
Evaluation of the efficacy and safety of neoadjuvant immunotherapy in locally advanced esophageal squamous cell carcinoma
Highlight box
Key findings
• This study showed that camrelizumab-based neoadjuvant immuno-chemotherapy was effective in the treatment of locally advanced esophageal squamous cell carcinoma (ESCC). Moreover, tumor regression grade (TRG) was found to be significantly associated with imaging assessment; these two approaches can be combined and employed to predict the efficacy of neoadjuvant immuno-chemotherapy.
What is known, and what is new?
• A number of studies have shown that camrelizumab plus chemotherapy as neoadjuvant regiment has good pathologic complete response and major pathologic response rates; however, the disease-free survival and overall survival outcomes require further follow-up.
• The TRG system and imaging assessment can be combined and employed to predict the efficacy of neoadjuvant therapy.
What is the implication, and what should change now?
• Neoadjuvant chemo-immunotherapy followed by surgery showed promising efficacy in the treatment of patients with locally advanced ESCC.
• Future studies should prioritize large-scale, biomarker-driven trials to determine whether immunotherapy should be integrated into standard neoadjuvant regimens and whether pathological complete response benefits can translate into improved long-term survival outcomes.
Introduction
Esophageal cancer is one of the most lethal malignant tumors worldwide (1). It is the seventh most common malignancy worldwide, and the sixth leading cause of cancer-related death worldwide (2). Surgery is one of the most effective ways to treat esophageal cancer. For newly diagnosed locally advanced esophageal cancer, the current treatment strategy is neoadjuvant therapy combined with surgery (3). Preoperative neoadjuvant treatments for esophageal cancer include chemotherapy and chemoradiotherapy, and extensive research of combination therapies associated with neoadjuvant immunotherapy is also being carried out.
Numerous studies have shown that neoadjuvant therapy significantly improves the survival of patients with a high pathological complete response (pCR) rate (4-7). The JCOG9907 study (5) reported that the pCR rate of neoadjuvant chemotherapy was 17.0%. Notable investigations into concurrent chemoradiotherapy include the CROSS trial (6) and the NEOCRTEC5010 trial (7), which have reported pCR rates of 49% and 43.2%, respectively. Nonetheless, the percentage of patients experiencing postoperative recurrence remains high, and the 5-year recurrence rate is 40–50% (8). Thus, the effectiveness of neoadjuvant therapy needs to be further improved, and ongoing clinical trials are actively exploring more advanced approaches for the treatment of esophageal cancer in the neoadjuvant setting.
Research on the use of neoadjuvant immunotherapy to treat esophageal cancer has yielded promising results (9). The CheckMate 577 study show that adjuvant immunotherapy after neoadjuvant treatment has demonstrated promising results in case of residual disease after neoadjuvant chemoradiotherapy (neoCRT) treatment followed by surgery with manageable toxicity profile (10). Initial findings also have shown that the combination of neoadjuvant chemotherapy and immunotherapy results in a superior tumor response compared to that of chemotherapy alone, and the combination therapy has the potential to increase the R0 resection rate. Thus, further investigations need to be conducted on this combination therapy.
Patients diagnosed with locally advanced, potentially resectable esophageal cancer face numerous unresolved issues related to preoperative neoadjuvant therapy, including the optimal number of cycles, drug selection, clinical effectiveness, safety, and postoperative complications. In light of this, we referred to various relevant domestic and international clinical studies, and chose the neoadjuvant treatment regimen of camrelizumab in combination with nab-paclitaxel and carboplatin (TC). We also analyzed the consistency between imaging efficacy assessment and postoperative tumor regression grades (TRGs) to gain valuable theoretical insights into the preoperative management of patients with locally advanced ESCC. We present this article in accordance with the STROBE reporting checklist (available at https://jtd.amegroups.com/article/view/10.21037/jtd-2025-215/rc).
Methods
Study subjects
Patients who were pathologically diagnosed with esophageal squamous cell carcinoma (ESCC) at The First Affiliated Hospital of Bengbu Medical University, the Hefei Cancer Hospital of Chinese Academy of Sciences, and The First Affiliated Hospital of Anhui Medical University, with the clinical stages of cT1–2N+M0, or cT3–4aN0, or N+M0, and non-cervical esophageal cancer from June 1, 2021 to June 1, 2023 were screened. All patients provided written informed consent before their enrollment in the study. This study was approved by the Ethics Committees of The First Affiliated Hospital of Bengbu Medical University (No. 2021KY014), the Hefei Cancer Hospital of Chinese Academy of Sciences (No. SL-KY2021-028), and The First Affiliated Hospital of Anhui Medical University (No. PJ2021-12-23). The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). In total, 106 patients were included in the study according to the inclusion and exclusion criteria. Detailed information about the medication was provided to the patients.
