
Krebs von den Lungen-6 as a biomarker for distinguishing between interstitial lung disease and interstitial lung abnormalities based on computed tomography findings
Highlight box
Key findings
• Serum Krebs von den Lungen-6 (KL-6) levels are significantly elevated in patients with interstitial lung disease (ILD) compared to those with interstitial lung abnormalities (ILA), indicating its potential as a diagnostic biomarker.
What is known and what is new?
• KL-6 is predominantly expressed by type II alveolar cells and is linked to the prognosis and severity of ILD.
• The study provides evidence that serum KL-6 concentrations are markedly elevated in clinically recognized and treated ILD compared to ILA. It also highlights the relationship between KL-6 levels and pulmonary function, specifically forced vital capacity.
What is the implication, and what should change now?
• Clinicians could consider incorporating KL-6 assessments into the diagnostic process for patients with suspected ILD to improve early detection and management strategies. Further research may be warranted to establish standardized protocols for KL-6 testing in clinical settings.
Introduction
Background
Interstitial lung disease (ILD) encompasses a diverse group of chronic lung conditions characterized by inflammation, fibrosis, and alterations in the lung interstitium (1). These conditions are classified based on their etiology, as well as their pathological and imaging features. In Korea, the incidence of ILD has been increasing, which is attributed to increased screening using computed tomography (CT) and an aging population. A study using claims data revealed that the prevalence of fibrotic ILD in South Korea increased from 18.8 per 100,000 individuals in 2011 to 38.9 per 100,000 individuals in 2018 (2).
High-resolution CT (HRCT) and pulmonary function tests (PFT) are crucial for diagnosing and assessing patients with ILD. Although lung biopsy remains the definitive diagnostic tool, its widespread use is limited due to its invasiveness. Thus, in recent times, biomarkers have gained significant attention owing to their potential as indicators of disease activity (3). One such biomarker, Krebs von den Lungen-6 (KL-6), a glycoprotein encoded by MUC1, was identified in 1989 (4,5). Elevated levels of KL-6 have been observed in various lung conditions, making it a reliable indicator of epithelial damage and fibrosis (4) and useful for disease detection, activity assessment, and prognostic prediction (5).
Rationale and knowledge gap
Interstitial lung abnormalities (ILA) are being increasingly detected with the widespread use of CT. Defined as alterations in non-dependent lung regions involving more than 5% of the lung area (6), ILA progress within 5 years in over 50% of cases, correlating with increased mortality rates (7,8). While ILA is observed in 3–10% of screening CT scans (9,10) there is no clear consensus regarding the follow-up of this condition in clinical practice.
Objective
This study aimed to compare patients with ILA and ILD—who were tested for KL-6 at the respiratory clinic of a single hospital—over time and evaluate the correlation between KL-6 levels and the clinical features of ILD. We present this article in accordance with the STARD reporting checklist (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1833/rc).
Methods
Study population and clinical data
This retrospective study included patients who visited the pulmonology clinic of a tertiary hospital between October 2022 and March 2023 and were examined for KL-6 levels. Clinical data collected from electronic medical records included information regarding age, sex, body mass index, comorbidities, smoking status, modified Medical Research Council (mMRC) dyspnea scale, and antifibrotic medication including pirfenidone and nintedanib. Additionally, we analyzed PFT and chest CT [including low-dose chest CT (LDCT) and HRCT] findings. PFT and CT were performed within 6 months of serum KL-6 sampling. Among the spirometric results, forced vital capacity (FVC) and diffusing capacity of the lungs for carbon monoxide (DLCO) were assessed and expressed as percentages of the normal predicted values.
