
The synergistic effects of PM2.5 and high-fat diet on Th1/Th2 balance in model mice with asthma
Highlight box
Key findings
• Particulate matter, ambient particulate matter with an aerodynamic equivalent diameter ≤2.5 µm (PM2.5) exposure and a high-fat diet modulate the Th1/Th2 balance in opposite directions, yet both exacerbate asthma. PM2.5 promotes asthma development through a Th2 response, whereas a high-fat diet does so through a Th1 response. The combination of PM2.5 exposure and a high-fat diet results in an attenuated shift towards Th2.
What is known and what is new?
• It is known that PM2.5 exposure and high-fat diets are risk factors for asthma, and studies have shown their individual effects on the immune system and asthma symptoms. The Th1/Th2 balance is crucial in the development and progression of asthma, with a skewed balance potentially contributing to asthma symptoms.
• This manuscript adds new insights into the interactions between PM2.5 exposure and high-fat diets on the Th1/Th2 balance in the context of asthma. The study demonstrates that while PM2.5 exposure favors a Th2 skew and high-fat diets favor a Th1 skew individually, the combined effects may lead to a moderation of Th2 polarization induced by PM2.5 exposure.
What is the implication, and what should change now?
• The findings suggest a complex interplay between PM2.5 exposure, high-fat diets, and the immune response in asthma development. Public health policies could focus on promoting healthier dietary patterns, especially in regions with high levels of air pollution, to potentially reduce asthma risk and severity.
Introduction
Asthma is a significant chronic, non-communicable disease (1). Currently, about 334 million people globally are affected by asthma, contributing to a substantial disease burden worldwide (2). Several risk factors are associated with the prevalence of asthma, including heredity, exposure to tobacco smoke, viral exposure, air pollution, obesity, genetic risk factors, socioeconomic status, and occupational exposures (3).
A nationwide analysis in 272 Chinese cities suggested a robust association between short-term PM2.5 exposure and increased mortality from several respiratory diseases (4). By activating the nuclear factor kappa-B (NF-κB) and mitogen-activated protein kinase (MAPK) pathways, PM2.5 exposure caused oxidative stress and inflammatory responses that then exacerbated allergic asthma (5). Obesity was initially thought to be a natural risk factor for asthma. In many cases of asthma, the pathogenesis of asthma is due to the activation of Th2 cells, the increased secretion of related inflammatory factors such as interleukin (IL)-4, IL-5, and IL-13 (6). In addition to “type 2-high” asthmatics, more research has shown that high-fat diet-induced obesity has a “type 2-low” signature, the presence of neutrophils, and unresponsiveness to corticosteroids (7). In conclusion, both PM2.5 exposure and a high-fat diet are risk factors for the development and exacerbation of asthma.
There are numerous biological regulatory processes involved in the onset of asthma. Among these, the Th1/Th2 imbalance is an essential immunological mechanism for inducing and exacerbating asthma. Th1 and Th2 are two main T cells, both originating from Th0 cells. Th0 cells also include other subpopulations such as Th17 (8). With the aid of T-bet, Th0 cells differentiate into Th1 cells, which regulate the secretion of cytokines such as IL-2, interferon-gamma (IFN-γ), and tumor necrosis factor-α (TNF-α) (9). IFN-γ and TNF-α are pleiotropic cytokines controls Th1/Th2 balance. Th0 cells can also differentiate into Th2 cells under the influence of GATA-3 (10). Th2 cells secrete cytokines including IL-4, IL-5, IL-6, IL-10, and IL-13 (10). The dynamic balance maintained by the reciprocal suppression of these cytokines is crucial. Previous studies have reported disruptions in this balance in patients with immunologic asthma (11,12). It has been observed that the onset and progression of asthma are related to a Th2 response, with Th1 cells thought to protect against allergic asthma by inhibiting the Th2 response (13,14); however, a further study indicate that a Th1 response might enhance allergy and airway hyperresponsiveness in allergic asthma (15). Thus, the over-differentiation of both Th1 and Th2 cells can induce and aggravate asthma.
