
Nomogram for predicting the prognosis of metastatic thymic epithelial tumors
Highlight box
Key findings
• Due to the rare incidence, disease outcomes, and possible prognostic factors are limited and inconsistent, particularly those associated with metastatic thymic epithelial tumors (TETs). We developed a nomogram model for metastatic TETs supporting survival estimation, the first one as far as we know.
What is known and what is new?
• Several survival predictors for resectable TETs have been proposed such as World Health Organization (WHO) histologic classification, stage of disease, the extent of resection, and tumor size. Nomograms have been widely used to predict the survival of cancer patients. Several prognostic nomogram models for resectable TETs or Masaoka-Koga stage I–IV thymomas have been developed.
• There are few reported prognostic factors of unresectable TETs and there is no prognostic nomogram model for patients with metastatic TETs.
What is the implication, and what should change now?
• In this study, WHO histologic classification, Masaoka-Koga stage, Karnofsky performance status score, and baseline serum albumin level were identified as the independent prognostic factors for overall survival in the patients with metastatic TETs. This study developed a nomogram for effectively predicting the prognosis of metastatic TETs, which can help to make clinical decisions on site more friendly and efficient. Patients in the high-risk group should be considered to be administered with anti-tumor drugs with sufficient potency, as well as providing with prompt and multi-disciplinary treatment with close surveillance.
Introduction
Thymic epithelial tumors (TETs) originate in the thymus which include thymoma and thymic carcinoma (1,2). They are rare tumors with low incidence (1.3–3.8/1,000,000) (3,4). TETs have a variable presentation, manifesting concurrently with myasthenia gravis (MG), with local symptoms, or asymptomatically as a mediastinal mass on chest radiography (5). The World Health Organization (WHO) histologic classification is used to categorize different histologic subtypes of TETs. Thymoma can be classified as type A (including an atypical variant), type AB, and type B thymoma (separated into B1, B2, and B3 thymomas). Thymic carcinoma is categorized as type C according to the WHO histologic classification, although it differs greatly from thymoma (1,2). Although there are several staging systems, the Masaoka-Koga staging system based on primary tumor extension and the degree of involvement of the surrounding organs has been the most widely accepted to predict the prognosis for both thymomas and thymic carcinomas (6,7). Due to the rare incidence, disease outcomes, and possible prognostic factors are limited and inconsistent, particularly those associated with metastatic TETs.
Several survival predictors for resectable TETs have been proposed such as WHO histologic classification, stage of disease, the extent of resection, and tumor size (8-10). However, there are few reported prognostic factors of unresectable TETs. Yang et al. reported that patients with lymph node involvement only had better survival than those with distant metastases in patients with stage IVb thymic carcinomas (11). Okuma et al. reported that the site of metastatic involvement affects the survival outcome of patients with advanced thymic carcinoma receiving palliative-intent chemotherapy (12).
Nomograms have been widely used to predict the survival of cancer patients. Several prognostic nomogram models for resectable TETs or Masaoka-Koga stage I–IV thymomas have been developed (13-16). However, there is no prognostic nomogram model for patients with metastatic TETs. Our study aims to evaluate the association between clinicopathological factors and overall survival (OS) in patients with metastatic TETs and develop a nomogram model to effectively predict the prognosis of this disease population by utilizing these factors. We present this article in accordance with the TRIPOD reporting checklist (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1837/rc).
Methods
Patients
Patients with histologically confirmed metastatic TETs at Sun Yat-sen University Cancer Center (SYSUCC) between July 2000 and November 2021 were retrospectively included. For the purpose of this study, patients were included if they had (I) histologically confirmed TETs; (II) distant metastases; and (III) palliative anti-tumor treatment. Patients with previous malignancy were excluded. All enrolled patients were restaged according to the Masaoka-Koga staging system. Clinical and pathological characteristics were collected from the medical chart of eligible cases. Follow-up information was obtained by telephone interview or from medical chart.
Ethical statement
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). Approval was obtained from the ethics committee of Sun Yat-sen University Cancer Center, Guangzhou, China (No. B2021-209). Written informed consent was waived by the ethics committee due to the anonymized retrospective nature of the analysis.
