
Clinical characteristics of the early responders in the treatment of chronic cough
Highlight box
Key findings
• Upper airway cough syndrome (UACS) and baseline cough numeric rating scale (NRS) scores is associated with early responder to treatment of chronic cough (CC).
What is known and what is new?
• There is lack of data about early responder of CC treatment. There are many cough-measuring index such as severity or quality of life but it did not used comprehensively.
• We found that UACS and higher baseline cough NRS are associated with early response to treat.
What is the implication, and what should change now?
• In case of UACS of CC, shorter treatment duration can be considered. Comprehensive cough evaluation is needed, and controlling cough severity is important especially.
Introduction
A cough is a basic protective mechanism of the airway, but it is also one of the most common reasons for patients to visit hospitals (1). Physicians usually divide cough according to its duration: acute (<3 weeks), subacute (3 to 8 weeks), and chronic (≥8 weeks) (2). Among them, chronic cough (CC) is a major health problem worldwide (3,4). The well-known causes of CC are upper airway cough syndrome (UACS), asthma, eosinophilic bronchitis (EB), or gastroesophageal reflux disease (GERD) (5-7).
However, patients frequently complains unmet needs such as unclear diagnosis, unsatisfactory treatment response, and adverse events during treatment (8). For overcoming these problems, many cough guidelines recommend following the guideline and treating the possible causes of CC simultaneously more than 4 weeks (9-11). In this circumstance, some patients could not avoid the overuse of medication. To avoid the unnecessary exposure of medication, defining the early responder in treatment of CC is necessary because it is most reasonable way of reducing the treatment duration. Until recently, we did not know much about the contributing factors of early responder because of complicated pathophysiology of CC (12).
When we assess coughing, we measure various factors, including intensity, frequency, and its impact on the patient’s quality of life (QOL). Cough intensity can be measured by using the cough numeric rating scale (NRS). The Cough Assessment Test (COAT) was developed for simplifying the assessment of CC by the Korean Academy of Tuberculosis and Respiratory Diseases (KATRD), which represent cough QOL and frequency (13-18). We planned to use these measurement tools for defining early responder in treatment of CC and for evaluating the characteristics of them. We present this article in accordance with the STROBE reporting checklist (available at https://jtd.amegroups.com/article/view/10.21037/jtd-22-1374/rc).
Methods
Study design and participants
This study was a multi-center observational study which collected the data retrospectively. Patients were included from 15 university hospitals from March 2016 to February 2018. During that period, all participating hospitals performed unified evaluation and treatment of CC according to 2014 Korean cough guideline of KATRD. All patients were followed by two and four weeks after treatment. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was approved by the Institutional Review Board of the Catholic University of Korea, South Korea (No. XC19REDI0027) and individual consent for this retrospective analysis was waived.
CC was defined by cough more than 8 weeks. Among screened patients, only patients who (I) were aged more than 18 years old; (II) were categorized by UACS, asthma/EB, and GERD; and (III) were completed both cough NRS and COAT score questionnaire voluntarily, were included initially. Patients were excluded if they (I) had abnormal structural lung diseases on chest imaging; (II) had current pulmonary diseases, such as interstitial lung disease, chronic obstructive pulmonary diseases, previously known asthma, bronchiectasis, or lung cancer; (III) were taking angiotensin-converting enzyme inhibitor; (IV) were pregnant; (V) did not complete both cough NRS and COAT; (VI) were current smoker; and (VII) were diagnosed as iatrogenic cough or cough hypersensitivity syndrome that should only be diagnosed after conducting a thorough examination and excluding other possible causes. The patients with incomplete dataset were excluded for avoiding potential bias.
