
Efficacy and safety of different forms of ablation therapies and antiarrhythmic drugs in atrial fibrillation: a systematic review and network meta-analysis of randomized clinical trials
Highlight box
Key findings
• Catheter ablation (CA) and surgical ablation (SA) more effectively reduce atrial fibrillation (AF) recurrence than antiarrhythmic drugs. CA has a lower complication rate, while SA carries a higher risk of adverse events. Additionally, no evidence indicates that radiofrequency ablation and SA significantly reduce the risk of mortality.
What is known and what is new?
• CA has become the first-line treatment for paroxysmal AF and symptomatic persistent AF.
• Among various CAs, cryoablation has the shortest procedure time, radiofrequency ablation may lower all-cause mortality rate, while SA is associated with more complications.
What is the implication, and what should change now?
• Clinicians should prioritize CA for the treatment of AF, especially for patients at higher risk of recurrence. Future research is needed to refine the evidence and guidelines for personalized treatment.
Introduction
Atrial fibrillation (AF), the most prevalent sustained cardiac arrhythmia, afflicts approximately 1–2% of the global population (1). It is estimated that by 2050, around 6–12 million individuals in the US (2) and by 2060, 17.9 million individuals in Europe (3) will suffer from this condition. AF is a progressive disease (4), and its progression is associated with an increased risk of myocardial infarction, thromboembolism, acute heart failure, and stroke, leading to considerable morbidity and mortality if these complications are left inadequately managed (5-7).
At present, three primary modalities are available for managing AF: catheter ablation (CA), surgical ablation (SA), and antiarrhythmic drugs (AADs). Although drug therapy remains the mainstay treatment for AF, its limited efficacy and potential side effects present significant challenges. The latest European guidelines (8) have endorsed CA, which includes radiofrequency ablation (RFA), cryoablation (CBA), laser balloon ablation (LBA), as the first-line therapy for paroxysmal AF (PAF), drug-refractory AF, and symptomatic AF. Furthermore, accumulating evidence has demonstrated the superiority of CA over AADs in preventing AF recurrence (9-11). Interestingly, Pokushalov et al. (12) found that SA outperformed CA in maintaining sinus rhythm in patients with PAF and persistent AF (PersAF) who had experienced failure with initial CA treatment.
As a result, the investigation of CA for individuals with AF remains a hotly debated topic for many scientists. Although several randomized controlled trials (RCTs) have been carried out on ClinicalTrials.gov (such as NCT04612244, NCT05534581, NCT03920917, and NCT04877327), there is still no consensus on the optimal ablation technique for patients with AF. Therefore, we conducted this comprehensive analysis and Bayesian network meta-analysis (NMA) to evaluate the effectiveness and safety of different ablation methods and AADs. We present this article in accordance with the PRISMA-NMA reporting checklist (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1545/rc) (13).
Methods
Study design and registration
As the NMA relies on previously published data, ethical clearance or consent to participate is not deemed necessary. The study’s methodology was duly registered on PROSPERO, with the registration number CRD42022371798.
Eligibility criteria and type of interventions
RCTs were included for systematic review if they compared CA and AADs or evaluated the efficacy of two or more ablation techniques for treating adult patients with AF (PAF and PersAF). Additionally, these trials were followed up for at least 3 months. RCTs investigating patients who underwent previous ablation procedures such as catheter, surgical, or atrioventricular node ablation were deemed not eligible for review.
Search strategy
We systematically searched PubMed, Embase, Web of Science, and Cochrane Library databases for RCTs published from the inception of the abovementioned databases to October 19, 2022. To avoid the risk of omission of newly published literature, we conducted additional searches of the databases on December 15, 2024. The search was conducted utilizing subject terms together with free words, without any restrictions on geographical area or language. A listing of the search terms and strategies employed can be found in Appendix 1. Additionally, our analysis exclusively considered publicly accessible RCTs or parallel controlled trials.
Literature screening
Retrieved studies were imported into Endnote to remove duplicates. The remaining studies were screened by reading the titles and abstracts. Then, the potentially eligible studies were reviewed by reading the full texts. Two investigators (Q.C. and X.X.) independently screened the studies obtained from databases. Any disagreement between them was resolved by discussion with a third member of the investigator team (L.L.).