Inclusion criteria
To be eligible for inclusion in this study, the patients had to meet the following inclusion criteria: (I) meet the diagnostic criteria for ESCC after histopathological examination, and have ≥1 lesion that could be measured or evaluated based on imaging data [stage T1b–4bN + M0 or T3–4bN0M0 according to American Joint Committee on Cancer (AJCC) 8th criteria]; (II) have not received chemotherapy or radiotherapy in the past; (III) Have an Eastern Cooperative Oncology Group (ECOG) performance score of 0–1; (IV) have an expected survival time >3 months; and (V) have complete clinical data and follow-up data.
Exclusion criteria
Patients were excluded from the study if they met any of the following exclusion criteria: (I) had severe liver, kidney, cardiopulmonary, or other dysfunctions, or a poor basic condition; (II) had rheumatism or immune system diseases; (III) had severe endocrine system dysfunction or diseases; (IV) had severe infectious diseases or blood system diseases; (V) had allergies; (VI) had other malignant tumors; (VII) had a mental illness; (VIII) had poorly controlled hypertension or a risk of gastrointestinal bleeding; and/or (IX) were a pregnant or lactating woman.
Treatment
After admission, the patients underwent relevant examinations, and once it was clear that there were no contraindications to chemotherapy, the patients were informed of the treatment and the potential side effects. The treatment regimen is shown in Figure 1. Specifically, 200 mg of camrelizumab (Jiangsu Hengrui Pharmaceutical Co., Ltd., Suzhou, China, National Drug Approval No. S20190027) was intravenously administered, while nab-paclitaxel (260 mg/m2, D1, Shiyao Inc., Sijiazhuang, China) and carboplatin (area under curve of 5 mg/mL/min, D1, Qilu Inc., Jinan, China) on the first day. A treatment cycle comprised 21 days, and the efficacy evaluation was conducted after two cycles. In cases in which surgical resection remained challenging, an additional two cycles of treatment were considered. One hundred patients (94.3%) completed the planned 2 cycles; 6 patients (5.7%) received additional cycles (median 3 cycles, range, 3–4 cycles). All patients underwent surgery, and the postoperative TRG of each patient was documented. Adverse drug reactions were monitored by assessing each patient’s blood routine results before and after treatment. Further, any other adverse reactions occurring during treatment were closely observed and promptly managed.

Observe indicators
Efficacy evaluation
Each patient’s post-treatment efficacy was evaluated by reviewing the imaging data after two or four cycles, and each patient’s postoperative pathological TRG was also assessed. The preoperative efficacy evaluation was based on the Response Evaluation Criteria for Solid Tumors (RECIST) (version 1.1). A complete response (CR) was defined as the disappearance of all target lesions, and a reduction to <10 mm in the short axis of all pathological lymph nodes; a partial response (PR) was defined as a 30% reduction in the sum of the diameters of all target lesions, and no new lesions; stable disease (SD) was defined as neither the response nor progression criteria being met; progressive disease (PD) was defined as the appearance of new lesions after treatment, or a 20% increase in the sum of the diameters of all target lesions. The objective response rate (ORR) was calculated as follows: ORR = (CR + PR)/total case number × 100%. While the disease control rate (DCR) was calculated as follows: DCR = (CR + PR + SD)/total case number × 100%.
The postoperative TRG was based on the National Comprehensive Cancer Network standards. Specifically, grade 0 indicated no residual tumor present (complete regression of the tumor tissue); grade 1 indicated <10% residual tumor present (near complete regression); grade 2 indicated 10–50% residual carcinoma present (partial regression); grade 3 indicated >50% residual tumor present (minimal/no regression).
Toxicity and side effects
Adverse reactions were assessed based on the World Health Organization’s criteria for grading the toxicity of anticancer drugs, which categorizes reactions into the following five levels: 0 (none), I (mild), II (moderate), III (severe), and IV (life threatening). Generally, grades III to IV are indicative of severe side effects.
All patients underwent assessments of their blood profiles, biochemical parameters, and other relevant tests before and after each treatment cycle. By comparing the changes in test results before and after treatment, and by monitoring the patients’ well-being post-discharge, the adverse effects and toxicity experienced by the patients were documented. The surgical procedures included lymph node dissection, transthoracic esophageal dissection, construction of the gastric tube, stomach mobilization, and left cervical esophagogastric anastomosis. Open and video-assisted minimally invasive esophagectomy techniques were used.
