
Early and midterm outcomes of concomitant coronary artery bypass grafting for coronary artery disease in surgical repair of type A aortic dissection
Highlight box
Key findings
• Coronary artery bypass grafting (CABG) for concurrent coronary artery disease (CAD) in type A aortic dissection (TAAD) patients shows better surgical outcomes but is associated with increased midterm mortality.
What is known and what is new?
• Concurrent CAD is frequently observed in patients with TAAD, but the impact of CABG for concurrent CAD in TAAD patients remains uncertain.
• This study demonstrates that CABG for concurrent CAD in TAAD patients reduces operative mortality but increases midterm mortality. A high incidence of graft occlusion is observed, particularly when the proximal end of the graft is anastomosed to the prosthesis.
What is the implication, and what should change now?
• The proximal end of the saphenous vein graft should be anastomosed to the native vessel tissue rather than to the prosthesis, as the latter negatively impacts long-term graft patency and patient prognosis.
Introduction
Concurrent coronary artery disease (CAD) is frequently observed in patients with type A aortic dissection (TAAD), with the reported incidence rates ranging from 6.0% to 46.4% (1,2). Once TAAD is diagnosed, urgent surgical intervention becomes imperative. However, the necessity of performing concomitant coronary artery bypass grafting (CABG) for CAD in TAAD patients remains uncertain. Two significant challenges impede the resolution of this issue: (I) the assessment of CAD in TAAD patients; and (II) the scarcity of high-quality studies.
Firstly, assessing CAD in TAAD patients is problematic, as coronary angiography is controversial due to the heightened risk of aortic rupture (3,4). Although preoperative coronary computed tomography angiography (CTA) is a safer alternative, it does not match the accuracy of coronary angiography in evaluating CAD. Secondly, few studies have specifically focused on the presence of CAD in TAAD patients, and those that do exist are often constrained by small sample sizes (4). More nuanced research questions, such as whether CABG should be performed for moderate coronary stenosis or whether complete revascularization is beneficial, have been extensively studied in isolated CABG procedures (5-7), but not in the context of TAAD.
Untreated coronary stenosis has been thoroughly investigated in isolated CABG, where it has been shown to increase the risk of acute peri-procedural myocardial infarction (8,9). However, its impact on the prognosis of TAAD patients remains unclear. Conversely, concomitant CABG undoubtedly prolongs the duration of surgery and may introduce the risk of competitive coronary flow, particularly when grafting non-severely stenotic coronary arteries (10,11). Additionally, it may increase the incidence of graft occlusion and bleeding events associated with antiplatelet therapy (12,13). Despite these considerations, the effect of concomitant CABG on patient outcomes during TAAD repair has not been thoroughly investigated.
At our institution, coronary CTA is routinely performed in the emergency department, enabling us to conduct a retrospective analysis. This study aims to evaluate the impact of concomitant CABG for co-existed CAD on both short-term and long-term outcomes following TAAD repair. We present this article in accordance with the STROBE reporting checklist (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1587/rc).
Methods
Participants
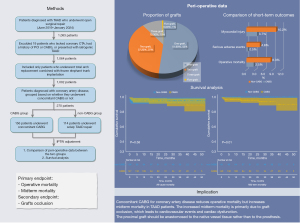
This project is a post-hoc substudy of a larger ambidirectional cohort study conducted at Fuwai Hospital. A total of 1,083 consecutive patients who underwent surgical repair for TAAD from June 2019 to January 2024 were included. Clinical data were collected from the hospital database. The inclusion criteria were: (I) patients who underwent TAAD surgical repair with total arch replacement combined with frozen elephant trunk implantation, and (II) patients diagnosed with CAD. The exclusion criteria included: (I) patients with a history of CABG or percutaneous coronary intervention, (II) patients lacking preoperative coronary CTA, and (III) patients with iatrogenic TAAD occurring during coronary angiography. The patient selection process and study flow are illustrated in Figure 1.

This study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This retrospective study was approved by the institutional review board of Fuwai Hospital on December 6th, 2021 (No. 2021-1490). The need for informed consent was waived for the retrospective nature of this study.