Data extraction
A standard data extraction table was drawn before we started to extract data, including the characteristics of included RCTs [study title, first author, study region, National Clinical Trial (NCT) number, year of publication, sample size, intervention strategy, follow-up duration] and the characteristics of participants [mean age, gender, type of AF, duration of AF, body mass index (BMI), left atrium diameter, left ventricular ejection fraction, blanking period, outcome indicators]. Two investigators (Q.C. and X.X.) independently extracted the data and the results were then cross-checked. Any disagreements were resolved by discussion with a third investigator (L.L.) to reach a consensus.
Outcomes
Primary outcomes
Efficacy
The primary endpoint was the duration from the completion of treatment to the first recurrence of symptomatic or asymptomatic atrial tachyarrhythmia [AF, atrial flutter (AFL), or atrial tachycardia (AT)] as determined by any form of monitoring methods including implantable loop recorder, pacemaker, defibrillator, electrocardiogram (ECG), or ambulatory-ECG, during a minimum follow-up period of 6 months after CA. An atrial tachyarrhythmia lasting no less than 30 seconds was considered an arrhythmia. To account for early AF recurrences, a standard 90-day blanking period was applied.
Safety
The primary safety endpoint was the total number of all-cause deaths, cerebrovascular events, or serious treatment-related adverse events during the periprocedural period or throughout the follow-up.
Secondary outcomes
The prespecified secondary endpoints reported in this review included procedure time and fluoroscopy time.
Risk of bias assessment
Two researchers (Q.C. and X.X.) used the RCT risk of bias assessment tool from the Cochrane Collaboration Network to assess the risk of bias across included studies. The following parameters were considered for study quality assessment: generation of random sequences, allocation concealment, blinding of subjects and intervention providers, blinding of outcome evaluators, incomplete outcome data, selective outcome reporting, and other sources of bias. A study was rated as having low, high, or unclear risk of bias regarding each of the abovementioned parameters.
Statistical analysis
A Bayesian random-effects model was employed to compare the effectiveness of various therapies for AF. After 50,000 simulation cycles, the modeling was finished using a Markov chain Monte Carlo (MCMC) method with four chains run in parallel and an annealing number of 20,000. In the event of a closed-loop mesh, the node splitting method was employed to evaluate local consistency, and the model fitting and global consistency were assessed using the deviation information criteria (DIC). Additionally, league tables were constructed to compare the effects of different intervention modalities, and a surface under the cumulative ranking curve (SUCRA) was used to rate the interventions. Data analyses were performed using the software Stata 16.0 (Stata Corporation, College Station, TX, USA) and R 4.2.3 (R Development Core Team, Vienna, Austria; http://www.rproject.org). Statistical significance was considered to be P<0.05.
Results
Literature search
Figure 1 illustrates the selection process for RCTs included in this review. The initial literature search yielded 4,812 articles that were potentially relevant to our research. After a thorough review of their titles and abstracts, 1,948 articles were excluded, leaving 2,864 trials for further evaluation. Upon further examination, 2,744 articles were excluded after reviewing their full text, resulting in 63 RCTs that met the predefined inclusion criteria involving 17,522 patients for further analysis.
Characteristics of included studies
Appendices 2,3 show the baseline characteristics and references for each study.
Quality assessment
Of the 63 RCTs, 27 did not provide a specific description of the random sequence generation, while four clearly indicated that treatment allocation was concealed. Given that CA requires informed consent before the procedures, it is not feasible to blind participants and personnel in practice. Regarding the blinding aspect, the comparator was medical therapy, so it was not possible to achieve blinding. Blinding would be feasible when two different energy sources are used in CA. Regarding detection bias, nine studies explicitly outlined protocols for blinding outcome assessment. All RCTs were judged as not having a risk of attrition bias or reporting bias. The quality assessment results are presented in Figure 2 and Appendix 4.
Primary outcomes
AF recurrence
The clinical outcome of AF recurrence was reported in 54 RCTs, which involved RFA, CBA, LBA, SA and AADs. Among them, RFA, CBA and AADs were the main measures used in most studies (Figure 3A). A random-effect model was employed. Analysis results showed that RFA [risk ratio (RR) =0.503, 95% confidence interval (CI) 0.423–0.594], CBA (RR =0.465, 95% CI: 0.370–0.579), LBA (RR =0.522, 95% CI: 0.306–0.891), and SA (RR =0.664, 95% CI: 0.392–1.12) all significantly reduced the risk of recurrent AF compared with AADs (Figure 3B), with no significant differences between ablation modalities regarding the risk of AF recurrence (Table 1). The ranking results showed CBA > LBA > RFA > SA > AADs (Figure 3C). Although the duration of follow-up for AF recurrence varied amongst trials, the meta-regression based on NMA revealed no correlation between follow-up duration and AF recurrence (Table 2). Moreover, we observed notable discrepancies in the risk of relapse between the PAF and PersAF cohorts, with the latter exhibiting a marginally increased risk. Thus, we conducted a subgroup analysis based on the study follow-up duration (Appendix 5) and the type of AF (Appendix 6).