Statistical analysis
Statistical analysis was performed by SPSS version 25.0 (IBM Corp., Armonk, NY, USA) and GraphPad Prism version 8 (GraphPad Software Inc., San Diego, CA, USA). These software packages were utilized for the descriptive data summary, univariate analysis, and multivariate analysis. R statistical packages (version 4.1.2; https://www.r-project.org/) [foreign, rms, time receiver operating characteristic (ROC), car, and ggDCA] were used to construct a nomogram, calculate the concordance index (C-index), generate the calibration curves and decision curve analysis (DCA) curves for internal validation.
The continuous data were converted to binary variables: white blood cell count, neutrophil count, lymphocyte count, platelet count, and C-reactive protein according to the upper limit of reference value; hemoglobin and albumin (ALB) according to the lower limit of reference value; neutrophil-lymphocyte ratio (NLR) and platelet-lymphocyte ratio (PLR) according to the best cutoff value by receiver operating characteristic (ROC) curves. An individual with body mass index below 18.5 kg/m2 was classified as being underweight (17). OS was defined as the time from the date of diagnosis of metastatic TETs to the date of death due to any cause or censoring at the date of data cutoff (January 30, 2022).
Univariate analysis was performed by a log-rank test. Variables with P values of less than 0.05 were further taken into the multivariate Cox proportional hazard model. In the multivariate Cox regression analysis, P values less than 0.05 were considered statistically significant. Clinicopathologic variables with statistical significance in the multivariate Cox regression were incorporated into the nomogram showing 2- and 3-year OS rates. The nomogram was used to calculate the prognostic risk score for each patient. According to the best cutoff value by ROC curves, patients’ scores were categorized into low-risk group and high-risk group. Their stratification effect was shown by the Kaplan-Meier curve.
Results
Patient characteristics
A total of 254 patients were enrolled into the analysis. Demography and disease characteristics are shown in Table 1. The median age was 49 years (range, 10–78 years) and 135 patients (53.15%) were aged younger than 50 years. The male patients were accounted for 61.42% (156/254). Patients with a Karnofsky performance status (KPS) score of 90–100 was accounted for 79.92% (203/254). Twenty-one (8.27%) patients had a MG history. According to WHO histologic classification system, 23 cases were type A or AB thymomas (9.06%); 106 cases were type B1, B2, or B3 thymomas (41.73%); and 125 cases were thymic carcinomas (49.21%). One hundred and ten (43.31%) patients were at Masaoka-Koga stage IVa while 144 (56.69%) patients were at Masaoka-Koga stage IVb. ALB levels were decreased (<40 g/L) in 69 (27.17%) patients.
Table 1
| Characteristics | Value |
|---|---|
| Age (years) | 49 [10–78] |
| ≤50 | 135 (53.15) |
| >50 | 119 (46.85) |
| Gender | |
| Male | 156 (61.42) |
| Female | 98 (38.58) |
| Smoking history | |
| No | 191 (75.20) |
| Yes | 63 (24.80) |
| Concurrent disease | |
| No | 167 (65.75) |
| Yes | 87 (34.25) |
| Family history of malignant tumor | |
| No | 233 (91.73) |
| Yes | 21 (8.27) |
| Underweight | |
| No | 224 (88.19) |
| Yes | 30 (11.81) |
| Myasthenia gravis history | |
| No | 233 (91.73) |
| Yes | 21 (8.27) |
| KPS score | |
| 90–100 | 203 (79.92) |
| 70–80 | 51 (20.08) |
| WHO histologic classification | |
| A–AB | 23 (9.06) |
| B1–B3 | 106 (41.73) |
| C | 125 (49.21) |
| Masaoka-Koga stage | |
| IVa | 110 (43.31) |
| IVb | 144 (56.69) |
| Metastatic site | |
| Intrathoracic only | 146 (57.48) |
| Intrathoracic and extrathoracic | 108 (42.52) |
| White blood cell count | |
| ≤10.0×109/L | 194 (76.38) |
| >10.0×109/L | 60 (23.62) |
| Neutrophils | |
| ≤6.3×109/L | 193 (75.98) |
| >6.3×109/L | 61 (24.02) |
| Lymphocytes | |
| ≤3.2×109/L | 230 (90.55) |
| >3.2×109/L | 24 (9.45) |
| Neutrophil-lymphocyte ratio | |
| ≤5.8 | 213 (83.86) |
| >5.8 | 41 (16.14) |
| Platelet count | |
| ≤300×109/L | 159 (62.60) |
| >300×109/L | 95 (37.40) |
| Platelet-lymphocyte ratio | |
| ≤212 | 171 (67.32) |
| >212 | 83 (32.68) |
| Hemoglobin | |
| <130 g/L | 114 (44.88) |
| ≥130 g/L | 140 (55.12) |
| Albumin | |
| <40 g/L | 69 (27.17) |
| ≥40 g/L | 185 (72.83) |
| C-reactive protein | |
| ≤3 mg/L | 120 (47.24) |
| >3 mg/L | 134 (52.76) |
Data are presented as median [range] or n (%). KPS, Karnofsky performance status; WHO, World Health Organization.