Both PM2.5 exposure and high-fat diets are risk factors for the development and exacerbation of asthma. PM2.5 increases the secretion of Th2-related cytokines (IL-4, IL-5, and IL-13) (16). A high-fat diet leads to an over-secretion of Th2-related cytokines (TNF-α) (17,18). We hypothesize that the Th1/Th2 imbalance plays a role in the onset of asthma induced by PM2.5 exposure and high-fat diets. In Beijing and other regions of China, polluted air with high concentrations of PM2.5 remains a top concern among the six conventional air pollutants. Meanwhile, with economic development, the prevalence of high-fat diets is also significant in these cities. Therefore, in many cities in China, PM2.5 exposure and high-fat diets coexist. Based on this evidence, investigating the synergistic effects of PM2.5 exposure and high-fat diets on the onset and progression of asthma is meaningful. We present this article in accordance with the ARRIVE reporting checklist (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1139/rc).
Methods
Animals
Sixty-four male BALB/c wild-type mice, aged 4 weeks, were obtained from Vital River Laboratory Animal Technology Co., Ltd. (Beijing, China). All mice were housed under standard conditions, including a 12-hour light/dark cycle, with free access to rodent chow and distilled water. After a 7-day acclimatization period, the mice were prepared for subsequent experiments. It has been shown that male and female mice respond differently to ovalbumin (OVA) sensitization, with female mice increasing experimental uncertainty due to estrogen, so we used male mice in the experiments.
Initially, the 64 mice were randomly divided into two main groups: a no-asthma group (n=32) and an OVA-induced asthma group (n=32). Each main group was further subdivided into four subgroups: control (Tables S1,S2), PM2.5 exposure (Figure S1), high-fat diet (Tables S3-S5), and combined PM2.5 and high-fat diet. This resulted in eight subgroups in total: wild type (C), PM2.5 exposure group (CP), high-fat diet group (CH), combined PM2.5 and high-fat diet group (CPH), asthma group (A), asthma with PM2.5 exposure group (AP), asthma with high-fat diet group (AH), and asthma with both PM2.5 and high-fat diet group (APH). All treatments, including PM2.5 exposure and high-fat diet, were administered for 30 days. The high-fat diets contained 60% fat, the table of ingredients for normal and high fat diets are in supplementary tables, the nutrient of both is essentially the same, fat is the only variable.
This study was approved by the Scientific Research Ethics Committee of the Chinese Research Academy of Environmental Sciences (project reference No. 005-o: 00e), and adhered to the Committee for Control and Supervision of Experiments on Animals’ regulations. A protocol was prepared before the study without registration.
Experimental OVA-driven allergic asthma model
For the experimental OVA-driven allergic asthma model, mice were immunized intraperitoneally (i.p.) with 20 µg OVA and 2 mg Alum on days 1 and 7 for sensitization. Then we performed provocation, mice were given 5% OVA in 50 µL phosphate buffered saline (PBS), is a buffer solution commonly used in biological and medical research by nebulized inhalation for 30 minutes every 2 days from day 14 to day 28 in the asthma groups. And the normal group was given saline by nebulized inhalation. On day 30, airway hyperresponsiveness (AHR) was assessed, and tissue samples were collected for further analysis.
PM2.5 exposure experimental design
There are two sets of animal exposure devices (Figure S1), divided into particulate matter exposure chambers and particulate matter purification chambers (1 units each) (the animal exposure devices have been granted a patent for the invention, Patent No. ZL201811275872.3). The unfiltered chamber is fed directly into the contaminated outdoor air, while the air fed into the filtered chamber is filtered by high efficiency particulate air (HEPA_ membranes (a mat of randomly aligned fibers, made from either glass or synthetic materials). PM2.5 concentrations were 12.6±3.8 µg/m3 at the filtered chamber in the control group and 139.2±32.3 µg/m3 at the unfiltered chamber in the PM2.5 exposure group. The temperature of chambers was controlled at 24±1 ℃.
Animal tissue preparation
After 30 days of administration, the mice euthanized with CO2 inhalation from the dry ice. Once euthanasia was confirmed, they were positioned on a clean laboratory table for sample collection. We harvested the lung and bronchoalveolar lavage fluid (BALF). The collected lung tissue was processed in two ways: (I) washed with PBS, then preserved in formalin for histological sectioning; (II) snap-frozen in liquid nitrogen and stored at −80 ℃.