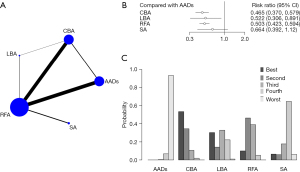
Table 1
| Treatment for AF | AADs | CBA | LBA | RFA | SA |
|---|---|---|---|---|---|
| AADs | 0.465 (0.370, 0.579) | 0.522 (0.306, 0.891) | 0.503 (0.423, 0.594) | 0.664 (0.392, 1.12) | |
| CBA | 2.152 (1.726, 2.705) | 1.123 (0.667, 1.9) | 1.083 (0.907, 1.293) | 1.429 (0.847, 2.429) | |
| LBA | 1.915 (1.123, 3.273) | 0.89 (0.526, 1.499) | 0.963 (0.577, 1.601) | 1.27 (0.626, 2.592) | |
| RFA | 1.989 (1.683, 2.364) | 0.924 (0.774, 1.103) | 1.039 (0.624, 1.733) | 1.32 (0.807, 2.175) | |
| SA | 1.507 (0.893, 2.548) | 0.7 (0.412, 1.18) | 0.787 (0.386, 1.598) | 0.758 (0.46, 1.239) |
Table 1 shows a league table of AF recurrence after different treatments. The row and column titles list AF treatments, including AADs, CBA, LBA, RFA, and SA. The diagonal divides the table into an upper triangle and a lower triangle. In the lower triangle, the reference treatment is on the right side of each comparison, while in the upper triangle, the reference treatment is on the left side. The presented RRs and their 95% CIs are crucial for interpreting the relative effectiveness of the treatments. When the 95% CI of RR does not include 1, the difference is statistically significant, with a P value less than 0.05. AAD, antiarrhythmic drug; AF, atrial fibrillation; CBA, cryoablation; CI, confidence interval; LBA, laser balloon ablation; RFA, radiofrequency ablation; RR, risk ratio; SA, surgical ablation.
Table 2
| AF recurrence | Beta (regression index) | 95% CI |
|---|---|---|
| RFA vs. AADs | 1.544 | 1.186 to 1.911 |
| RFA vs. CBA | −0.098 | −0.458 to 0.261 |
| RFA vs. LBA | 0.385 | −10.085 to 11.948 |
| RFA vs. SA | 0.598 | −0.564 to 1.766 |
Table 2 shows the meta-regression indices for AF recurrence. Each row represents a comparison between different treatments, namely AADs, CBA, LBA, RFA, and SA. The “beta (regression index)” column gives the regression coefficient values, showing how treatment differences affect AF recurrence in terms of magnitude and direction. The “95% CI” column offers the 95% CI of the regression coefficient, which helps evaluate the precision and statistical significance of the estimate. AAD, antiarrhythmic drug; AF, atrial fibrillation; CBA, cryoablation; CI, confidence interval; LBA, laser balloon ablation; RFA, radiofrequency ablation; SA, surgical ablation.
Adverse events
Forty-seven studies reported on the outcome of adverse events, involving RFA, CBA, LBA, SA, and AADs. Among them, RFA, CBA, and AADs were the main measures used in most studies (Figure 4A). A random-effect model was employed. The results of our NMA indicated that SA (RR =2.054, 95% CI: 0.999–4.369) may increase the risk of adverse events compared with AADs, but RFA (RR =0.917, 95% CI: 0.674–1.261) and LBA (RR =0.93, 95% CI: 0.358–2.46) were not associated increased risk of adverse events compared with AADs, with RR=1.462 (95% CI: 0.956–2.283) for CBA (Figure 4B), and there was no significant difference between ablation groups regarding the risk of adverse events (Table 3). The performance ranking results showed LBA > RFA > AADs > CBA > SA (Figure 4C).