Survival analysis
With a median follow-up of 40.27 months, the median OS was 53.03 months [95% confidence interval (CI): 40.56–65.51]. The 2- and 3-year OS rates were 74.6% and 61.2%, respectively.
Univariate analysis showed that KPS score (P=0.01), WHO histologic classification (P=0.001), Masaoka-Koga stage (P=0.01), NLR (P=0.001), PLR (P=0.04) and ALB (P=0.001) were significant prognostic factors of OS (Table 2). These clinicopathological factors (P<0.05) in the univariate analysis were further adjusted by multivariate analysis. In multivariate analysis, only WHO histologic classification (P=0.006), Masaoka-Koga stage (P=0.01), KPS score (P=0.03), and ALB (P=0.01) remained independent prognostic factors of OS (Table 3). Of note, MG history was not a prognostic factor of OS. WHO histologic types B1-B3 or C, Masaoka-Koga stage IVb, KPS score 70–80 and ALB <40 g/L were significant factors leading to the worse OS.
Table 2
| Clinicopathological characteristics | Median overall survival (months) | HR (95% CI) | P value |
|---|---|---|---|
| Age (years) | 0.18 | ||
| ≤50 | 64.20 (41.80–86.60) | 1.00 | |
| >50 | 48.87 (35.39–62.35) | 1.30 (0.89–1.90) | |
| Gender | 0.91 | ||
| Male | 56.30 (34.84–77.76) | 1.00 | |
| Female | 48.97 (34.88–63.05) | 0.98 (0.67–1.44) | |
| Smoking history | 0.24 | ||
| No | 53.03 (39.20–66.87) | 1.00 | |
| Yes | 39.73 (12.97–66.50) | 1.28 (0.83–1.97) | |
| Concurrent disease | 0.31 | ||
| No | 52.10 (44.51–59.69) | 1.00 | |
| Yes | 64.20 (45.72–82.68) | 0.82 (0.55–1.20) | |
| Family history of malignant tumor | 0.37 | ||
| No | 52.10 (41.86–62.34) | 1.00 | |
| Yes | 66.90 (43.80–90.00) | 0.72 (0.38–1.35) | |
| Underweight | 0.19 | ||
| No | 53.03 (39.06–67.01) | 1.00 | |
| Yes | 48.97 (15.27–82.66) | 1.43 (0.78–2.62) | |
| Myasthenia gravis history | 0.20 | ||
| No | 48.97 (39.00–58.93) | 1.00 | |
| Yes | 74.17 (39.27–109.06) | 0.61 (0.33–1.14) | |
| KPS score | 0.01 | ||
| 90–100 | 56.57 (38.54–74.59) | 1.00 | |
| 70–80 | 31.87 (5.71–58.02) | 1.73 (1.05–2.84) | |
| WHO histologic classification | 0.001 | ||
| A–AB | 102.37 (67.05–137.68) | 1.00 | |
| B1–B3 | 64.20 (50.10–78.30) | 1.74 (0.82–3.72) | 0.15 |
| C | 34.70 (24.26–45.14) | 3.15 (1.48–6.73) | 0.003 |
| Masaoka-Koga stage | 0.01 | ||
| IVa | 68.33 (49.8–86.80) | 1.00 | |
| IVb | 42.93 (24.59–61.28) | 1.62 (1.11–2.37) | |
| Metastatic site | 0.32 | ||
| Intrathoracic | 59.00 (42.45–75.55) | 1.00 | |
| Intrathoracic and extrathoracic | 44.97 (27.61–62.33) | 1.21 (0.82–1.80) | |
| White blood cell count | 0.08 | ||
| ≤10.0×109/L | 53.57 (39.89–67.25) | 1.00 | |
| >10.0×109/L | 38.67 (8.55–68.80) | 1.46 (0.91–2.34) | |
| Neutrophils | 0.11 | ||
| ≤6.3×109/L | 53.57 (40.00–67.14) | 1.00 | |
| >6.3×109/L | 38.67 (23.66–53.68) | 1.41 (0.88–2.24) | |