Lung histopathology sections with hematoxylin and eosin (H&E) staining
Briefly, 5 µm slices of paraffin-embedded lung tissue were prepared and mounted on poly-L-lysine coated slides. The tissues were stained with H&E and examined under a Nikon 80i Eclipse microscope (Nikon Corporation, Yokohama, Japan).
Enzyme-linked immunosorbent assay
Cytokines including IFN-γ, IL-2, IL-4, IL-5, IL-10, IL-17, and proteins such as GATA-3, and T-bet (all from Beijing Qisong Biotechnology Co., Ltd., Beijing, China), were quantified using ELISA kits according to the manufacturer’s instructions.
RNA isolation and RNA sequencing (RNA-seq)
Total RNA was extracted from lung samples using Trizol reagent (Invitrogen, Inc., Carlsbad, USA). RNA-seq provided comprehensive sequence and expression profiles of nearly all transcripts from the specified cells or tissues. We conducted differential gene expression analysis (Tables S6,S7), Gene Ontology (GO) analysis (GO enrichment analysis, to perform enrichment analysis on gene sets), and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis using RNA-seq data.
RNA isolation and real-time polymerase chain reaction (RT-PCR)
From the isolated RNA, 2 µg was used to synthesize first-strand complementary DNA (cDNA) using a Primescript RT Reagent Kit (BGI Genomics Co., Ltd., Shenzhen, China) as per the manufacturer’s protocol. RT-PCR was performed using a SYBR Green Mix kit (Takara Bio, Otsu, Japan). The primer sequences are detailed in Table S8. Quantitative PCR (qPCR) was conducted on a 7300 Plus Real-time PCR system (Thermo Fisher Scientific CN, Shanghai, China). The relative expression levels were calculated using the 2−ΔΔCT method, with normalization to β-actin messenger RNA (mRNA) (Table S8).
Statistical analysis
Data were presented as the mean ± standard deviation (mean ± SD). Statistical significance was assessed using Student’s t-test, after verifying homogeneity of variances and normality of distribution. All statistical analyses were carried out using GraphPad Prism (La Jolla), and a P value less than 0.05 was considered statistically significant.
Results
The effect of PM2.5 exposure and high-fat diet on the morphology of the lung
In the no-asthma group, PM2.5 exposure induced inflammatory cell infiltration and thickened alveolar septa; the alveolar walls tended to consolidate (Figure 1A,1B). A high-fat diet caused similar pathological changes as PM2.5 exposure and also led to increased alveolar fusion (Figure 1A,1C). After combined exposure, the inflammatory response intensified, and there was notable alveolar destruction, telangiectasia of the alveolar wall, congestion, and a decrease in capillaries (Figure 1A-1D).

In the OVA-sensitized asthma group, the lungs showed typical asthma-related pathology such as inflammatory responses and consolidated alveolar walls (Figure 1E). PM2.5 exposure further increased alveolar wall destruction and fusion (Figure 1E,1F). A high-fat diet exacerbated alveolar wall destruction and fusion (Figure 1E,1G). After combined exposure, alveolar destruction and lung consolidation were more pronounced (Figure 1E-1H).
The effect of PM2.5 exposure and high-fat diet on eosinophil cationic protein (ECP) levels of lung
In the no-asthma group, compared with the wild type (C) group, ECP levels were higher after PM2.5 exposure (CP) and combined PM2.5 and high-fat diet (CPH) but lower in the high-fat diet group (CH). In the asthma group, compared with the asthma (A) group, ECP levels were higher in asthma and PM2.5 (AP) and asthma with both PM2.5 and a high-fat diet (APH), however, lower in the asthma with a high-fat diet group (AH) (Figure 2).

The effect of PM2.5 exposure and high-fat diet on Th1/Th2 balance
IFN-γ and IL-2 are secreted by Th1 cells. IL-4, IL-5, and IL-10 are secreted by Th2 cells. In the no-asthma group, PM2.5 exposure decreased IL-2 but increased IL-4, IL-5 and IL-10 levels indicating a shift in the Th1/Th2 balance toward Th2. The high-fat diet did not alter the levels of IL-2, IL-4, IL-5, and IL-10, showing no impact on the Th1/Th2 balance. In the combined group (CPH), IL-4, IL-10 and IFN-γ levels were significantly increased, while IL-2 and IL-5 showed no significant change (Figure 3). These results suggest that PM2.5 exposure and a high-fat diet influence the Th1/Th2 balance differently.