Table 3
| Treatment for AF | AADs | CBA | LBA | RFA | SA |
|---|---|---|---|---|---|
| AADs | 1.462 (0.956, 2.283) | 0.93 (0.358, 2.46) | 0.917 (0.674, 1.261) | 2.054 (0.999, 4.369) | |
| CBA | 0.684 (0.438, 1.046) | 0.636 (0.243, 1.647) | 0.628 (0.428, 0.91) | 1.407 (0.657, 3.047) | |
| LBA | 1.075 (0.407, 2.794) | 1.572 (0.607, 4.108) | 0.985 (0.392, 2.471) | 2.214 (0.71, 6.943) | |
| RFA | 1.091 (0.793, 1.483) | 1.594 (1.099, 2.337) | 1.015 (0.405, 2.553) | 2.244 (1.165, 4.407) | |
| SA | 0.487 (0.229, 1.001) | 0.711 (0.328, 1.521) | 0.452 (0.144, 1.409) | 0.446 (0.227, 0.859) |
Table 3 shows a league table about the adverse events of AF after different treatments. The row and column titles list AF treatments like AADs, CBA, LBA, RFA, and SA. The diagonal divides the table into an upper triangle and a lower triangle. In the lower triangle, the reference treatment is on the right side of each comparison, while in the upper triangle, the reference treatment is on the left side. The presented RRs and their 95% CIs are crucial for interpreting the relative effectiveness of the treatments. When the 95% CI of RR does not include 1, the difference is statistically significant, with a P value less than 0.05. AAD, antiarrhythmic drug; AF, atrial fibrillation; CBA, cryoablation; CI, confidence interval; LBA, laser balloon ablation; RFA, radiofrequency ablation; RR, risk ratio; SA, surgical ablation.
All-cause mortality
Eleven studies reported on the outcome of all-cause mortality, these studies only covered RFA, SA, and AADs. In most studies, RFA and AADs were predominantly used (Figure 5A). Among the 108 patients who underwent SA, there were three RCTs. Random-effect models were employed. RFA (RR =0.745, 95% CI: 0.552–1.014) and SA (RR =0.777, 95% CI: 0.198–3.18) were not associated with reduced risk of adverse events compared with AADs (Figure 5B), and no significant difference emerged between RFA and SA groups in all-cause mortality (Table 4). The performance ranking showed RFA > SA > AADs (Figure 5C). The results of secondary outcomes (procedure duration and fluoroscopy time) were shown in Appendices 7,8.

Table 4
| Treatment for AF | AADs | RFA | SA |
|---|---|---|---|
| AADs | 0.745 (0.552, 1.014) | 0.777 (0.198, 3.18) | |
| RFA | 1.342 (0.986, 1.813) | 1.043 (0.276, 4.104) | |
| SA | 1.287 (0.314, 5.04) | 0.959 (0.244, 3.62) |
Table 4 shows a league table about the all-cause modality of AF after different treatments. The row and column titles list AF treatments like AADs, RFA, and SA. The diagonal divides the table into an upper triangle and a lower triangle. In the lower triangle, the reference treatment is on the right side of each comparison, while in the upper triangle, the reference treatment is on the left side. The presented RRs and their 95% CIs are crucial for interpreting the relative effectiveness of the treatments. When the 95% CI of RR does not include 1, the difference is statistically significant, with a P value less than 0.05. AAD, antiarrhythmic drug; AF, atrial fibrillation; CI, confidence interval; RFA, radiofrequency ablation; RR, risk ratio; SA, surgical ablation.
Discussion
Principal findings
To our knowledge, this is the first NMA to evaluate the efficacy and safety of different ablation techniques and AADs for the initial treatment of AF patients. The following are the key findings: (I) both CA and SA were found to reduce post-operative AF recurrence compared with AADs, irrespective of the duration of follow-up; (II) SA may be associated with a higher risk of adverse events; (III) compared to AADs, no evidence showed that RFA and SA notably decreased the mortality risk; and (IV) CBA took the shortest time to perform.
RFA vs. CBA vs. LBA
The present meta-analysis compared the efficacy of RFA, CBA, and LBA as initial treatments for AF. The analysis was based on 21 RFA vs. CBA, three RFA vs. LBA, and two CBA vs. LBA RCTs. The results revealed that three ablation techniques showed comparable efficacy, irrespective of AF type and follow-up time. However, CBA took the shortest time to complete, although no significant difference was observed in terms of fluoroscopy time.