| Lymphocytes | 0.95 | ||
| ≤3.2×109/L | 52.83 (39.27–66.40) | 1.00 | |
| >3.2×109/L | 68.33 (39.25–97.41) | 1.02 (0.54–1.91) | |
| Neutrophil-lymphocyte ratio | 0.001 | ||
| ≤5.8 | 59.00 (46.31–71.69) | 1.00 | |
| >5.8 | 31.77 (16.51–47.02) | 2.18 (1.16–4.09) | |
| Platelet count | 0.13 | ||
| ≤300×109/L | 56.30 (45.10–67.50) | 1.00 | |
| >300×109/L | 34.29 (15.10–53.30) | 1.36 (0.90–2.06) | |
| Platelet-lymphocyte ratio | 0.04 | ||
| ≤212 | 65.47 (49.91–81.03) | 1.00 | |
| >212 | 39.73 (25.00–54.46) | 1.52 (0.98–2.35) | |
| Hemoglobin | 0.07 | ||
| <130 g/L | 47.70 (34.94–60.47) | 1.00 | |
| ≥130 g/L | 68.33 (46.39–90.28) | 0.71 (0.48–1.04) | |
| Albumin | 0.001 | ||
| <40 g/L | 32.10 (20.43–43.77) | 1.00 | |
| ≥40 g/L | 65.47 (47.27–83.67) | 0.52 (0.33–0.81) | |
| C-reactive protein | 0.19 | ||
| ≤3 mg/L | 56.57 (44.44–68.69) | 1.00 | |
| >3 mg/L | 39.73 (11.89–67.58) | 1.29 (0.88–1.88) |
CI, confidence interval; HR, hazard ratio; KPS, Karnofsky performance status; WHO, World Health Organization.
Table 3
| Clinicopathological characteristics | HR (95% CI) | P value |
|---|---|---|
| WHO histologic classification | 0.006 | |
| A–AB | 1.00 | |
| B1–B3 | 1.89 (0.88–4.08) | 0.10 |
| C | 3.02 (1.40–6.52) | 0.005 |
| Masaoka-Koga stage | 0.01 | |
| IVa | 1.00 | |
| IVb | 1.62 (1.11–2.37) | |
| KPS score | 0.03 | |
| 90–100 | 1.00 | |
| 70–80 | 1.65 (1.05–2.60) | |
| Neutrophil-lymphocyte ratio | 0.15 | |
| ≤5.8 | 1.00 | |
| >5.8 | 1.51 (0.86–2.64) | |
| Platelet-lymphocyte ratio | 0.59 | |
| ≤212 | 1.00 | |
| >212 | 1.14 (0.72–1.80) | |
| Albumin | 0.01 | |
| <40 g/L | 1.00 | |
| ≥40 g/L | 0.58 (0.39–0.88) |
CI, confidence interval; HR, hazard ratio; KPS, Karnofsky performance status; WHO, World Health Organization.
Construction and validation of the nomogram
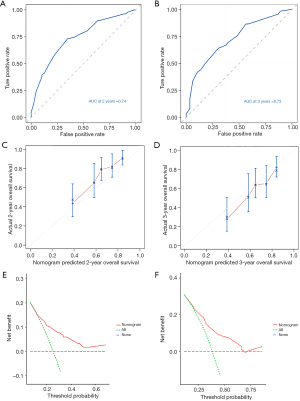
A nomogram to evaluate the prognostic factors of OS in the study population was established upon the outcome of multivariate analysis (Figure 1). To estimate the 2- and 3-year OS rates, we identified the score for each factor based on the point scale at the top of the nomogram and the sum of the points for each factor. The C-index was 0.68 (95% CI: 0.59–0.79). We also utilized the ROC curve to assess the performance of the nomogram. The area under the ROC curve (AUC) values at 2 and 3 years were 0.74 and 0.73, respectively, indicating that the nomogram exhibited good predictive accuracy (Figure 2A,2B). The calibration curves based on bootstrap resampling validation demonstrated the consistency between predicted and actual survival (Figure 2C,2D). The DCA curves of the nomogram have a high net benefit, which indicates that the nomogram demonstrates high clinical feasibility (Figure 2E,2F).