Data collection
An accurate assessment of CAD was essential in this study, primarily relying on coronary CTA. At our institution, only patients with documented computed tomography (CT) images were eligible for surgery. Coronary CTA was performed on all TAAD patients in the emergency room, except for those with renal failure or those who refused surgery. CT images were retrieved from the picture archiving and communication system. CAD was defined as severe stenosis (>50%) in the major branches of the coronary arteries and diagnosed by experienced radiologists, with oversight from a senior radiologist. Compromised coronary ostia were identified through coronary CT images and surgical notes and categorized into three subtypes according to the Neri classification (Neri A, Neri B, and Neri C) (14).
Clinical data were collected from electronic medical records, and surgical notes were retrospectively reviewed for details of CABG, including the number and proportion of grafts, target vessels (e.g., right coronary artery), and the use of graft materials (e.g., left internal mammary artery). Concomitant CABG could be performed simultaneously due to CAD and coronary artery involvement by TAAD, which was easily distinguishable, with overlapping cases being categorized into both groups.
Patients were informed and asked to attend annual outpatient clinical visits, during which coronary and aortic CTA were prescribed. To improve response rates, telephone interviews were conducted to complement follow-up data collection from April to July 2024. The primary endpoint was mortality during the follow-up period. The secondary endpoint was graft occlusion, as confirmed by CTA images.
Definition of clinical outcomes
Operative mortality was defined as deaths occurring within 30 days of surgery (30-day mortality) or during the hospitalization period (in-hospital mortality). Midterm mortality was defined as all deaths occurring after the operative period (all-cause mortality, excluding operative mortality). Serious adverse events (SAEs) included operative mortality, the need for intra-aortic balloon pump or extracorporeal membrane oxygenation support post-surgery, and stroke.
Surgical procedures
All patients were positioned supine and underwent total arch replacement with frozen elephant trunk implantation, coupled with hypothermic circulatory arrest and selective cerebral perfusion as cerebral protection measures.
The decision to perform CABG to address stenotic lesions during TAAD repair was primarily based on the severity of coronary stenosis. Generally, CABG was performed for significant stenosis (≥50% stenosis in the left main coronary artery or ≥70% in any other major branches), while the decision for less severe stenosis was at the surgeon’s discretion, as there is currently no consensus. Most surgeons performed CABG for moderate stenosis (50–69%) located in non-major coronary branches, while others opted not to.
Following a median sternotomy, arterial cannulation was performed via the axillary and/or femoral arteries, and right atrial cannulation was established for venous return. If the patient was in stable condition, the left internal mammary artery may be harvested before incising the pericardium. Graft material was harvested, and cooling commenced with the initiation of cardiopulmonary bypass (CPB). Once the temperature reached 32 ℃, the ascending aorta was clamped and incised, followed by the administration of cardioplegia. If antegrade coronary ostial perfusion was not feasible, retrograde perfusion was employed. The great saphenous vein could also be anastomosed for graft perfusion.
Upon completion of the aortic root surgery, total arch replacement with frozen elephant trunk implantation was performed in accordance with our previously established protocols (12). After rewarming, the proximal ascending aorta was replaced and anastomosed to a four-branched prosthesis, followed by weaning from CPB. Meanwhile, the distal end of the left internal mammary artery was anastomosed to the left descending coronary artery, if feasible and necessary, and the proximal end of the vein graft was anastomosed to the prosthetic vascular grafts or native vessels.
Statistical analysis
R (version 4.3.1, R Foundation) and RStudio were utilized for statistical analysis. Continuous variables following a normal distribution were expressed as mean (standard deviation), while non-normally distributed data were expressed as median and interquartile range. Categorical variables were presented as number (percentage).
To address baseline discrepancies between the two groups, particularly disparities in coronary ostial involvement, inverse probability treatment weighting (IPTW) was applied to minimize confounding biases. Variables included in the IPTW analysis were age, gender, hypertension, diabetes, oral anticoagulant use, aortic regurgitation, bicuspid aortic valve, pericardial effusion, preoperative left ventricular ejection fraction, coronary malperfusion, condition of left and right coronary artery ostia, preoperative serum creatinine, total bilirubin, alanine transaminase, aspartate Transaminase, international normalized ratio, and troponin-I. Comparisons between the CABG and non-CABG groups were made before and after IPTW adjustments. Survival analysis was primarily conducted using Kaplan-Meier curves to assess the all-cause mortality and mid-term mortality. The log-rank test was used to compare survival probability and freedom from graft occlusion. P values of ≤0.05 (two-tailed) were considered statistically significant.
