expressed differently (5,8,22). A landmark retrospective study conducted at the Dana-Farber Cancer Institute revealed the frequency and differences in oncogenic genomic alterations between these age groups in 2,237 patients of diverse racial backgrounds. Patients aged 50 or younger had a significantly higher frequency of targetable genotype mutations, such as mutations in epidermal growth factor receptor (EGFR), anaplastic lymphoma kinase (ALK), ROS1, human epidermal growth factor receptor 2 (HER2), and v-Raf murine sarcoma viral oncogene homolog B (BRAF) than did older patients (68% vs. 52%, P<0.01). This statistically significant difference suggests that younger patients with lung adenocarcinoma may benefit from targeted therapies, which improves treatment outcomes (8). Kohsaka et al. reported that Asian patients aged 40 years or younger had a significantly higher frequency of EGFR mutations (46.7% vs. 25%), no patient had EGFR mutations in patients aged 30 years or younger, and most major genotype mutation was HER2 in these individuals (40).
When we focused on somatic EGFR mutations, several conflicting results have been reported in young patients with lung adenocarcinoma (9,10,23,40-42). Three patterns have been reported for frequency. As the former described, Kohsaka et al. reported that higher incidence of EGFR mutations in Asian patients. However, the PIONEER study, focused on Asian patients with lung adenocarcinoma, found no significant age-related correlation with EGFR mutations (42). By contrast, as typified by research on Caucasian populations, several reports have revealed a notably higher incidence of EGFR mutations in older patients (25). Furthermore, the type of EGFR mutation can vary with age. For example, EGFR exon deletion 19 and exon 20 insertions are more common in younger patients, whereas the L858R mutation and de novo T790M are more prevalent in older patients (9,10,23). Two studies by Wu et al. revealed that although common mutations are less frequent, uncommon mutations occur more often in younger patients than in older individuals (11,12). These findings indicate a complex relationship between age and somatic EGFR mutation patterns in lung adenocarcinoma, thus highlighting the need for age-specific considerations in genetic profiling and targeted therapies.
Etiology of lung cancer in young patients
In this section, we describe the risk factors for lung cancer in young individuals with respect to two predispositions: internal and external (Figure 1) (32,33,43-49). Previous studies have reported that lung cancer does not have a hereditary predisposition, and the familial clustering of lung cancer has varied between studies, ranging from 1% to 30% (15,43). Interestingly, only one comparative study has identified notable differences in familial incidence between younger and older patients with lung cancer. Previous research has reported that several germline genetic alterations were associated with hereditary lung cancer, such as mutations in EGFR T790M, TP53, BRCA, HER2, YAP1, CHEK2, and other genes (43). Despite these findings, it is important to note that genetic predispositions cannot fully explain the development of lung cancer in young patients.

The etiology of lung cancer in younger patients may involve a complex predisposition to genetic, environmental, and lifestyle factors. Several external factors have been identified as potential contributors to lung cancer development in younger patients. Canadian research reported that radon, a radioactive noble gas produced naturally through the decay of uranium in rocks and soil, is a risk factor for lung cancer in younger generations (44). In Canada, indoor environments account for approximately 90% of radon exposure, and cumulative indoor radon exposure during childhood has been recognized as a significant risk factor for lung cancer in young Canadian adults. However, it should be noted that this study is highly region specific. Smoking and air pollution, such as PM2.5, remain primary risk factors for lung cancer across all age groups (45). Recently, the mortality rate of lung cancer attributable to PM2.5 has been increasing in Asian countries, and this matter affects both older and young individuals (46). Lung cancer incidences reflect temporal trends in smoking prevalence because cigarette smoking causes >80% of lung cancers in the United States (32). Although this proportion is gradually attenuating as fewer people smoke, 72% of women and 81% of men aged 20 to 49 years who were recently diagnosed with lung cancer had smoking habits (2). However, considering a higher proportion of young female patients diagnosed with lung cancer than young male patients, this sex gap cannot be sufficiently explained by smoking habits. Worldwide, males smoke more frequently than females; however, this gap has narrowed considerably among the white population born in the 1960s and later (31). While smoking remains a risk factor for lung cancer (1,2), previous studies have suggested that other factors also play important roles. Environmental exposure to biomass fuels, arsenic, cannabis, air pollution, and occupation exposure such as asbestos, silica, and diesel may also partially explain the early onset of lung cancer (50,51). Furthermore, the high lung cancer rates among Chinese women are postulated to reflect increased outdoor ambient air pollution and exposure to the burning of solid fuels in households for heating and cooking (33).