Several meta-analyses have investigated the efficacy and safety of CBA and RFA in different AF populations. In the PAF cohort (14), PersAF cohort (15), and both PAF and PersAF cohort (16), these three meta-analyses investigating diverse populations of AF types have demonstrated that both CBA and RFA exhibit equivalent efficacy and safety, with CBA taking a shorter time to perform. Another meta-analysis by Wei et al. (17) showed that there was no statistically significant difference in atrial arrhythmia recurrence between LBA vs. CBA at 12 months [RR (95% CI): 0.91 (0.64–1.28), P=0.59], but LBA tended to be more effective than CBA in PAF patients [RR (95% CI): 0.70 (0.47–1.03), P=0.07]. The latest NMA by Zhao et al. (18) reported no significant difference in the incidence of major adverse events among different ablation modalities. These findings are consistent with the results of our NMA. Noteworthy, Ravi, Wei, and Kim’s studies (15-17) primarily included observational studies, while Murray’s study (14) was limited to only four RCTs, and Zhao’s study (18) only focused on the safety aspect and did not compare the effectiveness of various ablation methods and drug treatments for AF. Therefore, the latter study may have been affected by selection bias and other study design confounders.
RFA has advantages over CBA and LBA due to its flexibility and accessibility to non-pulmonary vein targets such as the posterior wall and roof ablation lines. CBA is a standardized and single-shot procedure that achieves efficient pulmonary vein isolation (PVI) using hypothermia for which CBA can only be performed once per vein, and its success rate is generally high [approximately 93% in the STOP-AF trial (19)]. However, when CBA fails to isolate the pulmonary veins, additional RFA may be necessary, particularly in the inferior pulmonary veins. This presents a challenge for CBA (20). Compared to CBA and RFA, LBA utilizes a miniature endoscope allowing for segment-by-segment ablation, enabling a more precise ablation of the targets (21).
The above-mentioned three ablation methods present varying degrees of risk for complications. RFA carries the risk of cardiac perforation and tamponade due to tissue overheating, whereas LBA and CBA can cause phrenic nerve palsy due to the expansion of the balloon and consequent malformation of the right upper lobe venous system, thereby reducing the distance from the phrenic nerve (22). Paralysis is rare and most individuals recover before discharge. Our study confirmed previous findings by revealing no significant difference in adverse events associated with RFA, LBA, and CBA (14,16). Over the last decade, CA techniques have reduced complications due to a shorter learning curve, the use of contact pressure catheters, more precise septal penetration with intracardiac echocardiography, and safer procedures with real-time monitoring of intraoperative pericardial effusion.
CA vs. SA
The results of our study suggest that although SA and CA are similarly effective in treating AF, SA is associated with more complications than CA. Phan et al. (23) conducted a meta-analysis that revealed SA was more effective than CA in reducing arrhythmias in patients on and off AAD at 6- and 12-month follow-up. Similarly, the meta-analysis by Rattanawong et al. (24) demonstrated that SA resulted in higher freedom from AF, higher pacemaker placement rates, and equivalent rates of neurological sequelae and cardiac tamponade compared to CA. However, it is important to note that these studies included case-control and cohort studies, which can lead to highly heterogeneous results and therefore undermine the credibility of the results. Two additional meta-analyses (25,26) that exclusively included RCTs revealed that SA led to a higher success rate compared to CA. However, these studies focused on recurring patients who had previously undergone unsuccessful CA, rather than using it as an initial treatment, which conflicts with our study’s inclusion criteria and hence yielded different results. Interestingly, Yi et al.’s report (27) showed that in the sub-analysis of patients with PAF or early PersAF, SA did not provide higher efficacy results than the initial invasive procedure, which is in line with our findings.