Results
A total of 270 patients with CAD who met the eligibility criteria were enrolled in the study. Among them, 156 patients who underwent concomitant CABG due to CAD were categorized into the CABG group, while 114 patients who did not undergo CABG for concurrent CAD were assigned to the non-CABG group.
Peri-operative data
Baseline characteristics are summarized in Table 1. A significant difference was observed in the prevalence of left coronary ostial compromise between the non-CABG and CABG groups (28.07% vs. 16.67%, P=0.04). Additionally, significant bias were noted in age, severe pericardial effusion, preoperative serum creatinine levels, and preoperative international normalized ratio (all P<0.05). Following IPTW adjustment, the baseline data were well-balanced, as illustrated in Figure 2.
Table 1
| Item | Overall study population | IPTW matched population | |||||
|---|---|---|---|---|---|---|---|
| Non-CABG (n=114) | CABG (n=156) | P value | Non-CABG (n=269) | CABG (n=258) | P value | ||
| Age (years) | 55.07±10.15 | 57.94±10.49 | 0.03 | 56.84±10.20 | 57.46±10.53 | 0.67 | |
| Male | 91 (79.82) | 120 (76.92) | 0.67 | 217 (80.67) | 202 (78.29) | 0.63 | |
| Body mass index (kg/m2) | 26.47±3.66 | 26.42±4.13 | 0.91 | 26.29±3.57 | 26.37±4.05 | 0.86 | |
| Coronary malperfusion | 13 (11.40) | 15 (9.62) | 0.78 | 22 (8.18) | 25 (9.69) | 0.66 | |
| Coronary ostial involvement | 71 (62.28) | 84 (53.85) | 0.21 | 146 (54.28) | 141 (54.65) | 0.96 | |
| Left coronary ostial involved | 32 (28.07) | 26 (16.67) | 0.04 | 61 (22.68) | 47 (18.22) | 0.47 | |
| Right coronary ostial involved | 69 (60.53) | 82 (52.56) | 0.24 | 144 (53.53) | 139 (53.88) | 0.97 | |
| Hypertension | 102 (89.47) | 136 (87.18) | 0.70 | 237 (88.10) | 224 (86.82) | 0.74 | |
| Diabetes | 12 (10.53) | 15 (9.62) | 0.97 | 33 (12.27) | 27 (10.47) | 0.72 | |
| Oral anticoagulant | 15 (13.16) | 19 (12.18) | 0.96 | 34 (12.64) | 35 (13.57) | 0.87 | |
| Bicuspid aortic valve | 4 (3.51) | 3 (1.92) | 0.67 | 6 (2.23) | 5 (1.94) | 0.78 | |
| Severe pericardial effusion | 9 (7.89) | 3 (1.92) | 0.04 | 11 (4.09) | 7 (2.71) | 0.53 | |
| Aortic insufficiency | 0.50 | 0.96 | |||||
| No regurgitation | 53 (46.49) | 69 (44.23) | 126 (46.84) | 116 (44.96) | |||
| Mild | 32 (28.07) | 37 (23.72) | 60 (22.30) | 64 (24.81) | |||
| Moderate | 21 (18.42) | 31 (19.87) | 50 (18.59) | 50 (19.38) | |||
| Severe | 8 (7.02) | 19 (12.18) | 33 (12.27) | 28 (10.85) | |||
| LVEF (%) | 60.95±5.44 | 60.62±4.16 | 0.57 | 60.74±5.36 | 60.63±4.03 | 0.88 | |
| Troponin-I (µg/L) | 0.10±0.32 | 0.07±0.30 | 0.57 | 0.07±0.28 | 0.07±0.30 | 0.91 | |
| SCr (μmol/L) | 101.18±54.79 | 86.99±39.49 | 0.01 | 94.06±45.42 | 90.81±43.59 | 0.57 | |
| CCr (mL/min) | 90.72±35.44 | 96.10±36.10 | 0.22 | 91.44±33.16 | 94.41±36.97 | 0.51 | |
| BUN (mmol/L) | 7.61±2.97 | 7.07±2.85 | 0.13 | 7.42±2.78 | 7.17±2.86 | 0.52 | |
| Total bilirubin (μmol/L) | 16.43±10.33 | 15.48±7.49 | 0.38 | 15.78±9.32 | 15.56±7.99 | 0.86 | |
| ALT (U/L) | 31.76±49.17 | 34.29±120.85 | 0.83 | 30.35±43.28 | 31.46±106.51 | 0.90 | |
| AST (U/L) | 37.56±61.56 | 37.37±119.30 | 0.99 | 33.39±51.15 | 34.82±105.30 | 0.87 | |
| INR | 1.13±0.14 | 1.09±0.13 | 0.02 | 1.11±0.12 | 1.10±0.15 | 0.81 | |
Data are presented as mean ± SD or n (%). ALT, alanine transaminase; AST, aspartate transaminase; BUN, blood urea nitrogen; CABG, coronary artery bypass grafting; CCr, creatinine clearance; INR, international normalized ratio; IPTW, inverse probability of treatment weight; LVEF, left ventricular ejection fraction; SCr, serum creatinine; SD, standard deviation.