These findings suggest that multiple factors may play a significant role in young-onset lung cancer. The next section focuses on viral infections, which have received little attention in young patients with lung cancer thus far.
Viral infection and lung cancer
It has been reported that viral infections contribute to approximately 20–25% of human cancers (47), and cancer-associated viruses include the human papilloma virus (HPV) (48), Epstein-Barr virus (EBV) (52), hepatitis B virus (53), hepatitis C virus (54), and human T-cell leukemia virus type I (55). However, hepatitis B virus and human T-cell leukemia virus type I have not been associated with lung cancer; therefore, in this section, we focus on HPV and EBV.
First, HPV is responsible for an estimated 30% of cancer incidences related to viral infections (47). The presence of HPV in the lungs is well known through the occurrence of respiratory papillomatosis. This uncommon benign condition, typically caused by HPV types 6 and 11, occasionally progresses to malignancy (56,57). Bloodstream and oral transmission have also been mentioned as routes of infection, but there is no established evidence (58). Oral transmission, such as multiple sexual partners and oral-genital contact, has been reported to be a risk factor for adult recurrent respiratory papillomatosis, and other types of high-risk HPV, such as types 16 and 18, may be transmitted through oral transmission (59). Generally, HPV oncoproteins E6 and E7 inactivate tumor suppressor genes such as p53 and retinoblastoma, which eventually cause carcinogenesis (Figure 2) (60). A significant challenge in determining whether HPV is a lung carcinogen is to identify the function of the virus within lung cancer, regardless of whether the same process occurs in other cancers. de Oliveira et al. reported the presence of HPV in patients with lung cancer, particularly in younger patients (48). Their research revealed that HPV was detected in 33% of NSCLCs in patients under 55 years old. This high prevalence suggests a potential causal relationship between HPV infection and lung cancer development in younger populations, which warrants further investigation. If future studies reveal an association between young people with lung cancer and HPV infection, then HPV vaccination has the potential to control the prevalence of lung cancer in young people.
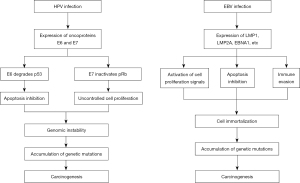
The EBV was the first viral entity directly linked to human cancer development. Subsequent research has established EBV’s crucial involvement of EBV in the onset and progression of two specific malignancies: Burkitt lymphoma and nasopharyngeal carcinoma (61). EBV infects B lymphocytes as well as epithelial cells and NK/T cells, which results in malignant transformation. EBV produces oncoproteins such as LMP1, LMP2A, and EBNA1, which stimulate cell proliferation, apoptosis inhibition, immune evasion, and genetic mutations and eventually lead to carcinogenesis (Figure 2) (52). Although EBV is dormant in the respiratory system, evidence of an association between EBV infection and lung cancer is relatively scarce. Furthermore, although pulmonary lymphoepithelioma-like carcinoma associated with EBV was reported in 1987 (62) and was classified as NSCLC, the World Health Organization’s 2015 histological classification recognizes lymphoepithelioma-like carcinoma as an exceptionally rare subtype of unclassified lung cancer (63). Lymphoepithelioma-like carcinomas tend to have more favorable prognoses than other types of lung cancers. Furthermore, the mean age of patients diagnosed with lymphoepithelioma-like carcinoma is relatively low, and Chen et al. reported lymphoepithelioma-like carcinoma in a patient aged 15 years (64). Previous research has also revealed the presence of EBV in tumor cells in an uncommon lymphoepithelioma-like carcinoma subtype as well as in NSCLC subtypes, including lung squamous cell carcinoma and lung adenocarcinoma (49). This finding led us to hypothesize a potential role of EBV in lung cancer pathogenesis.
With regards to HPV and EBV, although associations have been proposed for both, more reasonable scientific evidence is needed to establish causal relationships. Building an understanding of these relationships has the potential to improve prevention strategies, earlier detection methods, and potentially novel treatment approaches, particularly for younger patients at a risk of lung cancer.