Statistical analysis
All categorical variables were compared using the Chi-squared or Fisher’s exact test. Comparisons between patients with a pathologic/radiologic response and non-response were performed using Pearson’s Chi-squared test for the categorical variables and the Wilcoxon rank-sum test for the continuous variables. A P value <0.05 indicated a statistically significant difference. SPSS 21 software (IBM Corp., Armonk, NY, USA) was used for the statistical analysis.
Results
Clinical characteristics of the patients
The clinical data of the 106 patients with locally advanced ESCC were collected, including their sex, age, tumor location, stage, smoking history, drinking history, and ECOG score. Detailed clinical information is shown in Table 1.
Table 1
| Clinical characteristics | Group | Case number | Percentage (%) |
|---|---|---|---|
| Sex | Male | 93 | 87.7 |
| Female | 13 | 12.3 | |
| Age (years) | ≥65 | 60 | 56.6 |
| <65 | 46 | 43.4 | |
| Tumor location | Upper | 8 | 7.6 |
| Middle | 34 | 32 | |
| Lower | 64 | 60.4 | |
| Eastern Cooperative Oncology Group score | 2 | 98 | 92.5 |
| >2 | 8 | 7.5 | |
| T stage | T1–2 | 50 | 47.1 |
| T3–4 | 56 | 52.9 | |
| N stage | N0 | 5 | 4.8 |
| N+ | 101 | 95.2 | |
| Smoking history | Yes | 65 | 61.3 |
| No | 41 | 38.7 | |
| Drinking history | Yes | 72 | 68 |
| No | 34 | 32 |
Effectiveness analysis (preoperative efficacy evaluation and postoperative TRG)
In total, 106 (100%) patients successfully completed treatment. All patients received surgery. The pCR (ypT0N0) rate was 33% (35/106), and 50% (53/106) of the patients showed a CR of the primary tumor, and only had residual disease in the lymph nodes (ypT0N+). The major pathologic response (MPR) rate, which was defined as the presence of <10% of residual cancer cells in the primary tumor surgical specimen, was 49% (52/106). According to the RECIST (version 1.1), the preoperative imaging evaluation of the patients showed that 30.2% (32/106) had a CR, 33% (35/106) had a PR, 30.2% (32/106) had SD, and 6.6% (7/106) had PD.
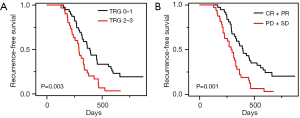
In the post-hoc analysis, we also evaluated the relationship between the baseline characteristics and treatment response (Table 2). No significant associations were found in relation to the patient factors [i.e., age, sex, cigarette-smoking, drinking history, T stage (pT), N stage (pN), and primary location]; however, an association was found between the TRG and pathologic grade after surgery reduction, and the pre-surgery treatment response (P=0.005 and P<0.001). In terms of the postoperative TRG, 14.15% of the patients (15/106) had grade 0, 48.11% (51/106) had grade 1, 27.36% (29/106) had grade 2, and 10.38% (11/106) had grade 3. The ORR obtained from the preoperative computed tomography (CT) evaluation was significantly negatively correlated with the pathological TRG (R=−0.63, P=0.001). The patients who achieved a MPR had a significantly higher 2-year recurrence-free survival (RFS) rate (77.1% vs. 45.9%; P=0.003) (Figure 2).