Intraoperative details are presented in Table 2. Prior to IPTW adjustment, the proportion of patients undergoing CABG was significantly higher in the CABG group compared to the non-CABG group (100.00% vs. 11.40%, P<0.001). The CABG group had a greater number and proportion of grafts (P<0.001), more frequent CABG of the right coronary artery as a target vessel (P=0.001), and higher utilization of the left internal mammary artery as graft (P<0.001). Specifically, in the CABG group, 81 patients (51.92%) received one graft, 58 (37.18%) received two grafts, 16 (10.26%) received three grafts, and 1 (0.64%) received five grafts. In contrast, in the non-CABG group, CABG was performed exclusively due to compromised coronary artery ostia or unplanned revascularization, 10 patients (8.77%) received one graft, 3 (2.63%) received two grafts, and 1 (0.88%) received three grafts. The left internal mammary artery was used in 31 patients (19.87%) in the CABG group, compared to only 2 patients (1.75%) in the non-CABG group. These statistical differences largely remained significant after IPTW adjustment. Additionally, CPB time and cross-clamp time were significantly longer in the CABG group compared to the non-CABG group (217.99 vs. 192.49 minutes, P=0.006; 140.72 vs. 118.91 minutes, P<0.001).
Table 2
| Item | Overall study population | IPTW Matched population | |||||
|---|---|---|---|---|---|---|---|
| Non-CABG (n=114) | CABG (n=156) | P value | Non-CABG (n=269) | CABG (n=258) | P value | ||
| CABG | 13 (11.40) | 156 (100.00) | <0.001 | 51 (18.96) | 258 (100.00) | <0.001 | |
| CABG for coronary ostial involved | 4 (3.51) | 31 (19.87) | <0.001 | 27 (10.04) | 37 (14.34) | 0.54 | |
| The number of bypass grafts | <0.001 | <0.001 | |||||
| None | 100 (87.72) | 0 | 214 (79.55) | 0 | |||
| One-graft | 10 (8.77) | 81 (51.92) | 46 (17.10) | 142 (55.04) | |||
| Two-graft | 3 (2.63) | 58 (37.18) | 7 (2.60) | 89 (34.50) | |||
| Three-graft | 1 (0.88) | 16 (10.26) | 1 (0.37) | 25 (9.69) | |||
| Five-graft | 0 | 1 (0.64) | 0 | 2 (0.78) | |||
| SVG to RCA graft | 12 (10.53) | 43 (27.56) | 0.001 | 46 (17.10) | 66 (25.58) | 0.27 | |
| LIMA to LAD graft | 2 (1.75) | 31 (19.87) | <0.001 | 5 (1.86) | 53 (20.54) | <0.001 | |
| Surgical strategy of aortic root | |||||||
| Sinus reconstruction | 38 (33.33) | 56 (35.90) | 0.76 | 91 (33.83) | 95 (36.82) | 0.68 | |
| Bentall procedure | 20 (17.54) | 29 (18.59) | 0.95 | 53 (19.70) | 46 (17.83) | 0.71 | |
| David I procedure | 3 (2.63) | 1 (0.64) | 0.41 | 9 (3.35) | 1 (0.39) | 0.04 | |
| Aortic valve replacement | 0 | 5 (3.21) | 0.14 | 0 | 8 (3.10) | 0.03 | |
| Aortic valvuloplasty | 8 (7.02) | 10 (6.41) | >0.99 | 15 (5.58) | 17 (6.59) | 0.66 | |
| Reinitiation of CPB | 9 (7.89) | 8 (5.13) | 0.50 | 25 (9.29) | 11 (4.26) | 0.11 | |
| Concurrent branches procedure | 16 (14.04) | 25 (16.03) | 0.78 | 41 (15.24) | 40 (15.50) | 0.95 | |
| RBC transfusion (U) | 0.83±1.94 | 1.01±2.17 | 0.49 | 0.71±1.74 | 0.90±1.97 | 0.41 | |
| Plasma transfusion (mL) | 264.91±363.36 | 271.79±424.38 | 0.89 | 202.00±314.57 | 258.38±390.92 | 0.19 | |