Diagnostic and management protocols of CC
All evaluations for diagnosis of CC were performed simultaneously according to the 2014 Korean cough guidelines of KATRD (1). The etiology of CC was determined based on suspected symptoms or positive evaluation tests. Simultaneous diagnoses were allowed according to the patient’s findings and symptoms. The details are as follows. UACS was diagnosed if the participants had post-nasal drip or mucosa showing a cobble stone appearance. Patients who were diagnosed with rhinitis, rhinosinusitis, or laryngopharyngitis were also classified as UACS. If the patients had abnormal findings on paranasal sinus X-ray, such as mucosal thickening, haziness, or air-fluid level, they were also diagnosed as UACS (19). Based on the treatment guidelines, we recommend first-generation antihistamines, but second-generation antihistamines are also allowed depending on the patient’s concurrent conditions. Asthma or cough variant asthma was diagnosed when bronchial hyperresponsiveness was confirmed by methacholine or mannitol provocation tests. This was also confirmed by a positive bronchodilator response (20). EB was diagnosed after exclusion of the other possible causes of CC and confirmed by an increased sputum eosinophil count of more than 3% in induced sputum tests (21). For treatment of asthma/EB, inhaled corticosteroids ± long-acting β-2 agonists were prescribed. GERD was suspected if the patient had typical acid regurgitation or heartburn symptoms. Patients with endoscopic findings of GERD were also included (22). GERD was treated by proton pump inhibitor. Multiple diagnoses were allowed, and specific treatments were performed according to each diagnosis.
Cough measurement tools (COAT and cough NRS) and definition of early responder by minimal important difference (MID)
COAT is a unique cough evaluation tool that was developed by the Cough Research Group of KATRD. It is well correlated with cough NRS and the Korean version of the Leicester Cough Questionnaire (K-LCQ) (23). The total score of COAT ranges from 0 to 20, which is composed of five factors ranging from 0 to 4: cough frequency, daily activity limitation, sleep disturbance due to cough, fatigue, and cough hypersensitivity (13). The MID of COAT were more than 2.0 points (13).
Cough NRS is an alternative expression of the cough visual analog scale (VAS), which is a traditional rating scale that represents the severity, intensity, or frequency of cough. It ranges from 0 (no cough at all) to 10 (maximal cough) (1). The MID of cough NRS is not specified until recently. Instead, we adopted the MID of VAS according to the previous articles (≥3 points) (24,25). In this study, both COAT and cough NRS were measured together three times: at baseline (before treatment), 2 weeks after treatment, and 4 weeks after treatment. The rate of improvement was calculated as changes in points per week (ΔCOAT and ΔNRS).
Early responder is defined by the patients who showed the improvement both COAT and cough NRS within 2 weeks of treatment, and who keep the response until 4 weeks of treatment. The improvement was defined by the decrease of score more than MID. The others were classified as non-early responder. We analyzed the contributing factors for each cough NRS and COAT in cases that meet the criteria for early responders through subgroup analysis.
Statistical analysis
For continuous variables, normality was tested using the Shapiro-Wilk test. The equality of variance was evaluated using Bartlett’s test. Kruskal-Wallis tests were performed to compare the means between the three groups. Pearson’s chi-square test or Fisher’s exact test was used for categorical variables. To compare the changes along with treatment, repeated measures analysis of variance (RM ANOVA) was used to compare the results from cough NRS and COAT by Bonferroni correction for post-hoc analysis. Binomial univariate and multivariate logistic regression analyses were performed to evaluate the variables in estimation of early responder. A P value of <0.05 was considered to indicate statistical significance. All statistical analyses were performed using RStudio Team [2020] RStudio: Integrated Development for R. RStudio, PBC, Boston, MA, USA (http://www.rstudio.com/).