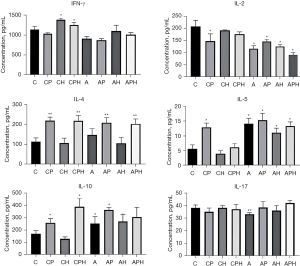
In the OVA-sensitized asthma group, after PM2.5 exposure (AP), IL-2, IL-4 and IL-10 were significantly up-regulated. After treatment with a high-fat diet (AH), IL-2 was significantly up-regulated, IL-5 was significantly down-regulated, while IFN-γ, IL-4, and IL-10 showed no significant changes. In the combined exposure group (APH), IL-2 and IL-5 was significantly down-regulated, IL-4 were significantly up-regulated, while levels of IFN-γ and IL-10 did not show significant changes (Figure 3). Similar as the no-asthma group, PM2.5 exposure and a high-fat diet had opposite effects on the Th1/Th2 balance.
The effect of PM2.5 exposure and high-fat diet on Th17
Th17 cells, which express IL-17, play a crucial role in inducing nonatopic asthma (19,20). But in our study, IL-17 expression did not significantly change with either PM2.5 or high-fat exposure. This indicates that the alteration in Th1/Th2 balance resulting from PM2.5 exposure and a high-fat diet is not significantly correlated with IL-17. This may be due to the involvement of other related genes or signaling pathways, which will be investigated in subsequent studies (Figure 3).
The effect of PM2.5 exposure and high-fat diet on Th1/Th2 balance GATA-3 and T-bet express level
Th1/Th2 balance were assessed by calculating the relative expression (Figure 4) and rate of change of genes (Tables S9,S10) in the normal and asthma groups separately. In the no-asthma group, PM2.5 exposure resulted in the up-regulation of GATA-3 and the down-regulation of T-bet. A high-fat diet did not affect GATA-3 or T-bet expression. In the combined exposure group (CPH), GATA-3 expression was significantly up-regulated (Figure 4, Tables S9,S10). In the OVA-sensitized asthma group, PM2.5 exposure led to up-regulation of GATA-3 and down-regulation of T-bet, whereas the high-fat diet had the opposite effect. In the combined exposure group (APH), GATA-3 expression down-regulated and T-bet expression up-regulated (Figure 4, Tables S9,S10). These results further confirmed the opposing effects of PM2.5 exposure and high-fat diet on the Th1/Th2 balance.

The effect of PM2.5 exposure and high-fat diet on other factors referring to Th1/Th2 balance
Furthermore, we analyzed the differentially expressed genes (DEGs) in each experimental group using RNA-seq (Figures S2-S4). From the DEGs, we identified changes in the KEGG pathways affected by various treatments. Notably, in both the no-asthma and asthma groups, the PI3K-AKT pathway was enriched by PM2.5 exposure and combined treatments (Figure S5). This pathway also had the highest number of gene changes across the three groups.
Numerous studies have demonstrated that the PI3K-AKT pathway is involved in asthma (21-23). Consequently, we hypothesize that the PI3K-AKT signaling pathway is also critical in regulating the onset and progression of asthma induced by PM2.5 exposure and the combined treatment with PM2.5 exposure and a high-fat diet. The PI3K-AKT signaling pathway influences the mTOR and NF-κB signaling pathways (24-26), which affect the expression levels of T-bet and GATA-3 (27,28), key regulators of the Th1/Th2 balance. Within the PI3K-AKT signaling pathway, Nr4a1 (NUR77) and PKN2 are the top two functionally related genes. NUR77 gene expression levels were significantly down-regulated in all three groups, whereas PKN gene levels were only significantly down-regulated in the group experiencing combined effects (Figure 5). This suggests that the combined effects of PM2.5 exposure and a high-fat diet could influence the Th1/Th2 balance by down-regulating the expression of NUR77 and PKN genes (Figure 6).