Risk stratification of OS
Patients were then categorized into the low-risk group (0–112 points) and the high-risk group (113 points or higher) respectively (Figure 3A) by the median score of 112 in the nomogram. The 1-, 2-, and 3-year OS rates were 97.5%, 87.3%, and 74.7% in the low-risk group while 83.9%, 59.8%, and 45.1% in the high-risk group, respectively. The median OS was significantly worse in the high-risk group than the low-risk group [28.60 versus 74.17 months, hazard ratio (HR): 2.44; 95% CI: 1.65–3.62; P<0.001] (Figure 3B).

Discussion
As patients with TETs usually do not present any clinical symptoms in the early stage, they are often in the advanced stage at initial diagnosis. It is of great clinical significance to identify patients of metastatic TETs with different prognosis thus providing precise medical interventions to prolong the survival of this disease population. Previous nomogram models were only used to predict relapse-free survival of patients with TETs after surgery or predict prognosis of thymoma patients only (13-16). Our study aimed to identify the independent prognostic factors of OS in patients with metastatic TETs and develop a nomogram predicting OS for these patients. In this study, by analyzing the clinicopathological and survival data of 254 patients with metastatic TETs in a single university cancer center, WHO histologic classification, Masaoka-Koga stage, KPS score, and baseline serum ALB level were found to be independent prognostic factors of OS in patients with metastatic TETs. We developed a nomogram that can effectively visually predict the 2- and 3-year OS rates of patients with metastatic TETs.
The WHO histologic classification was first recommended in 1999 and the most recent 5th version was published in 2021. There are no truly benign epithelial tumors of the thymus, but different subtypes exhibit varying biological behaviors and levels of clinical aggressiveness. The metastatic potential varies among thymic tumors, escalating from type A and AB thymomas, through B1, B2, and B3 thymomas, to the most aggressive form, thymic carcinomas. This variation in metastatic potential leads to different treatment strategies and follow-up schedules after diagnosis (18). In the realm of thymic malignancies, it is widely recognized that the prognosis of thymic carcinoma is generally more adverse compared to that of thymoma. This observation aligns with the findings of our study. However, the literature presents a discordance regarding the prognostic significance of the WHO histologic classification system for thymoma patients. A subset of studies has identified the WHO histologic classification as a robust prognostic indicator for thymoma, facilitating the stratification of patients and guiding the selection of optimal therapeutic strategies (9,13,19). In contrast, some studies have reported that this classification does not hold predictive value for disease-free survival or OS in thymoma patients (8,16). Historically, the prognostic relevance of the WHO histologic classification has predominantly been reported in the context of surgically treated patients. The results of our study appear to indicate that the WHO histologic classification may serve as a significant prognostic determinant of OS in patients with metastatic thymoma, although no statistically significant differences were observed between thymoma types A–AB and B1–B3. The results underscore the need for further investigation to elucidate the prognostic implications of the WHO histologic classification across diverse patient populations.
The Masaoka-Koga staging system was proposed in 1981 (20), and a modified version (Masaoka-Koga stage) was released in 1994, which is most commonly used (6). For resectable thymic TETs, complete surgical resection is recommended. Radiotherapy or chemotherapy can be employed as postoperative adjuvant treatment. In cases of advanced or unresectable TETs, the primary treatments are radiotherapy and chemotherapy (21). The different treatments lead to different prognoses. And the Masaoka stage served as a perfect predictor of prognosis in patients with early-stage or limited-stage TETs in the present study (9,16,22). The Masaoka-Koga stage classifies the pleural or pericardial dissemination as stage IVa disease and lymphatic or hematogenous metastasis as stage IVb disease. Our study found that the Masaoka stage also served as a perfect predictor of prognosis in patients with advanced TETs. Masaoka-Koga stage IVb was a more negative prognostic factor of OS than Masaoka-Koga stage IVa.
Ogawa et al. conducted a retrospective analysis of 40 patients with locally advanced stage thymic carcinoma treated with surgical resection or radiotherapy with or without chemotherapy and found that compared with patients with KPS score <70, patients with KPS score ≥70 had a decreased risk of death (23). Our study found that metastatic TETs with KPS score 70–80 had poorer OS than those with KPS score 90–100. Patients with good KPS scores can better tolerate treatment-related adverse reactions (TRAEs) and are more likely to get comprehensive treatment. Patients with low KPS scores tend to have poor performance status, poor treatment response, and difficulty in tolerating TRAEs. Therefore, it is suggested that patients with metastatic TETs should receive treatment at a good KPS score to achieve better treatment response and obtain improved survival time.