Results
Demographics of early responder in treatment of CC
Among 633 CC patients who undertook cough questionnaire voluntarily, 304 patients were enrolled after met inclusion and exclusion criteria of the study (Figure 1). Table 1 described the demographics of early responder and non-early responder group. The early responder group, comprising 56.6% (n=172), did not exhibit any statistically significant differences from the non-early responder group in terms of age, sex, and cough duration. However, there was a statistically significant difference in final diagnoses of CC. The proportion of UACS was significantly higher in early responder group than in non-early responder group (65.7% vs. 49.2%, P=0.006). The proportions of asthma/EB and GERD between early responder group and non-early responder group were not statistically different. Baseline cough NRS and COAT were higher in early responder group than in non-early responder group (5.5±1.2 vs. 4.9±1.3, P<0.001; 9.2±2.9 vs. 8.1±3.1, P=0.002). Early responder group showed lower cough NRS and COAT in 2nd- and in 4th-weeks than non-early responder group (both P<0.01) (Table 1). We performed subgroup analyses of each cough NRS and COAT. In cough NRS subgroup analysis, early responder group showed higher proportion of UACS diagnosis than counterparts (P=0.006), and there was no statistically significant difference in other variables. In COAT subgroup analysis, early responder group showed higher proportion of more than sixties than counterparts, while other variables were not statistically different between the two groups (Table S1).
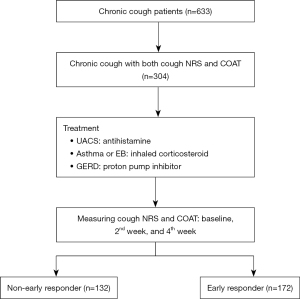
Table 1
| Treatment response | Study subject (n=304) | Non-early (n=132) | Early (n=172) | P value |
|---|---|---|---|---|
| Age (years) | 49.5±15.1 | 49.3±14.8 | 49.7±15.4 | 0.82 |
| <40 | 88 (28.9) | 36 (27.3) | 52 (30.2) | 0.16 |
| 40–59 | 134 (44.1) | 66 (50.0) | 68 (39.5) | |
| 60 or more | 82 (27.0) | 30 (22.7) | 52 (30.2) | |
| Female sex | 216 (71.1) | 96 (72.7) | 120 (69.8) | 0.66 |
| Cough duration (weeks) | 0.39 | |||
| 8–12 | 147 (48.4) | 59 (44.7) | 88 (51.2) | |
| 13–24 | 53 (17.4) | 26 (19.7) | 27 (15.7) | |
| 25–52 | 55 (18.1) | 28 (21.2) | 27 (15.7) | |
| >52 | 49 (16.1) | 19 (14.4) | 30 (17.4) | |
| Final diagnosis | ||||
| UACS | 178 (58.6) | 65 (49.2) | 113 (65.7) | 0.006 |
| Asthma/EB | 212 (69.7) | 95 (72.0) | 117 (67.9) | 0.54 |
| GERD | 43 (14.1) | 23 (17.4) | 20 (11.6) | 0.20 |
| Cough NRS (points) | ||||
| 0 week | 5.2±1.3 | 4.9±1.3 | 5.5±1.2 | <0.001 |
| 2nd week | 2.4±1.6 | 3.3±1.6 | 1.6±1.2 | <0.001 |
| 4th week | 1.3±1.4 | 1.9±1.6 | 0.8±0.8 | <0.001 |
| ΔNRS (points/week) | −1.0±0.4 | −0.7±0.4 | −1.2±0.3 | <0.001 |
| COAT (points) | ||||
| 0 week | 8.8±3.0 | 8.1±3.1 | 9.2±2.9 | 0.002 |
| 2nd week | 3.8±3.0 | 5.1±3.2 | 2.8±2.3 | <0.001 |
| 4th week | 2.0±2.2 | 2.8±2.7 | 1.4±1.5 | <0.001 |
| ΔCOAT (points/week) | −1.7±0.7 | −1.3±0.7 | −1.9±0.7 | <0.001 |
Data are presented as mean ± SD or n (%). Early responder showed higher percentage of UACS and baseline cough NRS and COAT score compared to counterpart. Δ, delta; COAT, Cough Assessment Test; EB, eosinophilic bronchitis; GERD, gastroesophageal reflux disease; NRS, numeric rating scale; SD, standard deviation; UACS, upper airway cough syndrome.