Discussion
In this study, we analyzed the effects of PM2.5 exposure and a high-fat diet on the Th1/Th2 balance and assessed their roles in the occurrence and development of asthma. In the no-asthma group, PM2.5 exposure shifted the Th1/Th2 balance toward Th2, whereas the high-fat diet shifted it toward Th1. After treatment with both a high-fat diet and PM2.5 exposure, the Th1/Th2 balance shifted toward Th2, with PM2.5 exposure being the dominant factor. In the asthma group, PM2.5 exposure led to a shift toward Th2, the high-fat diet shifted the balance toward Th1, and the combination of both had no significant effect on the Th1/Th2 balance.
Our results indicate that PM2.5 exposure leads to histopathological changes in the lung, accompanied by an increase in ECP, a marker strongly associated with asthma. This suggests that PM2.5 exposure may trigger the onset of asthma. Additionally, we observed a significant up-regulation of IL-4, IL-5, and IL-10 secreted by Th2 cells following PM2.5 exposure. Previous studies have reported that PM2.5 enhances airway hyperresponsiveness, elevates eosinophil counts, and promotes the secretion of cytokines such as IL-4, IL-5, and IL-13 by Th2 cells (7,29,30). Moreover, GATA-3, a critical factor in the differentiation of Th0 cells into Th2 cells, was found to be up-regulated after PM2.5 exposure in both this and prior studies (31). Conversely, T-bet, which facilitates the differentiation of Th0 cells into Th1 cells, was significantly reduced. These findings demonstrate that PM2.5 exposure shifts the Th1/Th2 balance towards Th2, thereby increasing the risk of Th2-type asthma.
Our study also shows that a high-fat diet induces histopathological changes in mouse lungs, upregulates the expression of IFN-γ and IL-2 secreted by Th1 cells, significantly decreases GATA-3 expression, and increases T-bet expression. This shift in the Th1/Th2 balance towards Th1 is evident. Some researchers have associated “type 2-low” asthma with obesity, characterized by neutrophil presence and corticosteroid unresponsiveness (7), and have noted that obesity-related asthma exhibits features of a T-helper 1 response (32). These results suggest that a high-fat diet shifts the Th1/Th2 balance towards Th1, increasing the risk of Th1-type asthma.
Under the combined influence of PM2.5 and a high-fat diet, we noted significant histopathological changes in the lungs. The level of ECP was elevated, suggesting an increased asthma risk. Additionally, IL-10 secretion by Th2 cells and GATA-3 expression were increased, while T-bet expression showed no significant change. These results indicate that the combined effects shift the Th1/Th2 balance towards Th2. However, compared to the PM2.5-only group, the combined treatment group exhibited a less pronounced shift towards Th2, likely due to the high-fat diet’s influence in shifting the balance towards Th1.
The activation of the PI3K/AKT signaling pathway, associated with asthma, has drawn our attention. NUR77 and PKN2 are pivotal genes in this pathway. NUR77, part of a nuclear receptor subfamily, is linked to the Th1/Th2 balance and stimulates inflammatory responses in various cell types (33,34). PKN2 has been identified as significantly upregulated in a population-based cohort study on childhood asthma (35), suggesting its role in the onset and development of asthma. Moreover, both NUR77 and PKN2 are crucial in the PI3K/AKT pathway. Our study found that the expression levels of these two genes were down-regulated following exposure to PM2.5, a high-fat diet, and their combined treatment. Therefore, we speculate that the PI3K/AKT pathway could be involved in regulating the Th1/Th2 imbalance induced by PM2.5 exposure, a high-fat diet, and their combination.
This study analyzed the mechanism of PM2.5 exposure, high-fat diet and combined effects on the occurrence and development of asthma in BALB/c mice, which is of great significance for the study of the mechanism of multi-factor combined effects on asthma, and is conducive to providing references for the study of the mechanism of human asthma. However, there have some limitations. The high-fat diet used in this study has a high fat content, and high-fat diets with different fat content and calories can be further studied to observe whether different results will appear. Second, the construction of asthma model is usually TH2 asthma induced by allergens, however, the mechanism of asthma is complex, including neutrophil asthma and asthma not induced by inflammatory cells, etc. Different asthma models can be constructed to further study the impact on the pathogenesis of different types of asthma.