Our study found that the low baseline serum ALB level (<40 g/L) was a poor prognostic factor for OS of metastatic TETs, which is consistent with previous research (12,13). Hypoalbuminemia is a marker of poor nutritional status in patients, which may be caused by reduced food intake, the host response to the tumor, and anticancer therapies (24). The malnutrition contributes to the impairment of immune function, performance status, muscle function, and quality of life (24). Furthermore, malnutrition may work as a significant factor that can attenuate the efficacy of oncological interventions, augment the likelihood and severity of TRAEs, and exacerbate cancer-associated complications. The confluence of these comorbidities may collectively contribute to diminished survival outcomes in cancer patients. Patients are recommended to strengthen nutritional support and maintain good nutritional status for improved survival.
WHO histologic classification, Masaoka-Koga stage, KPS score, and baseline serum ALB level were all independent prognostic factors of OS in patients with metastatic TETs in our study, but their relative weights in the model were totally different. In terms of weight, the histological type held the greatest significance, followed by ALB levels and KPS scores, with the stage carrying the least weight. The possible reasons are as follows. Histological classification is associated with the aggressiveness of the tumor, and tumor aggressiveness is an important factor in determining prognosis of tumor patients. Staging is also a prognostic factor for tumors, but in this study, all included patients were in stage IV, and most of the treatments were palliative, resulting in less significant differences in prognosis, hence the lowest weight.
We developed a nomogram model for metastatic TETs supporting survival estimation, the first one as far as we know. The prognostic factors used to predict survival outcome are easily collected from the routine clinical data without extra cost. Furthermore, this easy-to-use tool can help make clinical decisions on-site more friendly and efficiently. Given that the prognosis of patients in the high-risk group is much poorer than that in the low-risk group, the following treatment strategy is recommended to the metastatic TET patients in the high-risk group: (I) anti-tumor drugs with sufficient potency and starting the treatment without delay; (II) close surveillance; (III) multi-disciplinary treatment.
The limitations of this study are as follows. First, since this study is a retrospective study conducted at a single center, selection bias is inevitable. Future studies can be conducted at multiple centers to reduce the occurrence of bias. Second, although this is a large sample size study to predict the prognosis of metastatic TETs, a large-scale external validation cohort with multi-center datasets is needed to further confirm prognostic predictivity of this nomogram model.
Conclusions
In this study, WHO histologic classification, Masaoka-Koga stage, KPS score, and baseline serum ALB level were identified as the independent prognostic factors for OS in the patients with metastatic TETs. This study developed a nomogram for effectively predicting the prognosis of metastatic TETs, which can help to make clinical decisions on site more friendly and efficiently. Patients in the high-risk group should be considered to be administered with anti-tumor drugs with sufficient potency, as well as providing with prompt and multi-disciplinary treatment with close surveillance.
Acknowledgments
The work was accepted for an E-Poster Presentation at the IASLC 2024 World Conference on Lung Cancer (WCLC 2024).
Footnote
Reporting Checklist: The authors have completed the TRIPOD reporting checklist. Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1837/rc
Data Sharing Statement: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1837/dss
Peer Review File: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1837/prf
Funding: None.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1837/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Approval was obtained from the ethics committee of Sun Yat-sen University Cancer Center, Guangzhou, China (No. B2021-209). The procedures used in this study adhere to the tenets of the Declaration of Helsinki (as revised in 2013). Written informed consent was waived by the ethics committee due to the anonymized retrospective nature of the analysis.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Marx A, Chan JKC, Chalabreysse L, et al. The 2021 WHO Classification of Tumors of the Thymus and Mediastinum: What Is New in Thymic Epithelial, Germ Cell, and Mesenchymal Tumors? J Thorac Oncol 2022;17:200-13. [Crossref] [PubMed]
- National Comprehensive Cancer Network. Clinical Practice Guidelines in Oncology. Thymomas and Thymic Carcinomas, Version 1. 2022.