The aforementioned discrepancies in the treatment of AF can be attributed to two factors. On the one hand, traditional CA is often effective for PAF and early PersAF (defined as PersAF less than 3 months), which may obviate the need for a more invasive SA. However, it should be noted that if CA fails, SA can serve as a valuable alternative with demonstrated efficacy. On the other hand, for patients with long-standing PersAF or refractory AF with valvular problems, initial treatment with CA is likely to be ineffective. In such cases, SA may be a better treatment when. This may be explained by inherent differences in SA and CA techniques. Unlike CA, SA technique includes a greater variety of ablative lesions. Thoracoscopic SA includes PVI identical to CA, as well as epicardial ganglion plexus, left auricular resection, and additional left auricular line, Marshall’s ligament resection (12,28-30). Several studies (31,32) suggest that the ganglion plexus may be one of the substrates that perpetuate the AF refractory loop. Hence, removing these autonomic stimuli may reduce the incidence of arrhythmias. Similarly, left auricular has been identified as the trigger site in 27% of AF patients, and its removal may also contribute to AF freedom (33). According to Rodríguez-Mañero et al. (34), the vein of Marshall is a promising site for CA of AF since it has innervation, myocardial connections, and arrhythmogenic foci. In contrast, the point-by-point ablation performed by CA at the endocardium leaves patients susceptible to potential PV reconnection and AF recurrence.
SA is seldom utilized due to the significant risk of surgical adverse effects associated with it (28). The latest guidelines recommend SA in conjunction with surgical repair of other structural cardiac diseases (particularly mitral valve disease), whereas SA alone is usually not accepted as a primary treatment until CA has failed (8). A meta-analysis conducted by Phan et al. (23) revealed that rates of major complications were 3.5 times greater in the SA group than in the CA cohort, with higher rates of intraoperative pleural effusion and pneumothorax. These complications may be attributed to mechanical injury caused by pulmonary deflation during the procedure and damage to nearby pleural structures during epicardial ablation of the beating heart. With the improvement in SA techniques, thoracoscopic SA is now frequently performed without the need for open-heart exposure (29,30). However, small incisions to create ports for the endoscope and bipolar ablation forceps, as well as lung deflation are required to allow surgical access to epicardial pulsatile heart lesions. Furthermore, the shorter learning curve of CA compared to SA is one reason why there are higher rates of complications with SA. The application of ablation index, three-dimensional (3D) scaler systems, intra-cardiac ultrasonography, and visualization sheaths have also contributed to reducing the occurrence of adverse occurrences associated with CA.
Limitation
In the present study, a Bayesian NMA was conducted to examine 65 RCTs. Several limitations in this study must be considered. First, certain single-center studies with relatively low quality and small sample sizes were not excluded to ensure the inclusion of all relevant research. Although our NMA aimed to integrate the additional uncertainty associated with bias generated by multiple study designs, the capacity to estimate genuine effects may be constrained by the limited number of high-quality studies in the network. Second, this study compared four ablation techniques and pharmacological therapy, but some of the catheters initially used, such as non-contact pressure or non-saline irrigated RFA, LBA, and first-generation CBA, are not commonly used and may be susceptible to publication bias, as newer techniques are typically superior to established ones. Third, although the quality of the included studies is adequate, certain factors may have led to bias. For example, it was not possible to blind patients and operators, there were differences in operator experience and habits across cardiac centers, and the use of anti-arrhythmias after the blanking period varied. Fourth, although 63 studies were included, only 11 involved long-term follow-up and examined the effect of RFA and SA on all-cause mortality compared with AADs. The limited number of such studies might constrain the results. Hence, future research should perform further long-term follow-up on patients undergoing different ablation methods to determine whether these methods can reduce the mortality risk. Furthermore, the data are inconsistent in terms of endpoint definitions, follow-up durations, and arrhythmia recurrence monitoring protocols. Last, it should be noted that NMAs have an inherent limitation. The network graph reveals indirect measurements, which entail comparisons between groups that are not directly reported in published studies. However, neither the extent nor the number of these indirect measurements was specified in the original manuscript. The ambiguity of indirect comparisons may affect the interpretation and generalizability of our results, given the potentially variable reliability of such indirect evidence.