Changes of cough NRS and COAT according to the early response of treatment
Figure 2 showed that the outlines for being early responder. The proportion of early responders who met the MID for cough severity was approximately 57%, with the highest proportion observed in UACS (64.6%) followed by asthma/EB (55.7%) and GERD (46.5%). In contrast, the proportion of patients who met the criteria for cough-associated QOL was high at approximately 90%, with the highest proportion observed in UACS (91.6%), followed by asthma/EB (90.6%) and GERD (81.4%). These findings suggest that early response to treatment may differ depending on the underlying cause of CC, while improvement in cough-related QOL is more consistent across the three conditions. In the subgroup analyses of cough NRS, baseline scores were significantly higher in early responder group than in non-early responder group (5.5±1.2 vs. 4.9±1.3, P<0.001). Those of COAT were also higher in early responder group than in non-early responder group (8.9±2.9 vs. 7.5±3.4, P<0.001). After 2 weeks of treatment, early responder group showed lower scores than non-early responder group in both cough NRS (1.7±1.2 vs. 3.3±1.6, P<0.001) and in COAT (3.4±2.6 vs. 7.2±4.0, P<0.001). After four weeks of treatment, early responder group showed lower scores than non-early responder group in both cough NRS (0.9±0.9 vs. 1.9±1.6, P<0.001) and in COAT (1.9±2.1 vs. 3.3±2.6, P=0.001). The rate of improvement per weeks were different both in cough NRS (early responder group vs. non-early responder group; −1.2±0.3 vs. −0.7±0.4, P<0.001) and in COAT (early responder group vs. non-early responder group; −1.8±0.7 vs. −1.1±0.9, P<0.001) between early responder group and non-early responder group (Figure 3 and Table S1). We evaluated the RM ANOVA to evaluate the intra- and inter-group correlation effect on changes in COAT and cough NRS scores. Both COAT and cough NRS scores improved over time (P<0.001, both) and these were also shown both in cough NRS and COAT by whether early responder group or not (P<0.001, both), which showed that there was difference in changes of NRS and COAT according to the early responder group or not. In the post-hoc analyses by Bonferroni method, difference was showed between baseline (0-week) and 2nd-week, between 2nd- and 4th-week, and between 0- and 4th-week (P<0.001) (Figure 4).



Univariable and multivariable logistic regression analyses for being early responder
For analyzing the odds of being early responder, we selected the variables for the logistic regression analyses, such as female sex, age, cough duration, baseline cough NRS, baseline COAT, diagnosis of UACS, diagnosis of asthma/EB, and diagnosis of GERD. In the univariable analyses of early responder, higher baseline cough NRS [odds ratio (OR) 1.44; 95% confidence interval (CI): 1.19–1.74; P<0.001], higher baseline COAT (OR 1.13; 95% CI: 1.05–1.23; P=0.002), and diagnosis of UACS (OR 2.07; 95% CI: 1.30–3.30; P=0.004) were associated with being early responder group. After adjusting the confounding variables, higher cough NRS (OR 1.44; 95% CI: 1.06–1.97; P=0.021) and diagnosis of UACS (OR 1.76; 95% CI: 1.02–3.02; P=0.042) were still associated with being early responder group. In the subgroup analyses of cough NRS, baseline cough NRS (OR 1.48; 95% CI: 1.21–1.80; P<0.001), baseline COAT (OR 1.13; 95% CI: 1.04–1.22; P=0.004), and diagnosis of UACS (OR 2.07; 95% CI: 1.30–3.30; P=0.002) showed association with being early responder in the univariable logistic regression analyses. After adjusting confounding factors, cough NRS (OR 1.62; 95% CI: 1.17–2.23; P=0.003) and diagnosis of UACS (OR 1.86; 95% CI: 1.08–3.22; P=0.025) were still associated with being early responder. In the subgroup analyses of COAT, baseline COAT was only showed the association with early responder (OR 1.20; 95% CI: 1.03–1.40; P=0.018) in univariable logistic regression analyses. In the multivariable logistic regression analyses, lower cough NRS (OR 0.58; 95% CI: 0.35–0.98; P=0.042) and higher COAT (OR 1.42; 95% CI: 1.10–1.83; P=0.006) were still associated with being early responder (Table 2).