| Platelet transfusion (units) | 0.99±0.56 | 0.97±0.47 | 0.71 | 1.00±0.55 | 0.99±0.48 | 0.82 | |
| Blood loss (mL) | 842.71±454.76 | 872.24±707.66 | 0.70 | 818.58±385.88 | 855.76±612.38 | 0.50 | |
| Autologous fusion (mL) | 275.26±135.33 | 299.78±243.92 | 0.33 | 267.43±109.35 | 294.31±214.52 | 0.14 | |
| Operative time (min) | 390.66±123.94 | 424.60±113.41 | 0.02 | 388.80±120.39 | 417.42±107.62 | 0.07 | |
| CPB time (min) | 192.49±80.00 | 217.99±69.26 | 0.006 | 189.94±76.56 | 213.76±66.54 | 0.01 | |
| Cross-clamp time (min) | 118.91±47.41 | 140.72±45.27 | <0.001 | 117.52±44.69 | 138.18±45.20 | 0.001 | |
| HCA time (min) | 17.24±6.37 | 16.03±6.29 | 0.12 | 17.90±6.91 | 15.77±6.07 | 0.04 | |
Data are presented as mean ± SD or n (%). CABG, coronary artery bypass grafting; CPB, cardiopulmonary bypass; HCA, hypothermic circulatory arrest (lower extremity); IPTW, inverse probability of treatment weight; LAD, left anterior descending coronary artery; LIMA, left internal mammary artery; RCA, right coronary artery; RBC, red blood cell; SD, standard deviation; SVG, saphenous vein.
Postoperative outcomes are outlined in Table 3. After IPTW adjustment, the non-CABG group exhibited higher rates of operative mortality (4.46% vs. 0.39%, P=0.02), procedural myocardial infarction (10.04% vs. 3.49%, P=0.04), and SAEs (8.18% vs. 2.71%, P=0.048) compared to the CABG group.
Table 3
| Item | Overall study population | IPTW matched population | |||||
|---|---|---|---|---|---|---|---|
| Non-CABG (n=114) | CABG (n=156) | P value | Non-CABG (n=269) | CABG (n=258) | P value | ||
| Serious adverse events | 10 (8.77) | 4 (2.56) | 0.046 | 22 (8.18) | 7 (2.71) | 0.048 | |
| Operative mortality | 5 (4.39) | 1 (0.64) | 0.10 | 12 (4.46) | 1 (0.39) | 0.02 | |
| Procedural myocardial injury | 11 (9.65) | 7 (4.49) | 0.15 | 27 (10.04) | 9 (3.49) | 0.04 | |
| ECMO | 2 (1.75) | 1 (0.64) | 0.78 | 4 (1.49) | 2 (0.78) | 0.51 | |
| IABP | 3 (2.63) | 1 (0.64) | 0.41 | 11 (4.09) | 1 (0.39) | 0.051 | |
| Stroke | 4 (3.51) | 2 (1.28) | 0.42 | 6 (2.23) | 4 (1.55) | 0.67 | |
| Re-exploration | 6 (5.26) | 6 (3.85) | 0.80 | 12 (4.46) | 9 (3.49) | 0.71 | |
| Postoperative dialysis | 11 (9.65) | 13 (8.33) | 0.87 | 24 (8.92) | 19 (7.36) | 0.66 | |
| Pneumonia | 21 (18.42) | 32 (20.51) | 0.79 | 42 (15.61) | 52 (20.16) | 0.38 | |
| Tracheostomy | 1 (0.88) | 3 (1.92) | 0.85 | 2 (0.74) | 5 (1.94) | 0.44 | |
| Hs-TNI (ng/mL) | 8.76±23.84 | 7.63±16.60 | 0.65 | 12.26±36.52 | 7.22±14.44 | 0.39 | |
| Total bilirubin (μmol/L) | 37.70±19.52 | 40.29±19.97 | 0.29 | 36.14±18.57 | 40.26±20.17 | 0.12 | |
| ALT (U/L) | 55.57±148.31 | 66.15±297.32 | 0.73 | 42.39±105.35 | 56.26±254.99 | 0.47 | |
| AST (U/L) | 142.57±463.70 | 125.80±463.52 | 0.77 | 106.32±321.09 | 113.02±408.81 | 0.85 | |
| SCr-post (μmol/L) | 114.10±58.41 | 98.30±45.10 | 0.01 | 107.01±49.87 | 101.74±47.94 | 0.40 | |
| SCr-48h-peak (μmol/L) | 169.16±107.39 | 150.50±92.11 | 0.13 | 159.80±95.26 | 154.21±91.99 | 0.65 | |