Table 2
| Characteristics | CR (n=32) | PR (n=35) | PD (n=7) | SD (n=32) | P value |
|---|---|---|---|---|---|
| Age (years) | 68.7 [55–82] | 65.8 [46–81] | 64.4 [55–74] | 67.2 [44–84] | 0.51 |
| Sex | 0.48 | ||||
| Male | 28 (87.50) | 29 (82.86) | 5 (71.43) | 26 (81.25) | |
| Female | 4 (12.50) | 6 (17.14) | 2 (28.57) | 6 (18.75) | |
| Cigarette-smoking history | 0.50 | ||||
| Never | 12 (37.50) | 18 (51.43) | 4 (57.14) | 15 (46.87) | |
| Former | 20 (62.50) | 17 (48.57) | 3 (42.86) | 17 (53.13) | |
| Drinking history | 0.49 | ||||
| Never | 9 (28.13) | 12 (34.29) | 2 (28.57) | 8 (25.00) | |
| Former | 23 (71.87) | 23 (65.71) | 5 (71.43) | 24 (75.00) | |
| Tumor | 0.85 | ||||
| T1 | 0 | 0 | 1 (14.29) | 1 (3.12) | |
| T2 | 20 (62.50) | 10 (28.57) | 3 (42.86) | 14 (43.75) | |
| T3 | 12 (37.50) | 25 (71.43) | 2 (28.57) | 17 (53.13) | |
| T4 | 0 | 0 | 1 (14.29) | 0 | |
| Node | 0.83 | ||||
| N0 | 0 | 0 | 0 | 0 | |
| N1 | 3 (9.38) | 5 (14.28) | 0 | 8 (25.00) | |
| N2 | 17 (53.12) | 18(51.43) | 1 (14.29) | 10 (31.25) | |
| N3 | 12(37.50) | 12 (34.29) | 6 (85.71) | 14 (43.75) | |
| Primary location | 0.17 | ||||
| Lower | 22 (68.75) | 26 (77.14) | 6 (85.71) | 26 (81.25) | |
| Middle | 9 (28.13) | 8 (22.86) | 1 (14.29) | 6 (18.75) | |
| Upper | 1 (3.12) | 1 | 0 | 0 | |
| TRG | 0.46 | ||||
| 0 | 9 (28.13) | 2 (5.71) | 0 | 4 (12.50) | |
| 1 | 14 (43.75) | 21(60.00) | 2 (28.57) | 14 (43.75) | |
| 2 | 5 (15.62) | 12 (34.29) | 4 (57.14) | 8 (25.00) | |
| 3 | 4 (12.50) | 0 | 1 (14.29) | 6 (18.75) | |
| Pathologic grade | <0.001 | ||||
| 0 | 30 (93.75) | 2 (5.71) | 0 | 3 (9.37) | |
| 1 | 2 (6.25) | 28 (80.00) | 1 (14.29) | 3 (9.37) | |
| 2 | 0 | 5 (14.29) | 0 | 26 (81.25) | |
| 3 | 0 | 0 | 6 (85.71) | 0 |
Data are presented as median [range] or n (%). A P value of less than <0.05 was considered significant. RECIST, Response Evaluation Criteria for Solid Tumors; CR, complete response; PR, partial response; PD, progressive disease; SD, stable disease; TRG, tumor regression grade.
Subgroup analysis: extended vs. standard cycles
The extended-cycle group showed no significant improvement in pathological response (TRG 0–1: 45% vs. 33.3%, TRG 2–4: 55% vs. 66.7%, P=0.27), suggesting that prolonged therapy may not overcome intrinsic tumor resistance. All extended-cycle patients achieved R0 resection despite anatomical challenges, supporting the role of additional cycles in enhancing resectability for borderline cases. Higher grade ≥3 treatment-related adverse events (TRAEs) in the extended group (33.3% vs. 15%, P=0.15) highlight the need for careful patient selection (Table 3).
Table 3
| Parameter | cycle group (n=100) | Extend-cycle group (n=6) | P value |
|---|---|---|---|
| Surgical response | 0.27 | ||
| TRG 0–1 | 45% (45/100) | 33.3% (2/6) | |
| TRG 2–4 | 55% (55/100) | 66.7% (4/6) | |
| Surgical outcome | – | ||
| R0 | 100% (100/100) | 100% (6/6) | |
| Safety | |||
| Grade >3 TRAEs | 15.0% (15/100) | 33.3% (2/6) | 0.15 |
A P value of less than <0.05 was considered significant. TRG, tumor regression grade; TRAE, treatment-related adverse event; R0, resection with microscopically negative margins.
Adverse reactions and safety
All the patients experienced varying degrees of toxicity and side effects. Common adverse reactions included hypothyroidism (29.2%, 31/106), fever (28.3%, 30/106), gastrointestinal reactions (35.8%, 38/106), fatigue (40%, 36/106), liver function damage (22.6%, 24/106), and skin adverse reactions (20.8%, 22/106). All of the adverse reactions were grade I and II, and no patients died from adverse reactions. The adverse reactions are shown in Table 4. Moreover, surgery-related complications mainly included pneumonia (n=5.7%, 6/106), chylemia (n=3.8%, 4/106), and wound infection (n=2.8%, 3/106). No fatal adverse events (AEs) occurred during the 30–90-day of follow-up.