Conclusions
The efficacy and safety outcomes of several CA and SA approaches were comparable, and there is inadequate evidence to conclude that one strategy is preferable to another. Among the evaluated methods, CBA took the shortest time to perform, but it was associated with a somewhat high incidence of complications. Moreover, compared to AADs, no evidence shows that RFA and SA notably decrease the mortality risk. To make an informed decision about selecting a suitable ablation modality, some factors need to be considered, such as the expertise, skill, and habits of electrophysiologists, as well as the patient’s clinical condition and financial circumstances.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the PRISMA-NMA reporting checklist. Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1545/rc
Peer Review File: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1545/prf
Funding: This work was supported by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1545/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Lippi G, Sanchis-Gomar F, Cervellin G. Global epidemiology of atrial fibrillation: An increasing epidemic and public health challenge. Int J Stroke 2021;16:217-21. [Crossref] [PubMed]
- Patel NJ, Deshmukh A, Pant S, et al. Contemporary trends of hospitalization for atrial fibrillation in the United States, 2000 through 2010: implications for healthcare planning. Circulation 2014;129:2371-9. [Crossref] [PubMed]
- Krijthe BP, Kunst A, Benjamin EJ, et al. Projections on the number of individuals with atrial fibrillation in the European Union, from 2000 to 2060. Eur Heart J 2013;34:2746-51. [Crossref] [PubMed]
- Padfield GJ, Steinberg C, Swampillai J, et al. Progression of paroxysmal to persistent atrial fibrillation: 10-year follow-up in the Canadian Registry of Atrial Fibrillation. Heart Rhythm 2017;14:801-7. [Crossref] [PubMed]
- Mukherjee K, Kamal KM. Impact of atrial fibrillation on inpatient cost for ischemic stroke in the USA. Int J Stroke 2019;14:159-66. [Crossref] [PubMed]
- Ganesan AN, Chew DP, Hartshorne T, et al. The impact of atrial fibrillation type on the risk of thromboembolism, mortality, and bleeding: a systematic review and meta-analysis. Eur Heart J 2016;37:1591-602. [Crossref] [PubMed]
- Chiang CE, Naditch-Brûlé L, Murin J, et al. Distribution and risk profile of paroxysmal, persistent, and permanent atrial fibrillation in routine clinical practice: insight from the real-life global survey evaluating patients with atrial fibrillation international registry. Circ Arrhythm Electrophysiol 2012;5:632-9. [Crossref] [PubMed]
- Van Gelder IC, Rienstra M, Bunting KV, et al. 2024 ESC Guidelines for the management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J 2024;45:3314-414. [Crossref] [PubMed]
- Razzack AA, Lak HM, Pothuru S, et al. Efficacy and Safety of Catheter Ablation vs Antiarrhythmic Drugs as Initial Therapy for Management of Symptomatic Paroxysmal Atrial Fibrillation: A Meta-Analysis. Rev Cardiovasc Med 2022;23:112. [Crossref] [PubMed]
- Patel M, Changal K, Patel N, et al. Cryoablation versus antiarrhythmic therapy for initial treatment of atrial fibrillation: a systematic review and meta-analysis. Expert Rev Cardiovasc Ther 2021;19:667-71. [Crossref] [PubMed]
- Hakalahti A, Biancari F, Nielsen JC, et al. Radiofrequency ablation vs. antiarrhythmic drug therapy as first line treatment of symptomatic atrial fibrillation: systematic review and meta-analysis. Europace 2015;17:370-8. [Crossref] [PubMed]
- Pokushalov E, Romanov A, Elesin D, et al. Catheter versus surgical ablation of atrial fibrillation after a failed initial pulmonary vein isolation procedure: a randomized controlled trial. J Cardiovasc Electrophysiol 2013;24:1338-43. [Crossref] [PubMed]
- Hutton B, Salanti G, Caldwell DM, et al. The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: checklist and explanations. Ann Intern Med 2015;162:777-84. [Crossref] [PubMed]
- Murray MI, Arnold A, Younis M, et al. Cryoballoon versus radiofrequency ablation for paroxysmal atrial fibrillation: a meta-analysis of randomized controlled trials. Clin Res Cardiol 2018;107:658-69. [Crossref] [PubMed]
- Kim JA, Chelu MG. Comparison of cryoballoon and radiofrequency ablation for persistent atrial fibrillation: a systematic review and meta-analysis. J Interv Card Electrophysiol 2023;66:585-95. [Crossref] [PubMed]