Classification of ILD and ILA
The diagnosis of ILD was primarily based on clinical evaluations by the attending physician, differential diagnosis using CT, and considerations of comorbidities such as connective tissue disease. Herein, ILD subtypes included idiopathic pulmonary fibrosis (IPF), nonspecific interstitial pneumonia (NSIP), cryptogenic organizing pneumonia (COP), connective tissue disease-associated ILD (CTD-ILD), and unspecified subtypes, including pleuroparenchymal fibroelastosis (PPFE), combined pulmonary fibrosis and emphysema (CPFE), fibrosing mediastinitis, and unilateral fibrosis. IPF was diagnosed according to the Official American Thoracic Society (ATS)/European Respiratory Society (ERS)/Japanese Respiratory Society (JRS)/Latin American Thoracic Association (ALAT) Clinical Practice Guidelines [2022] (11). NSIP (12), COP (13), PPFE, CPFE, and fibrosing mediastinitis were diagnosed through discussion with a multidisciplinary team that included a radiologist, and CTD-ILD was diagnosed by rheumatologists who made the concomitant clinical diagnosis of ILD in conjunction with rheumatoid arthritis, Sjogren’s syndrome, and microscopic polyangiitis (MPA). ILA was defined according to the criteria of the Fleischner Society, referring to the incidental identification of non-dependent abnormalities, such as ground-glass opacities, reticular abnormalities, alveolar distortion, traction bronchiectasis, honeycombing, and non-emphysematous cysts, involving at least 5% of the lung zone, excluding ILD (6). Due to the ambiguity regarding the distinction between ILA and ILD in imaging findings, (14) we defined patients with ILA as those without clinically significant limiting dyspnea of mMRC grade 3 or higher and not undergoing pharmacologic therapy.
Semi-quantitative CT grade
Two experienced pulmonologists, both blinded to the clinical data, semi-quantitatively assessed the extent of ILD on CT images. Their evaluation was based on the grading system used in a previous study investigating the correlation between KL-6 levels and CTD-ILD (3). The grading scale was as follows: grade 1, 0–25% involvement; grade 2, 26–50% involvement; grade 3, 51–75% involvement; and grade 4, 76–100% involvement.
Measurement of KL-6
During patients’ outpatient visits, tests for KL-6 were performed, along with baseline serologic tests for ILD. KL-6 levels (U/mL) were measured using the high-quality Nanopia KL-6 immunoassay kit (Sekisui Medical Co., Ltd., Tokyo, Japan).
Statistical analysis
Descriptive statistics are presented as means with standard deviations (SDs). For the comparison of categorical variables, the Chi-squared test or Fisher’s exact test was employed, whereas Student’s t-test was used for continuous variables. Pearson correlation coefficients were used to assess the relationships between serum KL-6 levels and lung function parameters, including FVC and DLCO.
The diagnostic utilities of selected KL-6 levels for ILA were evaluated through the analysis of receiver operating characteristic (ROC) curves, including calculation of the area under the curve (AUC). In addition, the sensitivity and specificity of each KL-6 level was determined.
Statistical analyses and data management were conducted using Stata version 17.0 (StataCorp LLC, College Station, TX, USA). Statistical significance was set at P<0.05.
Ethical approval
This study was conducted in accordance with the Declaration of Helsinki (as revised in 2013) and approved by the Institutional Review Board of Veterans Health Service (VHS) Medical Center (IRB No. VHS 2023-02-002). Due to its retrospective nature, the requirement for informed consent was waived.
Results
Clinical characteristics of patients with ILD
This study included 147 patients; 17 patients (11.6%) were clinically diagnosed with ILA. Patients with ILA and ILD were similar in terms of age, sex, body mass index, and comorbidities such as cardiovascular diseases, diabetes, history of lung cancer, and chronic kidney disease. However, the ILD group had a higher prevalence of chronic kidney disease (26.2%, P=0.07) and a lower proportion of never-smokers (15.0% vs. 35.3%, P=0.08), although these differences were not statistically significant (Table 1).