Table 4
| Adverse reactions | I | II | III | IV | Total |
|---|---|---|---|---|---|
| Hypothyroidism | 21 | 6 | 4 | 0 | 31 |
| Fever | 16 | 9 | 3 | 2 | 30 |
| Gastrointestinal reactions | 26 | 7 | 5 | 0 | 38 |
| Anemia | 6 | 1 | 1 | 0 | 8 |
| Fatigue | 20 | 16 | 0 | 0 | 36 |
| Hyperlipidemia | 6 | 2 | 1 | 0 | 9 |
| Liver function damage | 14 | 6 | 2 | 2 | 24 |
| Reactive cutaneous capillary endothelial proliferation | 15 | 4 | 3 | 0 | 22 |
| Cardiotoxicity | 1 | 2 | 0 | 0 | 3 |
Discussion
With the emergence of the immunotherapy era, the treatment landscape for ESCC has transformed significantly. Presently, the combination of immunotherapy and chemotherapy in the treatment of locally advanced ESCC represents a popular area of research. Previous studies have shown that the response rate to chemotherapy in combination with immunotherapy generally surpasses that of chemotherapy alone, illustrating their synergistic effects in increasing treatment sensitivity (11,12). The KEYSTONE-001 study (13) reported that 47 patients with locally advanced ESCC received three cycles of pembrolizumab plus chemotherapy, and received surgery. Among 46 patients studied for efficacy, the MPR and pCR rates were 72% and 41%, respectively. These results highlight the potent anti-tumor activity and favorable safety profile of anti-programmed cell death 1 (PD-1) immunotherapy in combination with chemotherapy during neoadjuvant treatment for patients with locally advanced resectable ESCC. The ESPRIT study (14) is investigating the efficacy and safety of camrelizumab in combination with chemotherapy in the neoadjuvant treatment of ESCC. The preliminary findings of the study include that among the 11 patients who underwent surgical intervention, the pCR rate was 45.5% (5 out of 11), and it was independent of the patients’ programmed death-ligand 1 (PD-L1) expression. Further, the pT0 rate was 54.5% (6 of 11), and the R0 resection rate was 100%.
The combination of camrelizumab with paclitaxel and nedaplatin has shown good tolerability as a neoadjuvant therapy, and exhibited the ability to reduce tumor size and stage. The KEEP-G 03 study (15) evaluated the feasibility and safety of sintilimab combined with three-drug chemotherapy in the neoadjuvant treatment of resectable ESCC. Fifteen patients completed surgery, and had MPR and pCR rates of 50.0% (15/30) and 20.0% (6/30), and a R0 resection rate of 100%. The phase-II TD-NICE study (16) used tislelizumab combined with chemotherapy as the neoadjuvant treatment for resectable ESCC. In the study, 36 patients underwent surgery, and had a R0 resection rate of 80.5%, a MPR of 72% (25/36), and a pCR rate of 50% (18/36). The study also proposed for the first time that a tumor cell proportion score >1 was a more accurate predictive indicator of pCR than the combined positive score. A sequencing analysis showed that HLA-A02 gene mutations may be related to pCR.
In the present study, camrelizumab combined with nab-paclitaxel and carboplatin was used as the neoadjuvant regimen for locally advanced ESCC, and 106 patients underwent surgery. Of the patients, 39.6% (42/106) had a pCR (ypT0N0), 50% (53/106) had a CR to the primary tumor and only had residual disease in the lymph nodes (ypT0N+), and the MPR rate was 49% (52/106). Moreover, our study suggests that CT-based ORR and TRG may serve as a non-invasive predictor in neoadjuvant-treated ESCC. The safety profile included chemotherapy-related toxicity and immune-related toxicity. No new toxic reactions occurred, and the overall tolerability was acceptable. Thus, the neoadjuvant immuno-chemotherapy regimen was effective and safe.
Due to the close association between the treatment response and prognosis, the accurate evaluation of the response to neoadjuvant therapy is crucial (17). Currently, various methods are used to assess the effectiveness of neoadjuvant therapy. The RECIST is the gold standard for appraising the impact of therapy on solid tumors, but it requires the presence of measurable lesions (18). The latest version of the RECIST (version 1.1) requires the presence of measurable lesions with a diameter of ≥10 mm and a short axis of a single metastatic lymph node ≥15 mm (19). However, the diameters of esophageal cancer lesions and metastatic lymph nodes following neoadjuvant treatment often do not align with the measurement requirements of the RECIST. Further, the RECIST itself acknowledges the reproducibility limitation when measuring primary tumors of the digestive tract. Consequently, to assess the efficacy of neoadjuvant therapy in ESCC, the RECIST may not be the most suitable method.
There is substantial evidence that the TRG system may provide a more accurate assessment approach from a histopathological standpoint. Indeed, studies have shown that the TRG is an independent prognostic indicator of patient survival (20-23). The prognostic dominance of TRG over traditional biomarkers may stem from its integrative reflection of tumor-immune interactions. For instance, a study revealed that high TRG correlates with CD8+ T-cell clonal expansion and tertiary lymphoid structure formation, suggesting immune-mediated tumor eradication as a survival driver (24). Some studies have revealed that results obtained using the RECIST often tend to overestimate treatment effects, and that pathological assessment remains the most reliable means of evaluating residual tumor activity (25-27).