Prognosis and treatment
While some prior investigations have reported equivalent survival rates in younger and older patients with lung cancer, others have reported more favorable outcomes in younger patients. The National Cancer Database (173,856 patients between 2003 and 2009) provides significant insights into this topic, by revealing a statistically significant survival advantage for younger patients (34). Although the specific treatment strategy for lung cancer in young individuals have not reached a consensus, multimodal strategies including surgery and adjuvant chemotherapy are more common than for older patients. Although radical treatment is often the priority in younger patients as previously mentioned, conservative surgery is sometimes preferred. Ma et al. mentioned that sub-lobar resection without lymph node dissection may be an acceptable surgical procedure for teenage patients with GGO-featured lung cancer, because normal lymph node acts important function in anti-tumor immunity (36). However, prospective studies are required to further verify treatment strategy.
The clinical significance of this advantage varied depending on the cancer stage at diagnosis. In stages I and II [according to the International Association for the Study of Lung Cancer (IASLC) seventh-edition staging system], the absolute difference in 5-year overall survival (OS) between younger and older patient groups was 25%. However, the difference in 5-year OS between the age groups narrowed to only 2% for stage IV lung cancer. Although these results suggest that younger patients generally have a better prognosis, differences in cancer stage should be noted moving forward (34). Notably, the minimal difference in survival rates for advanced-stage disease indicates the importance of early detection and intervention, regardless of patient age.
Limitations
This study reviews the epidemiology, clinical, pathological and molecular features of lung cancer in young people. In addition, this article highlights internal and external risk factors for carcinogenesis, providing direction for future approaches to lung cancer in young people. However, this study presents a key paradox as a major limitation that remains unexplained in the current literature; how environmental factors that typically require long-term exposure significantly affect young patients who have had shorter exposure periods compared to older individuals. We propose several ways to resolve this issue. First, we should focus on the interaction between genetic and environmental predispositions. Second, this article focuses on viral infections, which have not been previously recognized as an external risk factor for lung cancer in young people. This is one of the environmental factors, but unlike other environmental factors, young people do not always have shorter exposure periods. However, there has not been sufficient evidence of an association with young age of onset, so this is one of the important issues to be investigated in the future.
Conclusions
Lung cancer in young individuals have unique epidemiological and clinical characteristics such as predominantly in women, nonsmokers with adenocarcinoma histology, higher frequency of targetable genotype mutations, and more advanced stages at diagnosis. The etiology of lung cancer in younger patients may involve a complex predisposition to genetic, environmental, and lifestyle factors. Further research is necessary to evaluate the etiology and to prevent cancer.
Acknowledgments
None.
Footnote
Peer Review File: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-2024-1950/prf
Funding: None.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jtd.amegroups.com/article/view/10.21037/jtd-2024-1950/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Bray F, Laversanne M, Sung H, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2024;74:229-63. [Crossref] [PubMed]