Table 1
| Variables | Total (n=147) | ILD (n=130) | ILA (n=17) | P value |
|---|---|---|---|---|
| Age (years) | 77.1±4.2 | 77.1±4.3 | 77.2±3.1 | 0.92 |
| Sex (female) | 4 (2.7) | 4 (3.1) | 0 | 0.99 |
| BMI (kg/m2) | 24.3±2.8 | 24.2±2.9 | 25.1±2.6 | 0.24 |
| Comorbidities | ||||
| Cardiovascular | 88 (59.9) | 80 (61.5) | 8 (47.1) | 0.25 |
| DM | 52 (35.4) | 46 (35.4) | 6 (35.3) | 0.98 |
| Lung cancer | 19 (12.9) | 17 (13.1) | 2 (11.8) | 0.99 |
| CKD | 35 (23.8) | 34 (26.2) | 1 (5.9) | 0.07 |
| Smoking status† | 0.08 | |||
| Never smoker | 25 (17.4) | 19 (15.0) | 6 (35.3) | |
| Ex-smoker | 106 (73.6) | 95 (74.8) | 11 (64.7) | |
| Current smoker | 13 (9.0) | 13 (10.2) | 0 | |
| ILD subtype | ||||
| IPF | – | 92 (70.8) | – | – |
| NSIP | – | 16 (12.3) | – | – |
| COP | – | 1 (0.8) | – | – |
| CTD-ILD | – | 9 (6.9) | – | – |
| Not specified | – | 12 (9.2) | – | – |
| ILA subtype | ||||
| Fibrotic | – | – | 12 (70.6) | – |
| Nonfibrotic | – | – | 5 (29.4) | – |
| Antifibrotics | ||||
| Pirfenidone | – | 70 (53.8) | – | – |
| Nintedanib | – | 28 (21.5) | – | – |
Data are expressed as mean ± SD or n (%). †, data were missing from three patients. AUC, area under curve; BMI, body mass index; CKD, chronic kidney disease; COP, cryptogenic organizing pneumonia; CTD-ILD, connective tissue disease-associated interstitial lung disease; DM, diabetes mellitus; ILD, interstitial lung disease; ILA, interstitial lung abnormality; IPF, idiopathic pulmonary fibrosis; NSIP, nonspecific interstitial pneumonia; SD, standard deviation.
In the ILD group, IPF was the most common diagnosis, observed in 92 patients (70.8%), followed by NSIP [16 patients (12.3%)], COP [1 patient (0.8%)], CTD-ILD [9 patients (6.9%)], and other conditions [12 patients (9.2%)]. Of these, 70 patients (53.8%) were taking pirfenidone and 28 patients (21.5%) were taking nintedanib (Table 1). Of all the patients with ILD, only two were diagnosed through pathology.
Among the patients with ILA, 12 (70.6%) had fibrotic ILA and 5 (29.4%) had non-fibrotic ILA.
KL-6 in patients with ILD and ILA
Figure 1A shows the differences in mean (SD) of KL-6 levels between the ILD and ILA groups [ILD 968.8 (901.9) vs. ILA 411.6 (298.8) U/mL, P<0.001]. However, no statistically significant differences in mean (SD) of KL-6 levels were observed among the ILD subgroups [IPF 931.4 (834.9) U/mL, NSIP 670.6 (377.4) U/mL, COP 1,434.3 U/mL (one case), CTD-ILD 1,394.5 (1,371.5) U/mL, and unspecified 987.4 (1,157.6) U/mL; one-way analysis of variance, P=0.06] (Figure 1B). In the comparison of the ILD and ILA groups, the ILD group exhibited higher mean (SD) of KL-6 levels [968.8 (901.9) vs. 411.6 (298.8) U/mL, P<0.001], and statistically significantly lower mean (SD) of FVC [2.79 (0.64) vs. 3.54 (0.59) L, P<0.001], lower mean (SD) of FVC (%) [67.1 (14.1) vs. 83.4 (12.5), P<0.001], and lower mean (SD) of DLCO (%) [70.1 (1.7) vs. 86.6 (22.4), P=0.002]. Regarding mMRC grades, patients with grades of 3, 4, or higher were present only in the ILD group; however, no statistically significant difference was observed between the ILD and ILA groups. Additionally, we observed a significantly higher proportion of wide-range CT involvement (grades 3 and 4) in patients with ILD (P=0.009) (Table 2).