Conclusions
In conclusion, PM2.5 exposure and short-term high-fat diet both exacerbate asthma, however, there has an opposite direction of modulation of the Th1/Th2 balance. PM2.5 promotes asthma development through a Th2 response, whereas short-term high-fat diet through a Th1 response. The combination of PM2.5 exposure and a high-fat diet results in an attenuated shift towards Th2.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the ARRIVE reporting checklist. Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1139/rc
Data Sharing Statement: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1139/dss
Peer Review File: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1139/prf
Funding: This work was supported by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-1139/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. This study was approved by the Scientific Research Ethics Committee of the Chinese Research Academy of Environmental Sciences (project reference No. 005-o: 00e), and adhered to the Committee for Control and Supervision of Experiments on Animals’ regulations.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Dharmage SC, Perret JL, Custovic A. Epidemiology of Asthma in Children and Adults. Front Pediatr 2019;7:246. [Crossref] [PubMed]
- Asthma--hope for the future? Lancet 2015;386:1014. [Crossref] [PubMed]
- Papi A, Brightling C, Pedersen SE, et al. Asthma. Lancet 2018;391:783-800. [Crossref] [PubMed]
- Chen R, Yin P, Meng X, et al. Fine Particulate Air Pollution and Daily Mortality. A Nationwide Analysis in 272 Chinese Cities. Am J Respir Crit Care Med 2017;196:73-81. [Crossref] [PubMed]
- Zhao C, Wang Y, Su Z, et al. Respiratory exposure to PM2.5 soluble extract disrupts mucosal barrier function and promotes the development of experimental asthma. Sci Total Environ 2020;730:139145. [Crossref] [PubMed]
- Rogozynski NP, Dixon B. The Th1/Th2 paradigm: A misrepresentation of helper T cell plasticity. Immunol Lett 2024;268:106870. [Crossref] [PubMed]
- Hammad H, Lambrecht BN. The basic immunology of asthma. Cell 2021;184:1469-85. [Crossref] [PubMed]
- Duddu AS, Andreas E, Bv H, et al. Multistability and predominant hybrid phenotypes in a four node mutually repressive network of Th1/Th2/Th17/Treg differentiation. NPJ Syst Biol Appl 2024;10:123. [Crossref] [PubMed]
- Zhang J, Le Gras S, Pouxvielh K, et al. Sequential actions of EOMES and T-BET promote stepwise maturation of natural killer cells. Nat Commun 2021;12:5446. [Crossref] [PubMed]
- Kanhere A, Hertweck A, Bhatia U, et al. T-bet and GATA3 orchestrate Th1 and Th2 differentiation through lineage-specific targeting of distal regulatory elements. Nat Commun 2012;3:1268. [Crossref] [PubMed]
- Zissler UM, Esser-von Bieren J, Jakwerth CA, et al. Current and future biomarkers in allergic asthma. Allergy 2016;71:475-94. [Crossref] [PubMed]
- León B, Ballesteros-Tato A. Modulating Th2 Cell Immunity for the Treatment of Asthma. Front Immunol 2021;12:637948. [Crossref] [PubMed]
- Ji T, Li H. T-helper cells and their cytokines in pathogenesis and treatment of asthma. Front Immunol 2023;14:1149203. [Crossref] [PubMed]
- Berker M, Frank LJ, Geßner AL, et al. Allergies - A T cells perspective in the era beyond the T(H)1/T(H)2 paradigm. Clin Immunol 2017;174:73-83. [Crossref] [PubMed]
- Bok SH, Seo JH, Bae CS, et al. Allium hookeri root extract regulates asthmatic changes through immunological modulation of Th1/Th2 related factors in an ovalbumin induced asthma mouse model. Mol Med Rep 2019;20:3215-23. [Crossref] [PubMed]
- Piao CH, Fan Y, Nguyen TV, et al. PM2.5 exposure regulates Th1/Th2/Th17 cytokine production through NF-κB signaling in combined allergic rhinitis and asthma syndrome. Int Immunopharmacol 2023;119:110254. [Crossref] [PubMed]