Table 2
| Variables | Both cough NRS and COAT | Cough NRS | COAT | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Univariate | Multivariate | Univariate | Multivariate | Univariate | Multivariate | ||||||||||||
| OR (95% CI) | P value | OR (95% CI) | P value | OR (95% CI) | P value | OR (95% CI) | P value | OR (95% CI) | P value | OR (95% CI) | P value | ||||||
| Gender | |||||||||||||||||
| Male | Reference | Reference | Reference | ||||||||||||||
| Female | 0.84 (0.51–1.39) |
0.50 | 0.91 (0.53–1.56) |
0.73 | 0.84 (0.51–1.39) |
0.50 | 0.87 (0.51–1.51) |
0.63 | 0.44 (0.16–1.19) |
0.10 | 0.45 (0.16–1.28) |
0.14 | |||||
| Age (years) | |||||||||||||||||
| <40 | Reference | Reference | Reference | ||||||||||||||
| 40–59 | 0.76 (0.44–1.30) |
0.32 | 0.78 (0.43–1.41) |
0.41 | 0.76 (0.44–1.30) |
0.32 | 0.84 (0.46–1.52) |
0.55 | 0.47 (0.19–1.15) |
0.10 | 0.56 (0.21–1.47) |
0.24 | |||||
| ≥60 | 1.20 (0.65–2.23) |
0.56 | 1.23 (0.62–2.42) |
0.55 | 1.20 (0.65–2.23) |
0.56 | 1.23 (0.62–2.44) |
0.55 | 2.28 (0.57–9.11) |
0.25 | 2.59 (0.61–10.95) |
0.20 | |||||
| Cough duration (weeks) | |||||||||||||||||
| 8–12 | Reference | Reference | Reference | ||||||||||||||
| 13–24 | 0.66 (0.35–1.24) |
0.19 | 0.70 (0.36–1.36) |
0.29 | 0.66 (0.35–1.24) |
0.19 | 0.65 (0.33–1.27) |
0.21 | 1.82 (0.59–5.61) |
0.30 | 2.17 (0.64–7.36) |
0.22 | |||||
| 25–52 | 0.61 (0.33–1.14) |
0.12 | 0.68 (0.35–1.32) |
0.25 | 0.61 (0.33–1.14) |
0.12 | 0.64 (0.33–1.23) |
0.18 | 1.48 (0.53–4.19) |
0.46 | 1.98 (0.64–6.11) |
0.24 | |||||
| >52 | 1.00 (0.52–1.94) |
1.00 | 1.07 (0.53–2.17) |
0.84 | 1.00 (0.52–1.94) |
1.00 | 0.97 (0.48–1.98) |
0.94 | 2.28 (0.64–8.05) |
0.20 | 2.52 (0.67–9.49) |
0.17 | |||||
| Cough NRS 0-week | 1.44 (1.19–1.74) |
<0.001 | 1.44 (1.06–1.97) |
0.02 | 1.48 (1.21–1.80) |
<0.001 | 1.62 (1.17–2.23) |
0.003 | 1.09 (0.81–1.47) |
0.57 | 0.58 (0.35–0.98) |
0.04 | |||||
| COAT 0-week | 1.13 (1.05–1.23) |
0.002 | 1.00 (0.87–1.14) |
0.98 | 1.13 (1.04–1.22) |
0.004 | 0.95 (0.83–1.09) |
0.50 | 1.20 (1.03–1.40) |
0.02 | 1.42 (1.10–1.83) |
0.007 | |||||
| UACS | 2.07 (1.30–3.30) |
0.004 | 1.76 (1.02–3.02) |
0.04 | 2.07 (1.30–3.30) |
0.002 | 1.86 (1.08–3.22) |
0.03 | 1.58 (0.75–3.33) |
0.23 | 1.49 (0.58–3.83) |
0.40 | |||||
| Asthma/EB | 0.81 (0.49–1.33) |
0.40 | 0.95 (0.51–1.74) |
0.86 | 0.81 (0.49–1.33) |
0.40 | 0.92 (0.50–1.71) |
0.80 | 1.30 (0.60–2.84) |
0.50 | 1.47 (0.53–4.11) |
0.46 | |||||
| GERD | 0.60 (0.32–1.16) |
0.13 | 0.68 (0.34–1.36) |
0.27 | 0.60 (0.32–1.16) |
0.13 | 0.64 (0.32–1.29) |
0.21 | 0.42 (0.18–1.02) |
0.06 | 0.49 (0.18–1.35) |
0.17 | |||||
Logistic regression analyses showed that higher baseline cough NRS and diagnosis of UACS were associated with being early responder to treatment in chronic cough after adjusting sex, age, cough duration, COAT, and confounding diagnosis of chronic cough. CI, confidence interval; COAT, Cough Assessment Test; EB, eosinophilic bronchitis; GERD, gastroesophageal reflux disease; NRS, numeric rating scale; OR, odds ratio; UACS, upper airway cough syndrome.
Discussion
A cough is a normal reflex that removes the stimulants of the airway (5). However, CC is detrimental to the QOL of patients (26). In a real-world survey, current care for CC is somewhat below the expected standard, such as unnecessarily longer exposure to medication (27). We hypothesized that there were clinical characteristics of early responder in CC patients.