Table 2
| Characteristics | Total (n=147) | ILD (n=130) | ILA (n=17) | P value |
|---|---|---|---|---|
| KL-6 level (U/mL) | 905.5±872.1 | 968.8±901.9 | 411.6±298.8 | <0.001 |
| FVC (L) | 2.79±0.69 (n=143) | 2.79±0.64 (n=126) | 3.54±0.59 (n=17) | <0.001 |
| FVC (%) | 69.0±14.9 (n=143) | 67.1±14.1 (n=126) | 83.4±12.5 (n=17) | <0.001 |
| DLCO (%) | 72.0±20.1 (n=138) | 70.1±1.7 (n=122) | 86.6±22.4 (n=16) | 0.002 |
| mMRC dyspnea scale (n=132) | 0.92 | |||
| 0 | 19 (14.4) | 17 (14.4) | 2 (14.3) | |
| 1 | 85 (64.4) | 76 (64.4) | 9 (64.3) | |
| 2 | 19 (14.4) | 16 (13.6) | 3 (21.4) | |
| 3 | 5 (3.8) | 5 (4.2) | 0 | |
| 4 | 4 (3.0) | 4 (3.4) | 0 | |
| Semiquantitative CT grade | 0.009 | |||
| 1 (0–25%) | 73 (49.7) | 58 (44.6) | 15 (88.2) | |
| 2 (26–50%) | 54 (36.7) | 53 (40.8) | 1 (5.9) | |
| 3 (51–75%) | 18 (12.2) | 17 (13.1) | 1 (5.9) | |
| 4 (76–100%) | 2 (1.4) | 2 (1.5) | 0 |
Data are expressed as mean ± SD or n (%). CT, computed tomography; DLCO, diffusing capacity of the lungs for carbon monoxide; FVC, forced vital capacity; ILA, interstitial lung abnormality; ILD, interstitial lung disease; KL-6, Krebs von den Lungen-6; mMRC, modified Medical Research Council; SD, standard deviation.
Correlation between KL-6 and lung function parameters
Negative correlations were observed between KL-6 levels and lung function in the overall analysis. The correlations between FVC, DLCO, and KL-6 levels were significant and presented negative coefficient values [FVC coefficient (coef) −0.29, P<0.001; DLCO coef −0.006, P=0.008] (Figure 2). In the subgroup analysis for DLCO, the correlation coefficients were −0.003, with a P value of 0.09, for ILD, and −0.017, with a P value of 0.38, for ILA. Regarding FVC (mL), the coefficients were −0.22, with a P value of 0.001, for ILD and −0.84, with a P value of 0.091, for ILA.

Optimal cut-off value for ILA diagnosis
The optimal serum KL-6 cut-off value for distinguishing ILA was determined through a plot of sensitivity versus specificity and ROC curve analysis (Figure 3A), which yielded an optimal cut-off value of 419.3 U/mL This value demonstrated a sensitivity of 80.0% and a specificity of 70.6% [AUC: 0.75, 95% confidence interval (CI): 0.64–0.87] (Table 3). In our analysis, KL-6 levels to differentiate ILA from ILD resulted in an AUC of 0.82 (95% CI: 0.70–0.93) (Figure 3B). Additionally, for the prediction of radiologic involvement of more than 50% in chest CT, the AUC was 0.77 (95% CI: 0.64–0.88) (Figure 3C), and for predicting an FVC of less than 50%, the AUC was 0.77 (95% CI: 0.62–0.89) (Figure 3D).