Further, research has established a strong association between the pathological response and postoperative overall survival (OS) or disease-free survival (DFS) (28). The present study found a significant relationship between the ORR in preoperative CT efficacy assessments and the pathological TRG, suggesting that imaging-based regression evaluation following neoadjuvant therapy can predict the pathological TRG to a certain extent (27). Notably, we still observed inconsistencies between the two assessment methods. This discrepancy may be attributed to the fact that the current pathological TRG system primarily focuses on the primary tumor and does not include lymph node evaluation, while imaging assessment considers changes in both the primary tumor and lymph nodes. As a result, there is a degree of variance between imaging and pathological evaluation, and combining the two could be more accurate in predicting tumor treatment responses.
This study had several limitations. First, all the patients included in this study had ESCC. Since ESCC and adenocarcinoma exhibit distinct epidemiological profiles, lesion sites, and disease progressions, it is challenging to extrapolate these findings to patients with adenocarcinoma. Second, endoscopic ultrasound was not applied in the study for more precise clinical T staging, and only a subset of patients underwent pre-surgery positron emission tomography (PET)/CT scans. In one patient, the clinical CR showed by preoperative PET/CT was consistent with the postoperative pCR. Third, prolonged treatment cycles (5.7% of patients received ≥3 cycles) improved the R0 resection rate but not TRG, suggesting that development of individualized regimen decision-making models based on the dynamic clearance of ctDNA or characteristics of the tumor microenvironment (TME) is needed. Furthermore, some unmeasurable factors influencing treatment efficacy may have affected the study’s results. Due to the study’s small sample size and relatively short follow-up period, tumor recurrence rates and 3-year, 5-year and even longer term survival data remains unclear.
However, there are still many issues that need to be further discussed in this study. Compared to the global standard of CROSS regimen (radiotherapy combined with surgery) (6), our chemoimmunotherapy regimen demonstrated the following potential advantages. First, comparable pathologic remission and surgical outcomes: a PCR rate of 39.6% (vs. 23.1% in CROSS) and an R0 resection rate of 97.2% (vs. 92% in CROSS) despite not combining radiotherapy, suggesting that immunotherapy may enhance local control by activating systemic anti-tumor immunity. Especially for patients with complex anatomy (e.g., aortic or tracheal involvement) or large lymph node metastases (cN3), chemoimmunotherapy may avoid radiotherapy-induced perivascular fibrosis and reduce the difficulty of surgery, as well as reduce the risk of distant recurrence through the immune-memory effect. Second, chemoimmunotherapy has no risk of radiation pneumonitis or esophagitis, providing a safer treatment option for patients with comorbid cardiorespiratory underlying disease. And last but not least, although PD-L1 was not routinely detected in this study, recent studies have suggested that the high tumor mutation burden (TMB) and PD-L1 positivity rate may predict a superior population for chemoimmunotherapy, which warrants future prospective validation (29-31).
This study found that neoadjuvant immuno-chemotherapy was both safe and effective in managing ESCC, and the treatment-related side effects were controllable and tolerable. Presently, challenges remain in determining the optimal neoadjuvant treatment strategy for locally advanced resectable esophageal cancer. Various combinations (e.g., chemotherapy with immunotherapy, radio-chemotherapy with immunotherapy, and targeted therapy with immunotherapy) have been investigated. Further research is required to determine whether the increased pCR rate resulting from neoadjuvant therapy results in long-term survival benefits for patients. Additionally, the identification of biomarkers to predict the effectiveness of neoadjuvant therapy and the selection of suitable candidates for this approach also requires further research. Thus, future research should seek to: (I) investigate the factors that affect patient response to immune checkpoint-based therapy; (II) identify predictive biomarkers for the checkpoint inhibitor-based immunotherapy of cancer patients; and (III) determine the effective duration of neoadjuvant immunotherapy, the cycles of neoadjuvant immunotherapy, the influence of the TME in the response to immunotherapy, the role of ethnicity, and how to surmount resistance to immunotherapy, and so on. We believe that gaining an understanding of the pathogenic mechanism of ESCC by multi-omics analysis and different perspectives will improve the clinical benefits for patients and enable precise treatment.
Conclusions
In conclusion, our analysis presented favorable pathological responses and promising outcome of neoadjuvant chemoimmunotherapy for locally advanced resectable ESCC. The treatment increased the likelihood of achieving a curative surgical outcome, and the side effects were generally manageable. Moreover, the TRG was significantly associated with imaging assessment, and the RFS of the TRG 0–1 group was significantly better than that of the TRG 2–3 group. These two approaches can be combined and employed to predict the efficacy of neoadjuvant immuno-chemotherapy.