We showed some interesting results in this study. First, we described the clinical characteristics of the treatment response of CC. Higher baseline cough NRS and diagnosis of UACS were associated with early response to treatment. These results showed that the possibility of short-term treatment in selective cases, such as who had UACS alone or had higher cough severity compared to others. Also, the 2 weeks of short-term application of inhaled corticosteroid in asthma/EB did not show clinical improvement unlike previous recommendation (11). Second, we demonstrated that baseline cough severity (cough NRS) was associated with early responder unlikely to that of cough QOL (COAT). Also, higher baseline cough NRS was associated with not meeting the MID of QOL. These findings highlight the importance of controlling cough severity to improve the clinical outcomes earlier. The classical way of estimating cough severity, cough NRS or VAS, may be the key for improving early outcomes of CC treatment. Third, these results suggested that the rates of improvement were different in severity and QOL. In this study, most of patient showed improvement of QOL after two weeks of treatment, while those of cough severity did not show the improvement. Therefore, this may be associated with the delayed improvement for CC treatment. We must explain the predicted course of treatment to ensure that the patients were comfortable even if they did not have feeling of improvement in severity of coughing, especially considering that the complaints of most patients have an unclear prognosis (27). Fourth, we found the importance of a comprehensive evaluation of cough through this study. As previously demonstrated, the cough severity and QOL influence each other and have different rates of treatment response. However, previous researches usually focused on single cough-measuring index to define improvement of cough or focused on etiology (25,28). Therefore, a composite scoring system should be considered in the upcoming study.
This study has some limitations. First, we only included the diagnoses of UACS, asthma/EB, and GERD. Other causes, such as idiopathic cough, somatic cough, or cough hypersensitivity, were excluded in this study. However, we could evaluate the factors which were associated with early responder by excluding those groups which were highly associated with uncontrolled CC in previous studies (29,30). Second, in this study, the concepts of refractory CC or unexplained CC were not applied. However, they were included in non-early responder group by its definition, and its proportion was not high because the overall patient group showed nearly 90% of treatment response rate at 4 weeks of treatment (1). Therefore, these factors have little influence on identifying the characteristics of the early responder group. Third, this was not a prospective study. However, the prospective cohort study may provoke selection bias because of the nature of the study (31). All participating hospitals regularly evaluated and managed CC patients according to the Korean cough guidelines, which reduced the weakness of the retrospective study design we used. Fourth, medication was not considered as factors in this study. However, all patients were treated according to the Korean cough guidelines that recommend antihistamines for UACS, inhaled corticosteroids for asthma/EB, and proton pump inhibitors for GERD. Previous study about this retrospective cohort showed that the diagnosis was matched to its medications close to 100% (1). Therefore, the factors related to medication were included in the diagnosis, and we supplemented them by utilizing the diagnosis as an independent variable in this study. Fifth, exclusion by incomplete dataset should be associated with selection bias. This occurred because we only selected participants who performed both COAT and cough NRS. However, most studies cannot avoid selection bias due to the study setting, and since we only wanted to analyze the characteristics of early responder compared to non-responder groups, we deemed these results useful for interpretation despite the possibility of bias.