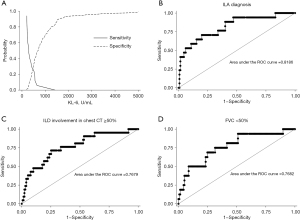
Table 3
| Cut-off value (U/mL) | AUC (95% CI) | Sensitivity (%) | Specificity (%) |
|---|---|---|---|
| 300 | 0.730 (0.606–0.854) | 93.9 | 52.9 |
| 410 | 0.727 (0.605–0.849) | 80.8 | 64.7 |
| 500 | 0.692 (0.577–0.814) | 68.5 | 70.6 |
AUC, area under curve; CI, confidence interval.
Discussion
Key findings
To our knowledge, this is the first study to demonstrate a significant difference in KL-6 levels between ILD and ILA in correlation with CT findings in an aged Korean population. While previous studies have focused on KL-6 concentrations in ILD (15) and CTD-ILD, this study was novel in its examination of KL-6 levels in ILA. We identified a potential cut-off value of KL-6 levels for distinguishing ILA from ILD, suggesting that a KL-6 level of 400 U/mL may indicate clinically significant ILD. This finding is in line with a previous meta-analysis indicating that KL-6 levels of 325 U/mL or higher can discriminate progressive ILD, corroborating our results (16).
Strengths and limitations
The principal strength of this study is the inclusion of patients from the real world. The study analyzed approximately 170 patients from a single institution, the majority of whom underwent pulmonary function tests and CT scans.
The limitations of our study include diagnosis of most patients through imaging without pathological confirmation and a lack of central consensus-based diagnostic criteria, the retrospective nature of the study, and the inclusion of only four women with ILD. This is a limitation related to the nature of clinical practice in this field, where older patients are often referred. The patients enrolled in the study were older adults, with a median age of 77.1 years, and included patients for whom the use of invasive diagnostic procedures, such as video-assisted thoracoscopic surgery or cryobiopsy, was difficult. Additionally, this study included a heterogeneous population of patients with ILD.
Furthermore, the small number of patients with ILA included herein is one of the limitations of this study. However, it is important to note that all patients with ILA in this study underwent KL-6 and PFT tests. We analyzed the data from these tests, and the correlations between KL-6 levels and FVC and DLCO in the study subjects demonstrated inverse relationships, which is consistent with the findings of previous studies; however, these findings were not statistically significant.
While including a homogenous ILD group with pathological confirmation is crucial for scientific analysis of causality, the heterogeneity of this study could be beneficial as it reflects real-world clinical practice.
Comparison with similar studies
The mean age of the patients included in our study was 77.1 years, and the majority (97.3%) were men. IPF was the most common form of ILD, accounting for 71% of all ILD cases in this study. The KL-6 levels observed herein were consistent with those reported in previous Korean studies on ILD (3,15,17). However, unlike earlier studies from other centers that focused on CTD-ILD (3,18,19), our study focused on ILA in an older, predominantly male population.
Data from the Korea IPF Cohort (KICO) registry, a Korean IPF cohort (20) indicate that approximately 76% of patients are male. In contrast, the proportion of males in our study is remarkably high. Furthermore, the prevalence of IPF has been reported to be approximately 50% in the Korean ILD study (2). However, a detailed analysis of the characteristics of our patient population reveals a distinct profile, with a notable proportion of older male patients with a history of smoking. The characteristics of VHS medical center that provides medical care for veterans could introduce a bias in our results.
The association of KL-6 with FVC has been documented in CTD-ILD (3), NSIP (21) and lung cancer (15) and a correlation between KL-6 and DLCO has been observed in CTD-ILD (22,23). Higher KL-6 levels were associated with restrictive physiology and decreased diffusion capacity, as evidenced by reduced FVC and DLCO (3,23) which is consistent with our findings.