- Engels EA. Epidemiology of thymoma and associated malignancies. J Thorac Oncol 2010;5:S260-5. [Crossref] [PubMed]
- Koizumi T, Otsuki K, Tanaka Y, et al. National incidence and initial therapy for thymic carcinoma in Japan: based on analysis of hospital-based cancer registry data, 2009-2015. Jpn J Clin Oncol 2020;50:434-9. [Crossref] [PubMed]
- Multidisciplinary Committee of Oncology, Chinese Physicians Association. Chinese guideline for clinical diagnosis and treatment of thymic epithelial tumors (2021 Edition). Chinese Journal of Oncology 2021;43:395-404. [Crossref] [PubMed]
- Koga K, Matsuno Y, Noguchi M, et al. A review of 79 thymomas: modification of staging system and reappraisal of conventional division into invasive and non-invasive thymoma. Pathol Int 1994;44:359-67. [Crossref] [PubMed]
- Detterbeck FC, Nicholson AG, Kondo K, et al. The Masaoka-Koga stage classification for thymic malignancies: clarification and definition of terms. J Thorac Oncol 2011;6:S1710-6. [Crossref] [PubMed]
- Zhao Y, Shi J, Fan L, et al. Surgical treatment of thymoma: an 11-year experience with 761 patients. Eur J Cardiothorac Surg 2016;49:1144-9. [Crossref] [PubMed]
- Yuan ZY, Gao SG, Mu JW, et al. Long-term outcomes of 307 patients after complete thymoma resection. Chin J Cancer 2017;36:46. [Crossref] [PubMed]
- Hashinokuchi A, Takamori S, Zhu J, et al. Prognostic Impact of Primary Tumor Size in Thymic Epithelial Tumor: An NCDB-Based Study. Ann Surg Oncol 2025;32:1662-9. [Crossref] [PubMed]
- Yang Y, Fan XW, Wang HB, et al. Stage IVb thymic carcinoma: patients with lymph node metastases have better prognoses than those with hematogenous metastases. BMC Cancer 2017;17:217. [Crossref] [PubMed]
- Okuma Y, Ko R, Shukuya T, et al. Prognostic factors for patients with metastatic or recurrent thymic carcinoma receiving palliative-intent chemotherapy. Lung Cancer 2020;148:122-8. [Crossref] [PubMed]
- Huang YY, Wu LL, Liu X, et al. Nomogram predict relapse-free survival of patients with thymic epithelial tumors after surgery. BMC Cancer 2021;21:847. [Crossref] [PubMed]
- Liu H, Gu Z, Qiu B, et al. A Recurrence Predictive Model for Thymic Tumors and Its Implication for Postoperative Management: a Chinese Alliance for Research in Thymomas Database Study. J Thorac Oncol 2020;15:448-56. [Crossref] [PubMed]
- Wang Y, Xu L, Du T, et al. A Nomogram Predicting Recurrence and Guiding Adjuvant Radiation for Thymic Carcinoma After Resection. Ann Thorac Surg 2018;106:257-63. [Crossref] [PubMed]
- Zhao M, Yin J, Yang X, et al. Nomogram to predict thymoma prognosis: A population-based study of 1312 cases. Thorac Cancer 2019;10:1167-75. [Crossref] [PubMed]
- Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet 2004;363:157-63. [Crossref] [PubMed]
- von der Thüsen J. Thymic epithelial tumours: histopathological classification and differential diagnosis. Histopathology 2024;84:196-215. [Crossref] [PubMed]
- Tseng YC, Tseng YH, Kao HL, et al. Long term oncological outcome of thymoma and thymic carcinoma - an analysis of 235 cases from a single institution. PLoS One 2017;12:e0179527. [Crossref] [PubMed]
- Masaoka A, Monden Y, Nakahara K, et al. Follow-up study of thymomas with special reference to their clinical stages. Cancer 1981;48:2485-92. [Crossref] [PubMed]
- Xu C, Zhang Y, Wang W, et al. Chinese expert consensus on the diagnosis and treatment of thymic epithelial tumors. Thorac Cancer 2023;14:1102-17. [Crossref] [PubMed]
- Koçer B, Kaplan T, Günal N, et al. Long-term survival after R0 resection of thymoma. Asian Cardiovasc Thorac Ann 2018;26:461-6. [Crossref] [PubMed]
- Ogawa K, Toita T, Uno T, et al. Treatment and prognosis of thymic carcinoma: a retrospective analysis of 40 cases. Cancer 2002;94:3115-9. [Crossref] [PubMed]
- Muscaritoli M, Corsaro E, Molfino A. Awareness of Cancer-Related Malnutrition and Its Management: Analysis of the Results From a Survey Conducted Among Medical Oncologists. Front Oncol 2021;11:682999. [Crossref] [PubMed]