Conclusions
Conclusively, we described the presence of early responder of CC and its characteristics. The early responder group had higher baseline cough severity and diagnosed as UACS group. Furthermore, we carefully recommend that physicians should consider the short-term duration of treatment in UACS group. They should explain to their patients that a slower improvement of cough severity is expected, also.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://jtd.amegroups.com/article/view/10.21037/jtd-22-1374/rc
Data Sharing Statement: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-22-1374/dss
Peer Review File: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-22-1374/prf
Funding: This study was supported by grant from
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jtd.amegroups.com/article/view/10.21037/jtd-22-1374/coif). C.K.R. serves as an unpaid editorial board member of Journal of Thoracic Disease. C.K.R. reports that he received consulting/lecture fees from AstraZeneca, GSK, Novartis, Mundipharma, Boehringer-Ingelheim, Teva, and Sanofi. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was approved by the Institutional Review Board of the Catholic University of Korea, South Korea (No. XC19REDI0027) and individual consent for this retrospective analysis was waived.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- An TJ, Kim JW, Choi EY, et al. Clinical Characteristics of Chronic Cough in Korea. Tuberc Respir Dis (Seoul) 2020;83:31-41. [Crossref] [PubMed]
- Rhee CK, Jung JY, Lee SW, et al. The Korean Cough Guideline: Recommendation and Summary Statement. Tuberc Respir Dis (Seoul) 2016;79:14-21. [Crossref] [PubMed]
- Johansson H, Johannessen A, Holm M, et al. Prevalence, progression and impact of chronic cough on employment in Northern Europe. Eur Respir J 2021;57:2003344. [Crossref] [PubMed]
- Song WJ, Chang YS, Faruqi S, et al. The global epidemiology of chronic cough in adults: a systematic review and meta-analysis. Eur Respir J 2015;45:1479-81. [Crossref] [PubMed]
- Chung KF, Pavord ID. Prevalence, pathogenesis, and causes of chronic cough. Lancet 2008;371:1364-74. [Crossref] [PubMed]
- Morice AH, Fontana GA, Sovijarvi AR, et al. The diagnosis and management of chronic cough. Eur Respir J 2004;24:481-92. [Crossref] [PubMed]
- Gibson PG, Vertigan AE. Management of chronic refractory cough. BMJ 2015;351:h5590. [Crossref] [PubMed]
- Kang SY, Won HK, Lee SM, et al. Impact of Cough and Unmet Needs in Chronic Cough: A Survey of Patients in Korea. Lung 2019;197:635-9. [Crossref] [PubMed]
- Joo H, Moon JY, An TJ, et al. Revised Korean Cough Guidelines, 2020: Recommendations and Summary Statements. Tuberc Respir Dis (Seoul) 2021;84:263-73. [Crossref] [PubMed]
- Irwin RS, French CL, Chang AB, et al. Classification of Cough as a Symptom in Adults and Management Algorithms: CHEST Guideline and Expert Panel Report. Chest 2018;153:196-209. [Crossref] [PubMed]
- Morice AH, Millqvist E, Bieksiene K, et al. ERS guidelines on the diagnosis and treatment of chronic cough in adults and children. Eur Respir J 2020;55:1901136. [Crossref] [PubMed]
- Lee KK, Davenport PW, Smith JA, et al. Global Physiology and Pathophysiology of Cough: Part 1: Cough Phenomenology - CHEST Guideline and Expert Panel Report. Chest 2021;159:282-93. [Crossref] [PubMed]
- Koo HK, Jeong I, Kim JH, et al. Development and validation of the COugh Assessment Test (COAT). Respirology 2019;24:551-7. [Crossref] [PubMed]
- Koo HK, Bae W, Moon JY, et al. Differential features of chronic cough according to etiology and the simple decision tree for predicting causes. Sci Rep 2021;11:10326. [Crossref] [PubMed]
- Satia I, Wahab M, Kum E, et al. Chronic cough: Investigations, management, current and future treatments. Can J Respir Crit Care Sleep Med 2021;5:404-16.