Acknowledgments
We would like to thank the pathologists and clinical research associates from The First Affiliated Hospital of Bengbu Medical University, Hefei Cancer Hospital, Chinese Academy of Sciences, and The First Affiliated Hospital of Anhui Medical University for their contribution to patient enrollment and sample collection.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://jtd.amegroups.com/article/view/10.21037/jtd-2025-215/rc
Data Sharing Statement: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-2025-215/dss
Peer Review File: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-2025-215/prf
Funding: This work was supported by grants from
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jtd.amegroups.com/article/view/10.21037/jtd-2025-215/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All patients provided written informed consent before their enrollment in the study. This study was approved by the Ethics Committees of The First Affiliated Hospital of Bengbu Medical University (No. 2021KY014), the Hefei Cancer Hospital of Chinese Academy of Sciences (No. SL-KY2021-028), and The First Affiliated Hospital of Anhui Medical University (No. PJ2021-12-23). The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013).
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Guo D, Jin J, Li D, et al. Analysis of the incidence and mortality trends of esophageal cancer in cancer registry areas of China and Japan. Int J Cancer 2024;155:1376-86. [Crossref] [PubMed]
- He F, Wang J, Liu L, et al. Esophageal cancer: trends in incidence and mortality in China from 2005 to 2015. Cancer Med 2021;10:1839-47. [Crossref] [PubMed]
- Li B, Chen H. The Best Surgery Should Be Applied for Locally Advanced Esophageal Cancer. J Clin Oncol 2021;39:3189-90. [Crossref] [PubMed]
- Lewis S, Lukovic J. Neoadjuvant Therapy in Esophageal Cancer. Thorac Surg Clin 2022;32:447-56. [Crossref] [PubMed]
- Ando N, Kato H, Igaki H, et al. A randomized trial comparing postoperative adjuvant chemotherapy with cisplatin and 5-fluorouracil versus preoperative chemotherapy for localized advanced squamous cell carcinoma of the thoracic esophagus (JCOG9907). Ann Surg Oncol 2012;19:68-74. [Crossref] [PubMed]
- Shapiro J, van Lanschot JJB, Hulshof MCCM, et al. Neoadjuvant chemoradiotherapy plus surgery versus surgery alone for oesophageal or junctional cancer (CROSS): long-term results of a randomised controlled trial. Lancet Oncol 2015;16:1090-8. [Crossref] [PubMed]
- Liu S, Wen J, Yang H, et al. Recurrence patterns after neoadjuvant chemoradiotherapy compared with surgery alone in oesophageal squamous cell carcinoma: results from the multicenter phase III trial NEOCRTEC5010. Eur J Cancer 2020;138:113-21. [Crossref] [PubMed]
- Eyck BM, van Lanschot JJB, Hulshof MCCM, et al. Ten-Year Outcome of Neoadjuvant Chemoradiotherapy Plus Surgery for Esophageal Cancer: The Randomized Controlled CROSS Trial. J Clin Oncol 2021;39:1995-2004. [Crossref] [PubMed]
- Doki Y, Ajani JA, Kato K, et al. Nivolumab Combination Therapy in Advanced Esophageal Squamous-Cell Carcinoma. N Engl J Med 2022;386:449-62. [Crossref] [PubMed]
- Kelly RJ, Ajani JA, Kuzdzal J, et al. Adjuvant Nivolumab in Resected Esophageal or Gastroesophageal Junction Cancer. N Engl J Med 2021;384:1191-203. [Crossref] [PubMed]
- Hirano H, Kato K. Systemic treatment of advanced esophageal squamous cell carcinoma: chemotherapy, molecular-targeting therapy and immunotherapy. Jpn J Clin Oncol 2019;49:412-20. [Crossref] [PubMed]
- Soeratram TT, Creemers A, Meijer SL, et al. Tumor-immune landscape patterns before and after chemoradiation in resectable esophageal adenocarcinomas. J Pathol 2022;256:282-96. [Crossref] [PubMed]
- Shang X, Zhao G, Liang F, et al. Safety and effectiveness of pembrolizumab combined with paclitaxel and cisplatin as neoadjuvant therapy followed by surgery for locally advanced resectable (stage III) esophageal squamous cell carcinoma: a study protocol for a prospective, single-arm, single-center, open-label, phase-II trial (Keystone-001). Ann Transl Med 2022;10:229. [Crossref] [PubMed]
- Ma J, Zhang J, Yang Y, et al. Camrelizumab combined with paclitaxel and nedaplatin as neoadjuvant therapy for locally advanced esophageal squamous cell carcinoma (ESPRIT): A phase II, single-arm, exploratory research. Journal of Clinical Oncology 2021;39:e16033.