| SCr-peak-post (μmol/L) | 195.90±156.74 | 189.39±174.21 | 0.75 | 182.50±140.66 | 192.96±176.86 | 0.60 | |
| BUN-post (mmol/L) | 9.79±3.31 | 9.14±3.13 | 0.10 | 9.47±3.21 | 9.28±3.20 | 0.68 | |
| BUN-48h-peak (mmol/L) | 17.41±5.53 | 17.72±6.36 | 0.68 | 16.82±5.05 | 17.95±6.49 | 0.13 | |
| BUN-peak-post (mmol/L) | 21.49±10.25 | 22.01±11.46 | 0.70 | 20.57±9.43 | 22.21±11.96 | 0.24 | |
| Ventilation time (hours) | 42.83±57.55 | 45.97±96.53 | 0.76 | 38.78±54.58 | 46.07±100.37 | 0.46 | |
| ICU stay (days) | 5.93±5.05 | 6.16±6.06 | 0.74 | 5.69±5.12 | 6.21±6.14 | 0.50 | |
| Hospital stays (days) | 12.98±6.57 | 15.37±8.35 | 0.01 | 13.64±7.24 | 15.17±8.21 | 0.16 | |
Data are presented as mean ± SD or n (%). ALT, alanine transaminase; AST, aspartate transaminase; BUN, blood urea nitrogen; CABG, coronary artery bypass grafting; ECMO, extracorporeal membrane oxygenation; Hs-TNI, high-sensitivity troponin I; IABP, intra-aortic balloon pump; ICU, intensive care unit; IPTW, inverse probability of treatment weight; SCr, serum creatinine; SD, standard deviation.
Survival analysis
Patients who were unreachable or did not attend outpatient follow-ups 3 months postoperatively were considered lost to follow-up. The median follow-up duration was 22.90 months (interquartile range: 23.97 months), with a response rate of 93.70%. Kaplan-Meier analysis revealed no significant differences in all-cause mortality between the two groups (log-rank P=0.58). The survival curve for the CABG group initially indicated better survival, followed by a decline compared to the non-CABG group, as shown in Figure 3A. Subsequent Kaplan-Meier survival analysis among surgical survivors indicated higher mid-term mortality in the CABG group (log-rank P=0.01), as depicted in Figure 3B. Notably, among surgical survivors, only eight deaths occurred, all within the CABG group. Cardiac dysfunction was the primary cause of death in three patients. One patient succumbed to a bleeding event, another to an acute ischemia stroke and hernia, two to gastric cancer and reoperation failure, and the cause of death remained unspecified in the final case. Among the CABG group (n=156), 84.61% of patients had documented coronary CTA. The incidence of graft occlusion was 25.96% at 2 years and 62.22% at 4 years (Figure 4). Out of a total of 269 grafts, 187 had their distal end anastomosed to a prosthesis, while 82 were either anastomosed to native vessels (n=49) or involved LIMA-LAD grafts (n=33). Among the grafts anastomosed to a prosthesis, 50 cases of occlusion were observed, compared to only 11 occlusions in the 82 grafts connected to native vessels, showing a statistically significant difference (P=0.02).


Discussion
The overview of the study and its key findings are illustrated in Figure 5. Our study demonstrates that performing CABG to address concurrent CAD in TAAD patients significantly reduces operative mortality, which was further validated through IPTW matching. However, midterm mortality was notably higher in the CABG group, accompanied by a high incidence of graft occlusion during the follow-up period.