- Kum E, Guyatt GH, Devji T, et al. Cough symptom severity in patients with refractory or unexplained chronic cough: a systematic survey and conceptual framework. Eur Respir Rev 2021;30:210104. [Crossref] [PubMed]
- Zhan W, Zhang L, Jiang M, et al. A new simple score of chronic cough: cough evaluation test. BMC Pulm Med 2020;20:68. [Crossref] [PubMed]
- Spinou A, Birring SS. An update on measurement and monitoring of cough: what are the important study endpoints? J Thorac Dis 2014;6:S728-34. [Crossref] [PubMed]
- Pratter MR. Chronic upper airway cough syndrome secondary to rhinosinus diseases (previously referred to as postnasal drip syndrome): ACCP evidence-based clinical practice guidelines. Chest 2006;129:63S-71S. [Crossref] [PubMed]
- 2021 GINA Report, Global Strategy for Asthma Management and Prevention, 2021. 2021. Available online: www.ginasthma.org
- Belda J, Leigh R, Parameswaran K, et al. Induced sputum cell counts in healthy adults. Am J Respir Crit Care Med 2000;161:475-8. [Crossref] [PubMed]
- Jung HK, Hong SJ, Jo YJ, et al. Updated guidelines 2012 for gastroesophageal reflux disease. Korean J Gastroenterol 2012;60:195-218. [Crossref] [PubMed]
- Birring SS, Prudon B, Carr AJ, et al. Development of a symptom specific health status measure for patients with chronic cough: Leicester Cough Questionnaire (LCQ). Thorax 2003;58:339-43. [Crossref] [PubMed]
- Martin Nguyen A, Bacci ED, Vernon M, et al. Validation of a visual analog scale for assessing cough severity in patients with chronic cough. Ther Adv Respir Dis 2021;15:17534666211049743. [Crossref] [PubMed]
- Zhang M, Sykes DL, Brindle K, et al. Chronic cough-the limitation and advances in assessment techniques. J Thorac Dis 2022;14:5097-119. [Crossref] [PubMed]
- Zeiger RS, Schatz M, Butler RK, et al. Burden of Specialist-Diagnosed Chronic Cough in Adults. J Allergy Clin Immunol Pract 2020;8:1645-1657.e7. [Crossref] [PubMed]
- Kang SY, Won HK, Lee SM, et al. Impact of Cough and Unmet Needs in Chronic Cough: A Survey of Patients in Korea. Lung 2019;197:635-9. [Crossref] [PubMed]
- Kang J, Seo WJ, Kang J, et al. Clinical phenotypes of chronic cough categorised by cluster analysis. PLoS One 2023;18:e0283352. [Crossref] [PubMed]
- McGarvey LP. Idiopathic chronic cough: a real disease or a failure of diagnosis? Cough 2005;1:9. [Crossref] [PubMed]
- Chung KF, McGarvey L, Song WJ, et al. Cough hypersensitivity and chronic cough. Nat Rev Dis Primers 2022;8:45. [Crossref] [PubMed]
- Biele G, Gustavson K, Czajkowski NO, et al. Bias from self selection and loss to follow-up in prospective cohort studies. Eur J Epidemiol 2019;34:927-38. [Crossref] [PubMed]