- Ravi V, Poudyal A, Pulipati P, et al. A systematic review and meta-analysis comparing second-generation cryoballoon and contact force radiofrequency ablation for initial ablation of paroxysmal and persistent atrial fibrillation. J Cardiovasc Electrophysiol 2020;31:2559-71. [Crossref] [PubMed]
- Wei Y, Zhang N, Jin Q, et al. Comparison of efficacy and safety of laser balloon and cryoballoon ablation for atrial fibrillation-a meta-analysis. J Interv Card Electrophysiol 2019;54:237-45. [Crossref] [PubMed]
- Zhao Y, Yi X. Safety of different therapies for atrial fibrillation: a network meta-analysis. Curr Probl Cardiol 2024;49:102795. [Crossref] [PubMed]
- Wazni OM, Dandamudi G, Sood N, et al. Cryoballoon Ablation as Initial Therapy for Atrial Fibrillation. N Engl J Med 2021;384:316-24. [Crossref] [PubMed]
- Pak HN, Park JW, Yang SY, et al. Cryoballoon Versus High-Power, Short-Duration Radiofrequency Ablation for Pulmonary Vein Isolation in Patients With Paroxysmal Atrial Fibrillation: A Single-Center, Prospective, Randomized Study. Circ Arrhythm Electrophysiol 2021;14:e010040. [Crossref] [PubMed]
- Dukkipati SR, Cuoco F, Kutinsky I, et al. Pulmonary Vein Isolation Using the Visually Guided Laser Balloon: A Prospective, Multicenter, and Randomized Comparison to Standard Radiofrequency Ablation. J Am Coll Cardiol 2015;66:1350-60. [Crossref] [PubMed]
- Chun JKR, Bordignon S, Last J, et al. Cryoballoon Versus Laserballoon: Insights From the First Prospective Randomized Balloon Trial in Catheter Ablation of Atrial Fibrillation. Circ Arrhythm Electrophysiol 2021;14:e009294. [Crossref] [PubMed]
- Phan K, Phan S, Thiagalingam A, et al. Thoracoscopic surgical ablation versus catheter ablation for atrial fibrillation. Eur J Cardiothorac Surg 2016;49:1044-51. [Crossref] [PubMed]
- Rattanawong P, Kewcharoen J, Kanitsoraphan C, et al. Surgical versus catheter ablation in atrial fibrillation: a systematic review and meta-analysis of randomized controlled trials. J Am Coll Cardiol 2020;75:460. [Crossref] [PubMed]
- Huang H, Wang Q, Xu J, et al. Comparison of catheter and surgical ablation of atrial fibrillation: A systemic review and meta-analysis of randomized trials. J Thorac Cardiovasc Surg 2022;163:980-93. [Crossref] [PubMed]
- Yonas E, Pranata R, Siswanto BB, et al. Comparison between surgical and catheter based ablation in atrial fibrillation, should surgical based ablation be implemented as first line? - A meta-analysis of studies. Indian Pacing Electrophysiol J 2020;20:14-20. [Crossref] [PubMed]
- Yi S, Liu X, Wang W, et al. Thoracoscopic surgical ablation or catheter ablation for patients with atrial fibrillation? A systematic review and meta-analysis of randomized controlled trials. Interact Cardiovasc Thorac Surg 2020;31:763-73. [Crossref] [PubMed]
- Adiyaman A, Buist TJ, Beukema RJ, et al. Randomized Controlled Trial of Surgical Versus Catheter Ablation for Paroxysmal and Early Persistent Atrial Fibrillation. Circ Arrhythm Electrophysiol 2018;11:e006182. [Crossref] [PubMed]
- Castellá M, Kotecha D, van Laar C, et al. Thoracoscopic vs. catheter ablation for atrial fibrillation: long-term follow-up of the FAST randomized trial. Europace 2019;21:746-53. [Crossref] [PubMed]
- Haldar S, Khan HR, Boyalla V, et al. Catheter ablation vs. thoracoscopic surgical ablation in long-standing persistent atrial fibrillation: CASA-AF randomized controlled trial. Eur Heart J 2020;41:4471-80. [Crossref] [PubMed]
- Scanavacca M, Pisani CF, Hachul D, et al. Selective atrial vagal denervation guided by evoked vagal reflex to treat patients with paroxysmal atrial fibrillation. Circulation 2006;114:876-85. [Crossref] [PubMed]
- Lemola K, Chartier D, Yeh YH, et al. Pulmonary vein region ablation in experimental vagal atrial fibrillation: role of pulmonary veins versus autonomic ganglia. Circulation 2008;117:470-7. [Crossref] [PubMed]
- Di Biase L, Burkhardt JD, Mohanty P, et al. Left atrial appendage: an underrecognized trigger site of atrial fibrillation. Circulation 2010;122:109-18. [Crossref] [PubMed]
- Rodríguez-Mañero M, Schurmann P, Valderrábano M. Ligament and vein of Marshall: A therapeutic opportunity in atrial fibrillation. Heart Rhythm 2016;13:593-601. [Crossref] [PubMed]