- Silva FMCE, Oliveira EE, Ambrósio MGE, et al. High-fat diet-induced obesity worsens TH2 immune response and immunopathologic characteristics in murine model of eosinophilic oesophagitis. Clin Exp Allergy 2020;50:244-55. [Crossref] [PubMed]
- Mohamed MME, Amrani Y. Obesity Enhances Non-Th2 Airway Inflammation in a Murine Model of Allergic Asthma. Int J Mol Sci 2024;25:6170. [Crossref] [PubMed]
- Kuramoto K, Morishima Y, Yoshida K, et al. Nrf2 Deficiency Accelerates IL-17-Dependent Neutrophilic Airway Inflammation in Asthmatic Mice. Antioxidants (Basel) 2024;13:818. [Crossref] [PubMed]
- Liu T, Woodruff PG, Zhou X. Advances in non-type 2 severe asthma: from molecular insights to novel treatment strategies. Eur Respir J 2024;64:2300826. [Crossref] [PubMed]
- Yuan J, Liu Y, Yu J, et al. Gene knockdown of CCR3 reduces eosinophilic inflammation and the Th2 immune response by inhibiting the PI3K/AKT pathway in allergic rhinitis mice. Sci Rep 2022;12:5411. [Crossref] [PubMed]
- Shao Y, Chong L, Lin P, et al. MicroRNA-133a alleviates airway remodeling in asthtama through PI3K/AKT/mTOR signaling pathway by targeting IGF1R. J Cell Physiol 2019;234:4068-80. [Crossref] [PubMed]
- Athari SS. Targeting cell signaling in allergic asthma. Signal Transduct Target Ther 2019;4:45. [Crossref] [PubMed]
- Vanhaesebroeck B, Stephens L, Hawkins P. PI3K signalling: the path to discovery and understanding. Nat Rev Mol Cell Biol 2012;13:195-203. [Crossref] [PubMed]
- Das J, Chen CH, Yang L, et al. A critical role for NF-kappa B in GATA3 expression and TH2 differentiation in allergic airway inflammation. Nat Immunol 2001;2:45-50. [Crossref] [PubMed]
- Wang S, Cheng Z, Cui Y, et al. PTPRH promotes the progression of non-small cell lung cancer via glycolysis mediated by the PI3K/AKT/mTOR signaling pathway. J Transl Med 2023;21:819. [Crossref] [PubMed]
- Zhang Y, Jing Y, Qiao J, et al. Activation of the mTOR signaling pathway is required for asthma onset. Sci Rep 2017;7:4532. [Crossref] [PubMed]
- Mowen KA, Glimcher LH. Signaling pathways in Th2 development. Immunol Rev 2004;202:203-22. [Crossref] [PubMed]
- Zahedi A, Hassanvand MS, Jaafarzadeh N, et al. Effect of ambient air PM(2.5)-bound heavy metals on blood metal(loid)s and children's asthma and allergy pro-inflammatory (IgE, IL-4 and IL-13) biomarkers. J Trace Elem Med Biol 2021;68:126826. [Crossref] [PubMed]
- Ogino K, Nagaoka K, Okuda T, et al. PM2.5-induced airway inflammation and hyperresponsiveness in NC/Nga mice. Environ Toxicol 2017;32:1047-54. [Crossref] [PubMed]
- Kanhere A, Hertweck A, Bhatia U, et al. T-bet and GATA3 orchestrate Th1 and Th2 differentiation through lineage-specific targeting of distal regulatory elements. Nat Commun 2012;3:1268. [Crossref] [PubMed]
- Nyambuya TM, Dludla PV, Mxinwa V, et al. Obesity-related asthma in children is characterized by T-helper 1 rather than T-helper 2 immune response: A meta-analysis. Ann Allergy Asthma Immunol 2020;125:425-432.e4. [Crossref] [PubMed]
- Banno A, Lakshmi SP, Reddy AT, et al. Key Functions and Therapeutic Prospects of Nur77 in Inflammation Related Lung Diseases. Am J Pathol 2019;189:482-91. [Crossref] [PubMed]
- Kurakula K, Vos M, Logiantara A, et al. Nuclear Receptor Nur77 Attenuates Airway Inflammation in Mice by Suppressing NF-κB Activity in Lung Epithelial Cells. J Immunol 2015;195:1388-98. [Crossref] [PubMed]
- Krautenbacher N, Flach N, Böck A, et al. A strategy for high-dimensional multivariable analysis classifies childhood asthma phenotypes from genetic, immunological, and environmental factors. Allergy 2019;74:1364-73. [Crossref] [PubMed]