The incidence of CAD in TAAD patients has long been underrecognized. Databases such as International Registry of Acute Aortic Dissection (IRAD) do not provide detailed data on the prevalence of concurrent CAD, possibly because coronary CTA is not routinely performed across institutions. A study has indicated that only 63% of patients in the IRAD database had documented aortic CTA, with even fewer having documented coronary CTA (15). Recently, a nationwide cohort study reported CAD prevalence of as high as 46.4% (2). Therefore, it is crucial to determine whether concomitant CABG is necessary for patients with both CAD and TAAD and to understand its impact on clinical outcomes. These questions remain unresolved due to the limited number of relevant studies with small sample sizes. However, as one of the largest studies to date, our research provides comprehensive data on nearly all inpatient cases, with patients who underwent surgery all having documented coronary CTA, thereby offering the strongest level of evidence currently available.
Our findings suggest that concomitant CABG for CAD during TAAD surgery reduces perioperative mortality, likely by enhancing coronary blood flow and decreasing the incidence of perioperative myocardial infarction. This contrasts with other studies where CABG was considered a risk factor for increased mortality (16,17). The discrepancy may stem from differing inclusion criteria; this study exclusively enrolled TAAD patients with concurrent CAD, whereas those studies included all TAAD patients and stratified them based on whether they underwent CABG.
Among surgical survivors, the midterm mortality rate was significantly higher in the CABG group, with most deaths attributable to cardiac and bleeding events. Additionally, a large proportion of TAAD patients who underwent concomitant CABG for CAD experienced graft occlusion during the follow-up period. This is largely attributed to the fact that the majority of graft materials were great saphenous veins, which were anastomosed to the prosthetic vascular graft (18,19). Previous research has shown that vein graft failure is associated with severe morbidity, including recurrent angina, myocardial infarction, and higher mortality rates (20). Moreover, the natural progression of coronary artery stenosis is accelerated in grafted coronary arteries compared to nongrafted ones, particularly with saphenous vein grafts (7). Thus, the grafts, being mostly saphenous veins anastomosed to the prosthetic conduit, are prone to occlusion, leading to adverse coronary events and substantially impacting patient prognosis. Therefore, we suggest that the proximal end of the graft should be anastomosed to the native supra-aortic branches rather than to the prosthesis.
Study limitations
There are several limitations in this study. We did not stratify stenosis severity into categories such as moderate (50–69%) and severe (≥70%), which may lead to varying conclusions across specific subgroups. Additionally, only eight deaths occurred during the follow-up period, all within the CABG group, underscoring the need for research with larger sample sizes and extended follow-up to further validate our findings. Furthermore, our surgeon’s preference for performing CABG in cases of less severe coronary stenosis resulted in a higher proportion of CABG procedures among the study population, which may not reflect practices at other centers, emphasizing the need for multicenter, prospective studies. Despite these limitations, our study addresses the issues discussed earlier, revealing the short- and midterm mortality associated with CABG in TAAD patients with concurrent CAD and highlighting the incidence of graft occlusion following concomitant CABG in this patient population.
Conclusions
Concomitant CABG for CAD in TAAD patients significantly reduces operative mortality but is associated with higher midterm mortality. Given the high incidence of graft occlusion observed during the follow-up period, and the high proportion of proximal end anastomosed to the prosthesis conduit, we recommend against anastomosing the proximal end of grafts to the prosthetic conduit, as this may improve long-term patency, reduce cardiac events, and improve patient prognosis.