Many studies have used KL-6 as a biomarker for ILD. In a study by d’Alessandro et al., which assessed KL-6 levels in patients with fibrotic ILD, the KL-6 cut-off value was 885 U/mL, and KL-6 was reported to be elevated in patients IPF, fibrotic hypersensitivity pneumonitis, and ILD associated with systemic sclerosis (SSc-ILD); moreover, similar to this study, inverse relationships between KL-6 and FVC and DLCO were identified (24). In patients with Sjögren’s syndrome, a study utilizing enzyme-linked immunosorbent assays reported a mean KL-6 level of 1,232 U/mL in the group that progressed to ILD and a mean KL-6 level of 764.5 U/mL in the group that did not progress to ILD (19). In another study of systemic sclerosis-related ILD, a KL-6 level exceeding 1,273 U/mL was significantly associated with progression to end-stage lung disease in a multivariate analysis (25). In a study of 69 patients with CTD-ILD, the Youden’s index of serum KL-6 for the diagnosis of ILD was 421.8 U/mL (23). Consistent with previous findings, guidelines suggest that KL-6 levels in patients with SSc-ILD and ILD associated with polymyositis/dermatomyositis (PM/DM-ILD) can be used as a marker to assess the severity of the disease (26). A European prospective cohort study comprising 179 patients reported a sensitivity of 82% and specificity of 86% for a cut-off value of 465 U/mL in 102 ILD patients compared with 77 controls (27). Similarly, in a meta-analysis, differences in KL-6 levels between ILD and non-ILD groups were identified, and an increase in KL-6 level was observed in cases of progressive ILD (28).
Studies on biomarkers for ILA are lacking. However, recent reports from the Framingham Heart Study and the Chronic Obstructive Pulmonary Disease Genetic Epidemiology (COPDGene) study (29) have shown a statistical association between ILA and growth differentiation factor 15—which belongs to the transforming growth factor-β superfamily (30) and is associated with aging (31)—with its levels being elevated in patients with ILA. This increase has also been reported in patients with IPF (32).
Explanations of findings
This study included 147 patients, of whom 130 (88.4%) were diagnosed with ILA. In the ILD group, IPF was the most common diagnosis, observed in 92 patients (70.8%). KL-6 levels were significantly higher in the ILD group, with a mean of 968.8 U/mL compared to 411.6 U/mL in the ILA group (P<0.001). The ILD group also exhibited lower FVC and DLCO values, and only patients with ILD had mMRC grades of 3 or higher. These findings suggest that KL-6 levels reflect the progression of fibrosis in the lungs of patients with ILD (33). KL-6 levels showed a negative correlation with lung function, and the optimal KL-6 cut-off for distinguishing ILA was found to be 419.3 U/mL, with a sensitivity of 80.0% and specificity of 70.6%.
Implications and actions needed
Herein, we aimed to use KL-6, a well-studied biomarker of ILD, to investigate the differences between ILA and ILD in a clinical setting. This study, which was conducted on a population of older Korean individuals diagnosed with ILD or idiopathic lung inflammation (ILA), consisted of mostly male subjects. Through clinical and radiological evaluation, the study suggests cut-off values for KL-6 and its application as a biomarker for patients requiring intensive treatment in real-world practice.
Conclusions
In summary, serum KL-6 levels were higher in patients with clinically diagnosed and treated ILD than in those with ILA. Elevated KL-6 levels were associated with lower FVC (%) and DLCO (%), suggesting their potential utility in distinguishing ILD patients requiring treatment.
Acknowledgments
We extend our sincere gratitude to Researcher Lee Young at the VHS Medical Research Institute for his invaluable statistical consultation.
Footnote
Reporting Checklist: The authors have completed the STARD reporting checklist. Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1833/rc
Data Sharing Statement: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1833/dss
Peer Review File: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1833/prf
Funding: This study was supported by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1833/coif). J.H.S. and H.R.K. report that this study was supported by a VHS medical research grant (grant No. VHSMC23003). The other author has no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. This study was conducted in accordance with the Declaration of Helsinki (as revised in 2013) and approved by the Institutional Review Board of VHS Medical Center (IRB No. VHS 2023-02-002). Due to its retrospective nature, the requirement for informed consent was waived.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Antoine MH, Mlika M. Interstitial Lung Disease. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2023.