- Li Z, Xu PF, Mao WM, et al. A study of neoadjuvant sintilimab combined with chemotherapy TP for locally advanced esophageal squamous cell carcinoma (ESCC). Journal of Clinical Oncology 2022;40:e16038.
- Yan X, Duan H, Ni Y, et al. Tislelizumab combined with chemotherapy as neoadjuvant therapy for surgically resectable esophageal cancer: A prospective, single-arm, phase II study (TD-NICE). Int J Surg 2022;103:106680. [Crossref] [PubMed]
- Gao L, Hong ZN, Wu L, et al. Residual tumor model in esophageal squamous cell carcinoma after neoadjuvant immunochemotherapy: Frequently involves the mucosa and/or submucosa. Front Immunol 2022;13:1008681. [Crossref] [PubMed]
- Leong WL. From RECIST to PERCIST: navigating the landscape of tumor response assessment. Eur Radiol 2024;34:3656-8. [Crossref] [PubMed]
- Berz AM, Dromain C, Vietti-Violi N, et al. Tumor response assessment on imaging following immunotherapy. Front Oncol 2022;12:982983. [Crossref] [PubMed]
- Hagi T, Makino T, Yamasaki M, et al. Pathological Regression of Lymph Nodes Better Predicts Long-term Survival in Esophageal Cancer Patients Undergoing Neoadjuvant Chemotherapy Followed by Surgery. Ann Surg 2022;275:1121-9. [Crossref] [PubMed]
- Tang YL, Li DD, Duan JY, et al. Prognostic analysis of rectal cancer patients after neoadjuvant chemoradiotherapy: different prognostic factors in patients with different TRGs. Int J Colorectal Dis 2024;39:93. [Crossref] [PubMed]
- Tang YL, Li DD, Duan JY, et al. Prognostic analysis of rectal cancer patients after neoadjuvant chemoradiotherapy: different prognostic factors in patients with different TRGs. Int J Colorectal Dis 2024;39:93. [Crossref] [PubMed]
- Luo D, Chen Y, Luo Z, et al. Prognostic Value of Tumor Regression Grade Combined with Pathological Lymph Node Status in Initially Node-Positive Rectal Cancer Treated with Neoadjuvant Chemoradiotherapy. J Invest Surg 2025;38:2449669. [Crossref] [PubMed]
- Xu Y, Lou X, Liang Y, et al. Predicting Neoadjuvant Chemoradiotherapy Response in Locally Advanced Rectal Cancer Using Tumor-Infiltrating Lymphocytes Density. J Inflamm Res 2021;14:5891-9. [Crossref] [PubMed]
- Lai MY, Kang SY, Sun YT, et al. Comparison of response evaluation criteria in solid tumors and tumor regression grade in evaluating the effect of preoperative systemic therapy of gastric cancer. BMC Cancer 2022;22:1031. [Crossref] [PubMed]
- Qu J, Zhang Y, Lu S, et al. Quantitative RECIST derived from multiparametric MRI in evaluating response of esophageal squamous cell carcinoma to neoadjuvant therapy. Eur Radiol 2022;32:7295-306. [Crossref] [PubMed]
- Tsagkalidis V, Blaszczyk MB, In H. Interpretation of Tumor Response Grade following Preoperative Therapy for Gastric Cancer: An Overview. Cancers (Basel) 2023;15:3662. [Crossref] [PubMed]
- Abboretti F, Mantziari S, Didisheim L, et al. Prognostic value of tumor regression grade (TRG) after oncological gastrectomy for gastric cancer. Langenbecks Arch Surg 2024;409:199. [Crossref] [PubMed]
- Yarchoan M, Albacker LA, Hopkins AC, et al. PD-L1 expression and tumor mutational burden are independent biomarkers in most cancers. JCI Insight 2019;4:e126908. [Crossref] [PubMed]
- Jeong AR, Trando AH, Thomas SD, et al. Higher tumor mutational burden and PD-L1 expression correlate with shorter survival in hematologic malignancies. Ther Adv Med Oncol 2024;16:17588359241273053. [Crossref] [PubMed]
- Muquith M, Espinoza M, Elliott A, et al. Tissue-specific thresholds of mutation burden associated with anti-PD-1/L1 therapy benefit and prognosis in microsatellite-stable cancers. Nat Cancer 2024;5:1121-9. [Crossref] [PubMed]
(English Language Editor: L. Huleatt)