- Siegel RL, Miller KD, Fuchs HE, et al. Cancer statistics, 2022. CA Cancer J Clin 2022;72:7-33. [Crossref] [PubMed]
- National Cancer Institute. Surveillance, epidemiology and end results program. SEER stat fact sheets: lung and bronchus cancer. Available online: https://seer.cancer.gov/statfacts/html/lungb.html
- Thomas A, Chen Y, Yu T, et al. Trends and Characteristics of Young Non-Small Cell Lung Cancer Patients in the United States. Front Oncol 2015;5:113. [Crossref] [PubMed]
- Catania C, Botteri E, Barberis M, et al. Molecular features and clinical outcome of lung malignancies in very young people. Future Oncol 2015;11:1211-21. [Crossref] [PubMed]
- Jemal A, Schafer EJ, Star J, et al. Lung cancer incidence rates in young women and men by state in the United States. Int J Cancer 2025;156:499-504. [Crossref] [PubMed]
- Liu M, Cai X, Yu W, et al. Clinical significance of age at diagnosis among young non-small cell lung cancer patients under 40 years old: a population-based study. Oncotarget 2015;6:44963-70. [Crossref] [PubMed]
- Sacher AG, Dahlberg SE, Heng J, et al. Association Between Younger Age and Targetable Genomic Alterations and Prognosis in Non-Small-Cell Lung Cancer. JAMA Oncol 2016;2:313-20. [Crossref] [PubMed]
- Tanaka K, Hida T, Oya Y, et al. Unique prevalence of oncogenic genetic alterations in young patients with lung adenocarcinoma. Cancer 2017;123:1731-40. [Crossref] [PubMed]
- Chen L, Hu X, Wu H, et al. Unique profiles of targetable genomic alterations and prognosis in young Chinese patients with lung adenocarcinoma. Pathol Res Pract 2019;215:152407. [Crossref] [PubMed]
- Wu SG, Chang YL, Yu CJ, et al. Lung adenocarcinoma patients of young age have lower EGFR mutation rate and poorer efficacy of EGFR tyrosine kinase inhibitors. ERJ Open Res 2017;3:00092-2016. [Crossref] [PubMed]
- Wu SG, Liu YN, Yu CJ, et al. Driver mutations of young lung adenocarcinoma patients with malignant pleural effusion. Genes Chromosomes Cancer 2018;57:513-21. [Crossref] [PubMed]
- Zhang L, Muscat JE, Chinchilli VM, et al. Trends in Cancer Incidence and Mortality in US Adolescents and Young Adults, 2016-2021. Cancers (Basel) 2024;16:3153. [Crossref] [PubMed]
- Hou H, Zhu H, Zhao H, et al. Comprehensive Molecular Characterization of Young Chinese Patients with Lung Adenocarcinoma Identified a Distinctive Genetic Profile. Oncologist 2018;23:1008-15. [Crossref] [PubMed]
- Viñal D, Martínez D, Higuera O, et al. Genomic profiling in non-small-cell lung cancer in young patients. A systematic review. ESMO Open 2021;6:100045. [Crossref] [PubMed]
- Tian P, Liu Y, Zeng H, et al. Unique molecular features and clinical outcomes in young patients with non-small cell lung cancer harboring ALK fusion genes. J Cancer Res Clin Oncol 2020;146:935-44. [Crossref] [PubMed]
- Suidan AM, Roisman L, Belilovski Rozenblum A, et al. Lung Cancer in Young Patients: Higher Rate of Driver Mutations and Brain Involvement, but Better Survival. J Glob Oncol 2019;5:1-8. [Crossref] [PubMed]
- Yang S, Song Z, Cheng G. Genomic alterations and survival in young patients aged under 40 years with completely resected non-small cell lung cancer. Ann Transl Med 2019;7:140. [PubMed]
- Pan X, Lv T, Zhang F, et al. Frequent genomic alterations and better prognosis among young patients with non-small-cell lung cancer aged 40 years or younger. Clin Transl Oncol 2018;20:1168-74. [Crossref] [PubMed]
- Kim L, Kim KH, Yoon YH, et al. Clinicopathologic and molecular characteristics of lung adenocarcinoma arising in young patients. J Korean Med Sci 2012;27:1027-36. [Crossref] [PubMed]
- Ye T, Pan Y, Wang R, et al. Analysis of the molecular and clinicopathologic features of surgically resected lung adenocarcinoma in patients under 40 years old. J Thorac Dis 2014;6:1396-402. [PubMed]
- He CH, Shih JF, Lai SL, et al. Non-small cell lung cancer in the very young: Higher EGFR/ALK mutation proportion than the elder. J Chin Med Assoc 2020;83:461-5. [Crossref] [PubMed]