- Joung KI, Park H, Park S, et al. Nationwide epidemiologic study for fibrosing interstitial lung disease (F-ILD) in South Korea: a population-based study. BMC Pulm Med 2023;23:98. [Crossref] [PubMed]
- Lee JS, Lee EY, Ha YJ, et al. Serum KL-6 levels reflect the severity of interstitial lung disease associated with connective tissue disease. Arthritis Res Ther 2019;21:58. [Crossref] [PubMed]
- Nakajima H, Harigai M, Hara M, et al. KL-6 as a novel serum marker for interstitial pneumonia associated with collagen diseases. J Rheumatol 2000;27:1164-70.
- Ishikawa N, Hattori N, Yokoyama A, et al. Utility of KL-6/MUC1 in the clinical management of interstitial lung diseases. Respir Investig 2012;50:3-13. [Crossref] [PubMed]
- Hatabu H, Hunninghake GM, Richeldi L, et al. Interstitial lung abnormalities detected incidentally on CT: a Position Paper from the Fleischner Society. Lancet Respir Med 2020;8:726-37. [Crossref] [PubMed]
- Putman RK, Gudmundsson G, Axelsson GT, et al. Imaging Patterns Are Associated with Interstitial Lung Abnormality Progression and Mortality. Am J Respir Crit Care Med 2019;200:175-83. [Crossref] [PubMed]
- Putman RK, Hatabu H, Araki T, et al. Association Between Interstitial Lung Abnormalities and All-Cause Mortality. JAMA 2016;315:672-81. [Crossref] [PubMed]
- Jin GY, Lynch D, Chawla A, et al. Interstitial lung abnormalities in a CT lung cancer screening population: prevalence and progression rate. Radiology 2013;268:563-71. [Crossref] [PubMed]
- Lee JE, Chae KJ, Suh YJ, et al. Prevalence and Long-term Outcomes of CT Interstitial Lung Abnormalities in a Health Screening Cohort. Radiology 2023;306:e221172. [Crossref] [PubMed]
- Raghu G, Remy-Jardin M, Richeldi L, et al. Idiopathic Pulmonary Fibrosis (an Update) and Progressive Pulmonary Fibrosis in Adults: An Official ATS/ERS/JRS/ALAT Clinical Practice Guideline. Am J Respir Crit Care Med 2022;205:e18-47. [Crossref] [PubMed]
- Travis WD, Costabel U, Hansell DM, et al. An official American Thoracic Society/European Respiratory Society statement: Update of the international multidisciplinary classification of the idiopathic interstitial pneumonias. Am J Respir Crit Care Med 2013;188:733-48. [Crossref] [PubMed]
- Raghu G, Meyer KC. Cryptogenic organising pneumonia: current understanding of an enigmatic lung disease. Eur Respir Rev 2021;30:210094. [Crossref] [PubMed]
- Chae KJ, Jin GY, Goo JM, et al. Interstitial Lung Abnormalities: What Radiologists Should Know. Korean J Radiol 2021;22:454-63. [Crossref] [PubMed]
- Cho EJ, Park KJ, Ko DH, et al. Analytical and Clinical Performance of the Nanopia Krebs von den Lungen 6 Assay in Korean Patients With Interstitial Lung Diseases. Ann Lab Med 2019;39:245-51. [Crossref] [PubMed]
- Zhang T, Shen P, Duan C, et al. KL-6 as an Immunological Biomarker Predicts the Severity, Progression, Acute Exacerbation, and Poor Outcomes of Interstitial Lung Disease: A Systematic Review and Meta-Analysis. Front Immunol 2021;12:745233. [Crossref] [PubMed]
- Park HK, Yoon CS, Na YO, et al. Serum KL-6 levels predict the occurrence and severity of treatment-related interstitial lung disease in lung cancer. Sci Rep 2023;13:18126. [Crossref] [PubMed]