Acknowledgments
Long Deng served as an excellent statistical consultant for this article. To him, we express our sincere gratitude and best wishes.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1587/rc
Data Sharing Statement: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1587/dss
Peer Review File: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1587/prf
Funding: The study was funded by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1587/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was approved and monitored by the ethics committee of the Fuwai Hospital (approval No. 2021-1490) and individual consent for this retrospective analysis was waived.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Zhu Y, Lingala B, Baiocchi M, et al. Type A Aortic Dissection-Experience Over 5 Decades: JACC Historical Breakthroughs in Perspective. J Am Coll Cardiol 2020;76:1703-13. [Crossref] [PubMed]
- Goldstone AB, Chiu P, Baiocchi M, et al. Interfacility Transfer of Medicare Beneficiaries With Acute Type A Aortic Dissection and Regionalization of Care in the United States. Circulation 2019;140:1239-50. [Crossref] [PubMed]
- Rizzo RJ, Aranki SF, Aklog L, et al. Rapid noninvasive diagnosis and surgical repair of acute ascending aortic dissection. Improved survival with less angiography. J Thorac Cardiovasc Surg 1994;108:567-74; discussion 574-5. [Crossref] [PubMed]
- Creswell LL, Kouchoukos NT, Cox JL, et al. Coronary artery disease in patients with type A aortic dissection. Ann Thorac Surg 1995;59:585-90. [Crossref] [PubMed]
- Hayward PA, Zhu YY, Nguyen TT, et al. Should all moderate coronary lesions be grafted during primary coronary bypass surgery? An analysis of progression of native vessel disease during a randomized trial of conduits. J Thorac Cardiovasc Surg 2013;145:140-8; discussion 148-9. [Crossref] [PubMed]
- Sabik JF 3rd, Olivares G, Raza S, et al. Does grafting coronary arteries with only moderate stenosis affect long-term mortality? J Thorac Cardiovasc Surg 2016;151:806-811.e3. [Crossref] [PubMed]
- Raza S, Blackstone EH, Houghtaling PL, et al. Natural History of Moderate Coronary Artery Stenosis After Surgical Revascularization. Ann Thorac Surg 2018;105:815-21. [Crossref] [PubMed]
- Gaba P, Gersh BJ, Ali ZA, et al. Complete versus incomplete coronary revascularization: definitions, assessment and outcomes. Nat Rev Cardiol 2021;18:155-68. [Crossref] [PubMed]
- Alamanni F, Dainese L, Naliato M, et al. On- and off-pump coronary surgery and perioperative myocardial infarction: an issue between incomplete and extensive revascularization. Eur J Cardiothorac Surg 2008;34:118-26. [Crossref] [PubMed]
- Akhrass R, Bakaeen FG. Intraoperative graft patency validation: Friend or foe? JTCVS Tech 2021;7:131-7. [Crossref] [PubMed]
- Doenst T, Sousa-Uva M. How to deal with nonsevere stenoses in coronary artery bypass grafting - a critical perspective on competitive flow and surgical precision. Curr Opin Cardiol 2022;37:468-73. [Crossref] [PubMed]
- Caliskan E, de Souza DR, Böning A, et al. Saphenous vein grafts in contemporary coronary artery bypass graft surgery. Nat Rev Cardiol 2020;17:155-69. [PubMed]
- Vendramin I, Piani D, Lechiancole A, et al. Do oral anticoagulants impact outcomes and false lumen patency after repair of acute type A aortic dissection? J Thorac Cardiovasc Surg 2023;166:38-48.e4. [PubMed]
- Neri E, Toscano T, Papalia U, et al. Proximal aortic dissection with coronary malperfusion: presentation, management, and outcome. J Thorac Cardiovasc Surg 2001;121:552-60. [PubMed]
- Berretta P, Patel HJ, Gleason TG, et al. IRAD experience on surgical type A acute dissection patients: results and predictors of mortality. Ann Cardiothorac Surg 2016;5:346-51. [PubMed]
- Zhang K, Dong SB, Pan XD, et al. Concomitant coronary artery bypass grafting during surgical repair of acute type A aortic dissection affects operative mortality rather than midterm mortality. Asian J Surg 2021;44:945-51. [PubMed]
- Morjan M, Reser D, Savic V, et al. Concomitant Coronary Artery Bypass in Patients with Acute Type A Aortic Dissection. Semin Thorac Cardiovasc Surg 2022;34:410-6. [Crossref] [PubMed]
- Hwang HY, Lee KH, Han JW, et al. Equivalency of Saphenous Vein and Arterial Composite Grafts: 5-Year Angiography and Midterm Clinical Follow-Up. Ann Thorac Surg 2016;102:580-8. [Crossref] [PubMed]
- Kawamura A, Yoshioka D, Toda K, et al. An evaluation of the long-term patency of the aortocoronary bypass graft anastomosed to a vascular prosthesis. Eur J Cardiothorac Surg 2020;58:832-8. [Crossref] [PubMed]
- Harskamp RE, Lopes RD, Baisden CE, et al. Saphenous vein graft failure after coronary artery bypass surgery: pathophysiology, management, and future directions. Ann Surg 2013;257:824-33. [Crossref] [PubMed]