- Zhong W, Zhao J, Huang K, et al. Comparison of clinicopathological and molecular features between young and old patients with lung cancer. Int J Clin Exp Pathol 2018;11:1031-5. [PubMed]
- Chen Z, Teng X, Zhang J, et al. Molecular features of lung adenocarcinoma in young patients. BMC Cancer 2019;19:777. [Crossref] [PubMed]
- Yang B, Li J, Li F, et al. Comprehensive analysis of age-related somatic mutation profiles in Chinese young lung adenocarcinoma patients. Cancer Med 2019;8:1350-8. [PubMed]
- Wang Y, Chen J, Ding W, et al. Clinical Features and Gene Mutations of Lung Cancer Patients 30 Years of Age or Younger. PLoS One 2015;10:e0136659. [PubMed]
- VandenBussche CJ, Illei PB, Lin MT, et al. Molecular alterations in non-small cell lung carcinomas of the young. Hum Pathol 2014;45:2379-87. [PubMed]
- Luo W, Tian P, Wang Y, et al. Characteristics of genomic alterations of lung adenocarcinoma in young never-smokers. Int J Cancer 2018;143:1696-705. [PubMed]
- Hou H, Zhang C, Qi X, et al. Distinctive targetable genotypes of younger patients with lung adenocarcinoma: a cBioPortal for cancer genomics data base analysis. Cancer Biol Ther 2020;21:26-33. [PubMed]
- Scarpino S, Rampioni Vinciguerra GL, Di Napoli A, et al. High prevalence of ALK+/ROS1+ cases in pulmonary adenocarcinoma of adoloscents and young adults. Lung Cancer 2016;97:95-8. [Crossref] [PubMed]
- Jemal A, Miller KD, Ma J, et al. Higher Lung Cancer Incidence in Young Women Than Young Men in the United States. N Engl J Med 2018;378:1999-2009. [Crossref] [PubMed]
- Islami F, Goding Sauer A, Miller KD, et al. Proportion and number of cancer cases and deaths attributable to potentially modifiable risk factors in the United States. CA Cancer J Clin 2018;68:31-54. [Crossref] [PubMed]
- Mu L, Liu L, Niu R, et al. Indoor air pollution and risk of lung cancer among Chinese female non-smokers. Cancer Causes Control 2013;24:439-50. [Crossref] [PubMed]
- Arnold BN, Thomas DC, Rosen JE, et al. Lung Cancer in the Very Young: Treatment and Survival in the National Cancer Data Base. J Thorac Oncol 2016;11:1121-31. [Crossref] [PubMed]
- Qu R, Tu D, Cai Y, et al. Clinical features and surgical outcomes of young patients with lung adenocarcinoma manifesting as ground glass opacity. Front Oncol 2022;12:979522. [PubMed]
- Ma Y, Fei X, Jiang C, et al. Lung adenocarcinoma manifested as ground-glass nodules in teenagers: characteristics, surgical outcomes and management strategies. Eur J Cardiothorac Surg 2024;66:ezae291. [PubMed]
- Rich AL, Khakwani A, Free CM, et al. Non-small cell lung cancer in young adults: presentation and survival in the English National Lung Cancer Audit. QJM 2015;108:891-7. [PubMed]
- Skarin AT, Herbst RS, Leong TL, et al. Lung cancer in patients under age 40. Lung Cancer 2001;32:255-64. [PubMed]
- Mauri D, Pentheroudakis G, Bafaloukos D, et al. Non-small cell lung cancer in the young: a retrospective analysis of diagnosis, management and outcome data. Anticancer Res 2006;26:3175-81. [PubMed]
- Kohsaka S, Hayashi T, Nagano M, et al. Identification of Novel CD74-NRG2α Fusion From Comprehensive Profiling of Lung Adenocarcinoma in Japanese Never or Light Smokers. J Thorac Oncol 2020;15:948-61. [PubMed]
- Bratova M, Brat K, Hurdalkova K, et al. Lung Cancer Versus "Young Cancer": Is Non-Small Cell Lung Cancer in Young Patients a Different Entity? J Adolesc Young Adult Oncol 2022;11:451-8. [PubMed]
- Shi Y, Au JS, Thongprasert S, et al. A prospective, molecular epidemiology study of EGFR mutations in Asian patients with advanced non-small-cell lung cancer of adenocarcinoma histology (PIONEER). J Thorac Oncol 2014;9:154-62. [PubMed]
- de Alencar VTL, Formiga MN, de Lima VCC. Inherited lung cancer: a review. Ecancermedicalscience 2020;14:1008. [PubMed]
- Chen J, Xie L. Contribution of Childhood Indoor Radon Exposure to Lung Cancer Incidence among Young Adults: A Population-Based Ecological Study in Canada. Radiation Environment and Medicine 2021;10:31-6.