- Kim YJ, Choe J, Moon SJ, et al. Blood KL-6 predicts prognosis in primary Sjögren's syndrome-associated interstitial lung disease. Sci Rep 2022;12:5343. [Crossref] [PubMed]
- Chiu YH, Chu CC, Lu CC, et al. KL-6 as a Biomarker of Interstitial Lung Disease Development in Patients with Sjögren Syndrome: A Retrospective Case-Control Study. J Inflamm Res 2022;15:2255-62. [Crossref] [PubMed]
- Jegal Y, Park JS, Kim SY, et al. Clinical Features, Diagnosis, Management, and Outcomes of Idiopathic Pulmonary Fibrosis in Korea: Analysis of the Korea IPF Cohort (KICO) Registry. Tuberc Respir Dis (Seoul) 2022;85:185-94. [Crossref] [PubMed]
- Sakamoto K, Taniguchi H, Kondoh Y, et al. Serum KL-6 in fibrotic NSIP: Correlations with physiologic and radiologic parameters. Respir Med 2010;104:127-33. [Crossref] [PubMed]
- Hu C, Wu C, Yang E, et al. Serum KL-6 is associated with the severity of interstitial lung disease in Chinese patients with polymyositis and dermatomyositis. Clin Rheumatol 2019;38:2181-7. [Crossref] [PubMed]
- He Q, Tang Y, Huang J, et al. The value of KL-6 in the diagnosis and assessment of interstitial lung disease. Am J Transl Res 2021;13:9216-23.
- d'Alessandro M, Conticini E, Bergantini L, et al. Krebs von den Lungen-6 as biomarker of the new progressive fibrotic phenotype of interstitial lung disease. Tissue Cell 2024;90:102516. [Crossref] [PubMed]
- Salazar GA, Kuwana M, Wu M, et al. KL-6 But Not CCL-18 Is a Predictor of Early Progression in Systemic Sclerosis-related Interstitial Lung Disease. J Rheumatol 2018;45:1153-8. [Crossref] [PubMed]
- Kondoh Y, Bando M, Kawahito Y, et al. Identification and management of interstitial lung disease associated with systemic sclerosis (SSc-ILD), rheumatoid arthritis (RA-ILD), and polymyositis/dermatomyositis (PM/DM-ILD): development of expert consensus-based clinical algorithms. Expert Rev Respir Med 2024;18:447-56. [Crossref] [PubMed]
- Millan-Billi P, Castellví I, Martinez-Martinez L, et al. Diagnostic Value of Krebs von den Lungen (KL-6) for Interstitial Lung Disease: A European Prospective Cohort. Arch Bronconeumol 2024;60:350-5. [Crossref] [PubMed]
- Wang C, Wang Q, Liu T, et al. Krebs von den Lungen-6 (KL-6) as a diagnostic marker for pulmonary fibrosis: A systematic review and meta-analysis. Clin Biochem 2023;114:30-8. [Crossref] [PubMed]
- Sanders JL, Putman RK, Dupuis J, et al. The Association of Aging Biomarkers, Interstitial Lung Abnormalities, and Mortality. Am J Respir Crit Care Med 2021;203:1149-57. [Crossref] [PubMed]
- Bootcov MR, Bauskin AR, Valenzuela SM, et al. MIC-1, a novel macrophage inhibitory cytokine, is a divergent member of the TGF-beta superfamily. Proc Natl Acad Sci U S A 1997;94:11514-9. [Crossref] [PubMed]
- Tanaka T, Biancotto A, Moaddel R, et al. Plasma proteomic signature of age in healthy humans. Aging Cell 2018;17:e12799. [Crossref] [PubMed]
- Zhang Y, Jiang M, Nouraie M, et al. GDF15 is an epithelial-derived biomarker of idiopathic pulmonary fibrosis. Am J Physiol Lung Cell Mol Physiol 2019;317:L510-21. [Crossref] [PubMed]
- Jehn LB, Costabel U, Boerner E, et al. Serum KL-6 as a Biomarker of Progression at Any Time in Fibrotic Interstitial Lung Disease. J Clin Med 2023;12:1173. [Crossref] [PubMed]