- Hill W, Lim EL, Weeden CE, et al. Lung adenocarcinoma promotion by air pollutants. Nature 2023;616:159-67. [PubMed]
- Liu X, Mubarik S, Wang F, et al. Lung Cancer Death Attributable to Long-Term Ambient Particulate Matter (PM(2.5)) Exposure in East Asian Countries During 1990-2019. Front Med (Lausanne) 2021;8:742076. [PubMed]
- Araldi RP, Sant'Ana TA, Módolo DG, et al. The human papillomavirus (HPV)-related cancer biology: An overview. Biomed Pharmacother 2018;106:1537-56. [Crossref] [PubMed]
- de Oliveira THA, do Amaral CM, de França São Marcos B, et al. Presence and activity of HPV in primary lung cancer. J Cancer Res Clin Oncol 2018;144:2367-76. [Crossref] [PubMed]
- Huber M, Pavlova B, Mühlberger H, et al. Detection of the Epstein-Barr virus in primary adenocarcinoma of the lung with Signet-ring cells. Virchows Arch 2002;441:25-30. [Crossref] [PubMed]
- Leiter A, Veluswamy RR, Wisnivesky JP. The global burden of lung cancer: current status and future trends. Nat Rev Clin Oncol 2023;20:624-39. [PubMed]
- Laguna JC, Tagliamento M, Lambertini M, et al. Tackling Non-Small Cell Lung Cancer in Young Adults: From Risk Factors and Genetic Susceptibility to Lung Cancer Profile and Outcomes. Am Soc Clin Oncol Educ Book 2024;44:e432488. [PubMed]
- Yin H, Qu J, Peng Q, et al. Molecular mechanisms of EBV-driven cell cycle progression and oncogenesis. Med Microbiol Immunol 2019;208:573-83. [PubMed]
- Adugna A. Histomolecular characterisation of hepatitis B virus induced liver cancer. Rev Med Virol 2023;33:e2485. [PubMed]
- Zakurdaeva K, Tsimafeyeu I. Immunotherapy in cancer patients with chronic hepatitis C viral infection. J Cancer Res Ther 2023;19:1093-7. [Crossref] [PubMed]
- Yasunaga J, Matsuoka M. Human T-cell leukemia virus type I induces adult T-cell leukemia: from clinical aspects to molecular mechanisms. Cancer Control 2007;14:133-40. [Crossref] [PubMed]
- Sirera G, Videla S, Saludes V, et al. Prevalence of HPV-DNA and E6 mRNA in lung cancer of HIV-infected patients. Sci Rep 2022;12:13196. [Crossref] [PubMed]
- Ouda AM, Elsabagh AA, Elmakaty IM, et al. HPV and Recurrent Respiratory Papillomatosis: A Brief Review. Life (Basel) 2021;11:1279. [Crossref] [PubMed]
- Lin FC, Huang JY, Tsai SC, et al. The association between human papillomavirus infection and female lung cancer: A population-based cohort study. Medicine (Baltimore) 2016;95:e3856. [Crossref] [PubMed]
- Cheng YW, Wu MF, Wang J, et al. Human papillomavirus 16/18 E6 oncoprotein is expressed in lung cancer and related with p53 inactivation. Cancer Res 2007;67:10686-93. [Crossref] [PubMed]
- Hoppe-Seyler K, Bossler F, Braun JA, et al. The HPV E6/E7 Oncogenes: Key Factors for Viral Carcinogenesis and Therapeutic Targets. Trends Microbiol 2018;26:158-68. [Crossref] [PubMed]
- Henle W, Henle G. The Epstein-Barr Virus (EBV) in Burkitt's lymphoma and nasopharyngeal carcinoma. Ann Clin Lab Sci 1974;4:109-14. [PubMed]
- Bégin LR, Eskandari J, Joncas J, et al. Epstein-Barr virus related lymphoepithelioma-like carcinoma of lung. J Surg Oncol 1987;36:280-3. [PubMed]
- Tang L, Chen N, He W, et al. The clinicopathological features and prognosis of primary pulmonary lymphoepithelioma-like carcinoma: A systematic review and meta-analysis. PLoS One 2020;15:e0240729. [PubMed]
- Chen SH, Miser JS, Liew PL, et al. Pulmonary lymphoepithelioma-like carcinoma: A complication of Epstein-Barr virus in a teenager. J Microbiol Immunol Infect 2023;56:886-8. [PubMed]


